Inhibiting the shuttle effect using artificial membranes with high lithium-ion content for enhancing the stability of the lithium anode†
Abstract
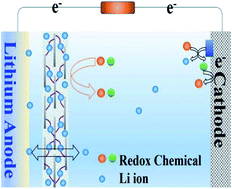
The low cycle stability of the lithium anode has become one of the bottlenecks restricting the development of lithium-metal batteries with high theoretical energy density. Serious side reactions between lithium and electrolyte components are one of the key reasons for the poor cycle stability of the lithium anode. Herein, lithiated graphene oxide (GO-Li) and lithium poly(styrene sulfate) (PSS-Li) are used to construct composite membranes for the protection of the Li-anode, which shows long-term operation over 1000 h in Li‖Li symmetric cells in the presence of redox chemicals that accelerate the cathodic reaction. The high content of Li+ of PSS-Li can not only inhibit the dissolution and diffusion of redox molecules in the membrane, but also improve the Li+ transport rate through the membrane. In our study, we take a lithium–oxygen (Li–O2) battery as the model device and 2,2,6,6-tetramethyl-1-piperidinyloxy as the model redox chemical to accelerate the cathodic reaction. Compared with conventional membranes, artificial membranes can effectively inhibit the side reaction between the redox molecules and the lithium anode. Consequently, the energy efficiency and cycle stability (over three times) of Li–O2 batteries are greatly improved. This provides an important theoretical basis and technical support for the design and preparation of membranes for high performance energy-conversion batteries.


 Please wait while we load your content...
Please wait while we load your content...