Polymorphs and hydrates of the anticancer drug erlotinib: X-ray crystallography, phase transition and biopharmaceutical studies†
Abstract
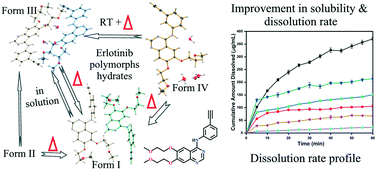
Erlotinib, a non-small cell lung cancer BCS class II drug, was found to occur as two polymorphs and two hydrates depending on the crystallization conditions. The monohydrate (form III), which has been reported in patents and publications, is the preferred crystalline phase from solution crystallization. The other forms (polymorphs, forms I and II, and a trihydrate, form IV) are solvent and condition-specific. Form I was exclusively and reproducibly obtained from dried non-polar solvents (viz. toluene, benzene, and xylene) using a solution crystallization method under controlled conditions. In contrast, form II was obtained by the rotary evaporation technique under reduced pressure from polar solvents (viz. dichloromethane, ethyl acetate, acetone, and acetonitrile). Slow evaporation from polar and non-polar solvents under open conditions yielded form III crystals, whereas evaporation from acetone–acetonitrile–water mixtures yielded form IV crystals. DSC and variable temperature PXRD studies revealed form I to be the most stable phase while the other solid forms displayed thermally induced polymorphic transitions into form I crystals. Further, form I and form III are found to be stable over the 30–90% RH range. Further, form I displayed solution-mediated transformation into form III (monohydrate) when left to stand in the mother liquor for a longer duration (1–2 days). Similarly, the unstable form IV crystals transformed into the stable form III crystals within 1–2 h when exposed to an open atmosphere. Fast crystallization from polar solvents using the rotary evaporation technique enabled us to capture the metastable polymorph, form II. Early separation of form I from its mother liquor prevented its solution-mediated transformation into the monohydrate, form III. The water activity study revealed that form III is the most preferred solid phase in the presence of water. Dissolution rate measurements showed that the rate of form II is almost comparable to that of the marketed erlotinib hydrochloride. In contrast, for the other forms, the dissolution profiles showed a considerable decrease. Although the crystal structures of form I and form III have been published previously, this manuscript gives a comprehensive overview of the free base solid forms of erlotinib, the phase transitions amongst them and their biopharmaceutical properties.
- This article is part of the themed collection: Supramolecular & Polymorphism


 Please wait while we load your content...
Please wait while we load your content...