Cyclisation strategies for stabilising peptides with irregular conformations
Abstract
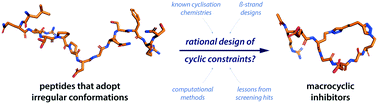
Cyclisation is a common synthetic strategy for enhancing the therapeutic potential of peptide-based molecules. While there are extensive studies on peptide cyclisation for reinforcing regular secondary structures such as α-helices and β-sheets, there are remarkably few reports of cyclising peptides which adopt irregular conformations in their bioactive target-bound state. In this review, we highlight examples where cyclisation techniques have been successful in stabilising irregular conformations, then discuss how the design of cyclic constraints for irregularly structured peptides can be informed by existing β-strand stabilisation approaches, new computational design techniques, and structural principles extracted from cyclic peptide library screening hits. Through this analysis, we demonstrate how existing peptide cyclisation techniques can be adapted to address the synthetic design challenge of stabilising irregularly structured binding motifs.
- This article is part of the themed collection: Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...