Open Access Article
Open Access ArticleThe differential effects of endogenous cathepsin and microorganisms on changes in the texture and flavor substances of grouper (Epinephelus coioides) fillets
Xicai Zhang abcde and
Jing Xie*abcd
abcde and
Jing Xie*abcd
aCollege of Food Science & Technology, Shanghai Ocean University, Shanghai 201306, China. E-mail: 116215986@qq.com; jxie@shou.edu.cn; Tel: +86 2161900391
bShanghai Engineering Research Center of Aquatic Product Processing and Preservation, Shanghai 201306, China
cShanghai Professional Technology Service Platform on Cold Chain Equipment Performance and Energy Saving Evaluation, Shanghai 201306, China
dNational Experimental Teaching Demonstration Center for Food Science and Engineering, Shanghai Ocean University, Shanghai 201306, China
eJingchu University of Technology, Jingmen, 448000, China
First published on 13th March 2020
Abstract
Grouper is an important seafood species in China and has high economic value. However, the edible value of grouper is seriously affected by deterioration in the texture and flavor during refrigeration. The purpose of this study was to investigate the effects of endogenous cathepsin and microorganisms on texture softening and flavor changes in refrigerated grouper fillets. Iodoacetic acid and ProClin 300 were used to inhibit endogenous protease activity and microbial growth separately. Iodoacetic acid can inhibit the activity of cathepsin B, L, and calpain. Moreover, iodoacetic acid does not significantly affect the growth of microorganisms. The total amounts of bacteria and Pseudomonas spp. in the samples treated with ProClin 300 were less than 2 log CFU g−1 and 1 log CFU g−1 on the 18th day, and the activity of protease was not significantly affected. On the 6th day, the hardness of the iodoacetic acid treatment group decreased by 8%, while the ProClin 300 treatment group decreased by 28%, and changes in the free amino acids and volatile substances significantly exceeded those of the iodoacetic acid treatment group, indicating that endogenous protease was the main factor in the texture deterioration. A first-order exponential decay model indicated that cathepsin L was the most important protease for reducing the hardness of grouper fillets, and changes in the content of free amino acids and volatile substances indicated that microorganisms played a more important role in the deterioration of flavor substances compared to that played by endogenous protease.
1. Introduction
The grouper (Epinephelus coioides) belongs to the order Perciformes, Serranidae, and Epinephelus. As a warm-water, offshore bottom fish, the grouper is called “marine chicken” as its meat tastes like chicken. Grouper has become an important economic fish in China's coastal areas owing to the huge market demand and the continuous development of artificial breeding technology.1 With China's economic development and the continuous improvement of people's living standards, consumption of precious seafood has continued to increase, and changes in the pace of life have led to changes in the supply for the grouper market. The younger generation no longer favor fresh seafood like the previous generation, and perfectly prepared foods such as refrigerated fish fillets are more suitable for the needs of modern urban people.2Grouper fillet is rich in nutrition, but is prone to deterioration under refrigerated conditions. The deterioration in the texture and flavor eventually leads to the loss of the aquatic products' commercial value, which is also an important factor influencing consumers' acceptance of refrigerated fillets.3,4 As many studies have shown, the deterioration in the quality of aquatic products during refrigeration is due to two factors: the change in the endogenous proteases and the action of microorganisms.5,6 In the process of refrigeration, the muscle softening of aquatic products is caused by the degradation of skeletal proteins, which is due to the highly active endogenous proteases and exogenous proteases secreted by microorganisms. Meanwhile, the change in the flavor substance is one of the important characteristics of the change in quality of aquatic products.
