Covalent and non-covalent interactions of cyanidin-3-O-glucoside with milk proteins revealed modifications in protein conformational structures, digestibility, and allergenic characteristics†
Abstract
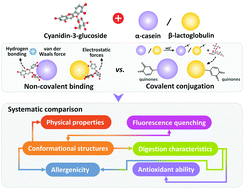
Currently, there is a need to explore the effects of different types of protein–anthocyanin complexations, as well as the possible changes in the nutrition and allergenicity of the formed complexes. Here, we systematically investigated the covalent and non-covalent interactions between cyanidin-3-O-glucoside (C3G) and two major milk proteins, α-casein (α-CN) and β-lactoglobulin (β-LG). Fluorescence quenching data showed that, under non-covalent conditions, C3G quenched the fluorescence of the two proteins via a static process, with the interaction forces being revealed; for covalent products, decreased fluorescence intensities were observed with red shifts in the λmax. Multiple spectroscopic analyses implied that C3G-addition induced protein structural unfolding through transitions between the random coil and ordered secondary components. With a two-stage simulated gastrointestinal (GI) digestion model, it was seen that covalent complexes, not their non-covalent counterparts, showed reduced protein digestibility, ascribed to structural changes resulting in the unavailability of enzyme cleaving sites. The GI digests displayed prominent 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) radical cation-scavenging abilities (3.8–11.1 mM Trolox equivalents per mL digest), in contrast to the markedly reduced 1,1-diphenyl-2-picrylhydrazyl radical-scavenging capacities. Additionally, covalent protein–C3G complexes, but not their non-covalent counterparts, showed lower IgE-binding levels in comparison to the native control. This study provides new understanding for the development of anthocyanin–milk protein systems as functional ingredients with health-beneficial properties.


 Please wait while we load your content...
Please wait while we load your content...