Polymorphism of asymmetric catalysts based on amphiphilic lipopeptides in solution†
Abstract
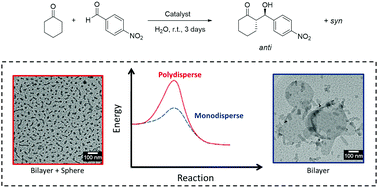
The self-assembly of model [P]RWG lipopeptides (P: L-proline, R: L-arginine, W: L-tryptophan, G: L-glycine), containing one or two aliphatic octadecyl (C18) chains in water and cyclohexanone/water solutions was examined. The self-assembly of mixtures of these RWG and PRWG lipopeptides was also investigated. These materials presented a similar critical aggregation concentration of ∼4.0 × 10−4 wt% and were characterized by unordered secondary structures with some β-sheet content. TEM and cryo-TEM revealed the presence of mainly nanotape structures with micelles observed for systems rich in PRWG(C18H37). Analysis of detailed SAXS form factor measurements revealed the presence of bilayers 3–4 nm thick while the PRWG(C18H37) micelles have a core radius of approximately 3 nm, and a shell thickness of 2 nm. For the cyclohexanone/water systems polymorphs containing cluster aggregates (with radius of 0.25 nm to 0.50 nm) and some elongated structures (with radius of 5.7 nm to 26.1 nm) were seen. Longer structures were formed with the increase of the proline-containing lipopeptide content. The catalytic activity of these peptides was assessed using a model nitro-aldol reaction. The concentration of water in the reaction system influenced the conversion, higher content promoted better efficiency for the water systems, but the opposite was observed for the cyclohexanone/water samples.
- This article is part of the themed collection: Peptide Soft Materials


 Please wait while we load your content...
Please wait while we load your content...