The catalysis of proteases is an important factor in the formation and the change in flavor, proteins are reduced to peptides by the protease, and then free amino acids are formed by aminopeptidase. Both peptides and free amino acids not only have flavor themselves, but are also the precursor substances of many flavor substances.7 Many studies have shown a strong link between protease and a fish flavor, Strecker degradation of amino acids may produce aldehydes and ketones with a peculiar smell, and the generation of amino acids is related to endogenous proteases and microorganisms.8 Xu found that branched aldehydes could also be produced from amino acids through Strecker degradation or microbial metabolism, and autochthonous microflora could also accelerate the formation of ketones and acids.9 These enzymes may come from endogenous proteases or from microorganisms, this means that the softening of muscles and the change in the flavor compounds in refrigerated aquatic products are believed to be caused by the degradation of highly active endogenous proteases and spoilage microorganisms.4,10 Many studies have shown that cathepsin B, L, and D and calcium-activated proteases have a high activity in aquatic products, which can lead to deterioration in the quality and structure.11,12 Owing to the different environments of different seafood, the endogenous protease activity and the key enzyme types that play a major role have obvious differences.13
At present, the mechanism of texture and flavor changes in refrigerated grouper fillets owing to the contribution of endogenous proteases and microorganisms is unclear. Therefore, this paper aims to compare the contribution of endogenous proteases and microorganisms on the texture and flavor in the muscles of refrigerated grouper. If endogenous proteases are the dominant factor in the quality changes, further experiments will determine the types of endogenous proteases that play a key role. In this experiment, ProClin 300 and iodoacetic acid (IAA) were used to inhibit the microorganisms and endogenous protease in the grouper fillets in order to study the differences in the texture deterioration and flavor changes in different treatment groups during refrigeration. This will be used to determine the role of endogenous proteases and microorganisms in the decline in fish quality. Then, the key protease types were identified through correlation analysis of different endogenous protease activities and the deterioration in texture. This study will be helpful for exploring the specific role of endogenous proteases and microorganisms in the texture and flavor deterioration of refrigerated grouper fillets. It will also contribute to the development of technology for the preservation of refrigerated grouper fillets, to control and improve the quality of grouper fillets in the process of refrigerated storage.
2. Materials and methods
2.1. Preparation and immersion pretreatment of grouper fillets
Fresh grouper (Epinephelus coioides) (500 ± 50 g; n = 30) were purchased from the seafood market of Luchao Port (Shanghai, China). The grouper was stunned and killed, washed with distilled water and drained on ice. Based on the cleaning process, the head, bones, and skin of the grouper were removed and drained under refrigerated temperatures.The dorsal part of the grouper muscle was filleted to 1 × 1 × 2 cm and divided into three groups. The samples in the control group (C group) were soaked in deionized water, the fillets in the protease inhibiting group (I group) were immersed in 1 mmol L−1 IAA solution at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (v/w), and the samples in the bacteria inhibiting group (P group) were immersed in 0.05% ProClin 300 solution at a ratio of 1
10 (v/w), and the samples in the bacteria inhibiting group (P group) were immersed in 0.05% ProClin 300 solution at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (v/w), all of the soaking times were 4 h. Each group of fillets was drained and placed in sterile bags, and then refrigerated at 4 °C for subsequent analysis.
10 (v/w), all of the soaking times were 4 h. Each group of fillets was drained and placed in sterile bags, and then refrigerated at 4 °C for subsequent analysis.
2.2. Activity assay for endogenous proteases
According to the method previously described by Hultmann,14 5 g of grouper muscle was homogenized with 20 mL of Tris–HCl buffer solution (20 mmol L−1, pH 7.5), then centrifuged at 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×g for 20 min at 4 °C. The supernatant provided a crude extraction of the grouper protease. The measurement of the activity of the protease was conducted using the method previously published by Yu [4]. The specific fluorescent substrates of cathepsin B, cathepsin L and calpain were Z-Arg-Arg-MCA, Z-Phe-Arg-MCA, and Suc-Leu-Tyr-7-MCA, respectively. The above mentioned fluorogenic substrates were purchased from Shanghai Sigma Aldrich Co., Ltd. The fluorescence spectrophotometer (Hitachi F-7000, Tokyo, Japan) was used to detect the 7-amino-4-methylcoumarin (AMC) which came from the reaction between the fluorogenic substrates and proteases. The excitation wavelength (λex) and emission wavelength (λem) were set to 380 and 460 nm with slits width of 5 nm. One unit of enzyme activity was defined as the amount of enzyme that releases 1 nmol L−1 AMC per minute at 40 °C.
