A multiscale biophysical model for the recruitment of actin nucleating proteins at the membrane interface†
Abstract
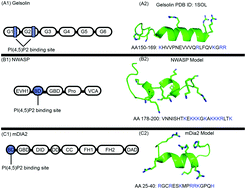
The dynamics and organization of the actin cytoskeleton are crucial to many cellular events such as motility, polarization, cell shaping, and cell division. The intracellular and extracellular signaling associated with this cytoskeletal network is communicated through cell membranes. Hence the organization of membrane macromolecules and actin filament assembly are highly interdependent. Although the actin-membrane linkage is known to happen through many routes, the major class of interactions is through the direct interaction of actin-binding proteins with the lipid class containing poly-phosphatidylinositols (PPIs). Among the PPIs, phosphatidylinositol bisphosphate (PI(4,5)P2) acts as a significant factor controlling actin polymerization in the proximity of the membrane by binding to actin-associated proteins. The molecular interactions between these actin-binding proteins and the membrane lipids remain elusive. Here, using molecular modeling, analytical theory, and experimental methods, we investigate the binding of three different actin-binding proteins, mDia2, NWASP, and gelsolin, to membranes containing PI(4,5)P2 lipids. We perform molecular dynamics simulations on the protein-bilayer system and analyze the membrane binding in the form of hydrogen bonds and salt bridges at various PI(4,5)P2 and cholesterol concentrations. Our experimental study with PI(4,5)P2-containing large unilamellar vesicles mimics the computational experiments. Using the multivalencies of the proteins obtained in molecular simulations and the cooperative binding mechanisms of the proteins, we also propose a multivalent binding model that predicts the actin filament distributions at various PI(4,5)P2 and protein concentrations.


 Please wait while we load your content...
Please wait while we load your content...