Cyclohexene esterification–hydrogenation for efficient production of cyclohexanol†
Abstract
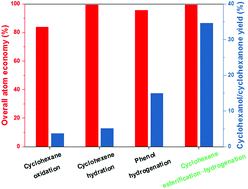
A novel process based on cyclohexene esterification–hydrogenation for the production of cyclohexanol, the key intermediate in the production of ε-caprolactam, was devised and validated for the first time. In this process, cyclohexene obtained from the partial hydrogenation of benzene is esterified with acetic acid to cyclohexyl acetate, followed by hydrogenation to cyclohexanol. The experimentally determined equilibrium conversion of cyclohexene for cyclohexene esterification at the stoichiometric ratio is always ≥68% in the temperature range of 333–373 K over the commercial Amberlyst 15 catalyst, which is substantially higher than that of cyclohexene hydration. The apparent activation energy (Ea) for the esterification of cyclohexene with acetic acid is 60.0 kJ mol−1, which is lower than that of cyclohexene hydration. In the hydrogenation of cyclohexyl acetate to cyclohexanol, high conversion of 99.5% and high selectivity of 99.7% are obtained on the La-promoted Cu/ZnO/SiO2 catalyst prepared by the co-precipitation method. This process shows both a high overall atom economy of 99.4% comparable to that of the cyclohexene hydration process and a much higher catalytic efficiency than the phenol hydrogenation process. On the basis of the above fundamental works, a pilot-scale demonstration unit with a capacity of 8000 tonnes per annum was developed and operated smoothly for more than 1000 h with no indication of deactivation.


 Please wait while we load your content...
Please wait while we load your content...