000×g for 20 min at 4 °C. The supernatant provided a crude extraction of the grouper protease. The measurement of the activity of the protease was conducted using the method previously published by Yu [4]. The specific fluorescent substrates of cathepsin B, cathepsin L and calpain were Z-Arg-Arg-MCA, Z-Phe-Arg-MCA, and Suc-Leu-Tyr-7-MCA, respectively. The above mentioned fluorogenic substrates were purchased from Shanghai Sigma Aldrich Co., Ltd. The fluorescence spectrophotometer (Hitachi F-7000, Tokyo, Japan) was used to detect the 7-amino-4-methylcoumarin (AMC) which came from the reaction between the fluorogenic substrates and proteases. The excitation wavelength (λex) and emission wavelength (λem) were set to 380 and 460 nm with slits width of 5 nm. One unit of enzyme activity was defined as the amount of enzyme that releases 1 nmol L−1 AMC per minute at 40 °C.
The activity of cathepsin D was measured using the acid-induced denaturation of bovine serum protein as detailed by Hagen et al.15 The polypeptide released by the reaction of cathepsin D in the crude enzyme solution and acid denatured bovine hemoglobin was determined using the method previously described by Lowry.16 The activity unit (U) was defined as the amount of enzyme that releases 1 μmol L−1 tyrosine per minute at 37 °C.
2.3. Microbiological analysis
Microbiological analysis was conducted according to the method previously described by Li.17 10 g of the sample was added to 90 mL of a sterile solution containing 0.85% NaCl under a sterile environment and homogenized. 1 mL of the solution was added to 9 mL of the sterile saline for gradient dilution, and three appropriate gradients (the number of colony-forming units should be between 30–300 when counting) were selected. The total viable counts (TVC) were cultured using plate count agar at 30 °C for 72 h, and Pseudomonas spp. was cultured in Pseudomonas selective media at 25 °C for 48 h, all of the above cultures were purchased from Qingdao Haibo Biotechnology Co., Ltd. The mean counts of the TVC and Pseudomonas spp. were calculated and expressed as log CFU g−1 muscle.2.4. Texture analysis
The hardness of the grouper fillets subjected to different treatments was analyzed at certain sampling intervals, and then a texture analyzer (TA.XT Plus, English stable micro system Ltd) with a flat cylindrical probe p/5 (5 mm in diameter) was used to test each sample after two compressions using the faceted texture analysis (TPA) mode. The average value of the hardness was obtained from six samples in each group.2.5. TCA-soluble peptides
According to the method of Sriket et al.,18 4 g of the muscle was homogenized with 27 mL trichloroacetic acid (TCA) solution (5%) for 1 min at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, and then placed in an ice bath for 1 h. The obtained homogenate was centrifuged at 5000×g for 5 min at 4 °C. The value of the TCA-soluble peptides was obtained according to the method described by Lowry et al.16
000 rpm, and then placed in an ice bath for 1 h. The obtained homogenate was centrifuged at 5000×g for 5 min at 4 °C. The value of the TCA-soluble peptides was obtained according to the method described by Lowry et al.16
2.6. Myofibril fragmentation index
The method of Aroeira et al. was used to evaluate the myofibril fragmentation index (MFI).19 4 g of the muscle was homogenized with 40 mL of the pre-chilled buffer solution (100 mM KCl, 20 mM potassium phosphate, 1 mM magnesium chloride, 1 mM sodium azide, and 1 mM ethylenediamine tetraacetic acid disodium salt) for 30 s (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm). This was then centrifuged at 1000×g for 15 minutes, and 40 mL of buffer solution was added to the precipitate, this was centrifuged again using the same parameters. Next, the precipitate was added to 10 mL of the buffer and homogenized for 30 s (15
000 rpm). This was then centrifuged at 1000×g for 15 minutes, and 40 mL of buffer solution was added to the precipitate, this was centrifuged again using the same parameters. Next, the precipitate was added to 10 mL of the buffer and homogenized for 30 s (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm), and then filtered through a polyethylene mesh (18 mesh). Finally, the concentration of the protein solution was adjusted to 0.5 mg mL−1, then the absorbance was measured at 540 nm, multiplying the absorbance by 200 gave the MFI value.
000 rpm), and then filtered through a polyethylene mesh (18 mesh). Finally, the concentration of the protein solution was adjusted to 0.5 mg mL−1, then the absorbance was measured at 540 nm, multiplying the absorbance by 200 gave the MFI value.
2.7. Volatile basic nitrogen
The muscle (5 g) was mixed with a moderate amount of light magnesium oxide, the volatile basic nitrogen (TVB-N) was measured according to GB-5009.228 2016 in China using DigiPREP TKN Systems (Kjeltec 2300, Denmark). The content of TVB-N was expressed as mg TVB-N per 100 g muscle.2.8. Free amino acid
Samples of free amino acid (FAA) extracts were prepared according to the method previously describe by Yu et al.20 Detection of the content of the FAA was performed according to the method described in our previous study using the automatic amino acid analyzer (L-8800, Hitachi Co. Ltd, Japan).212.9. Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
The extraction of myofibrillar was performed according to Ogawa's method with a slight modification.22 2 g of the muscle was washed five times with a volume of Tris–HCl buffer (pH 7.0) three times, then homogenized with five volumes of KCl–Tris buffer for 90 s (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm) in an ice bath with a short pause to prevent overheating. The homogenate was centrifuged at 5000×g for 10 min, a volume of 10 mmol L−1 Tris–HCl buffer (containing 0.6 mol L−1 NaCl, pH 7.0) was added to the precipitate five times, it was centrifuged for 10 min at a speed of 5000×g, and the supernatant obtained was the myofibrillar protein.
000 rpm) in an ice bath with a short pause to prevent overheating. The homogenate was centrifuged at 5000×g for 10 min, a volume of 10 mmol L−1 Tris–HCl buffer (containing 0.6 mol L−1 NaCl, pH 7.0) was added to the precipitate five times, it was centrifuged for 10 min at a speed of 5000×g, and the supernatant obtained was the myofibrillar protein.
Using the method reported by Verrez-Bagnis,23 sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) was performed at a 4–20% gradient, and a real-band three-color high-range protein marker purchased from Sangon Biotech (Sangon Biotech Co., Ltd, Shanghai, China) was adopted, and the optical density calculation was analyzed using Quantity One software (Quantity One 4.0, Bio-Rad Inc., USA).
2.10. Microstructures
Grouper fillets were sampled on day 0 and 9 for each treatment group. According to the method reported by Castejón,24 the samples were cut into 1 × 1 × 1 mm pieces along the direction of the myofibrils, then soaked with 2.5% glutaraldehyde solution and 2% tetroxide solution. Next, 30%, 50%, and 70% hexanol solutions were used to dehydrate the pieces for 10 min in turn. When finished, they were embedded in epoxy resin and thinly sliced with a frozen microtome. The slices were stained with uranyl acetate and lead citrate and observed using a transmission electron microscope (Tecnai G2 Spirit, Thermo Fisher Scientific, USA) with a magnification of 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times, the smallest unit in the image acquisition was 1 μm.
000 times, the smallest unit in the image acquisition was 1 μm.
2.11. Solid-phase microextraction gas chromatography mass spectrometry analysis
The solid-phase microextraction gas chromatography mass spectrometry (SPME-GC-MS) analysis was performed according to the method reported by Jaffrès et al. with some adjustments.25 5 g of the fish muscle was weighed and crushed on days 0, 9 and 18 for each experimental group, then the muscle was added into 20 mL vials with polypropylene nuts and PTFE/silicone septa before being sealed (Supelco, Bellefonte, PA, USA). The samples were equilibrated at 40 °C for 50 min and extracted with a 100 μm PDMS extraction head (Supelco, USA) for 25 min to absorb the volatile compounds onto the SPME fibers. The fibers of the SPME were introduced into the inlet of a GC-MS (5977A-SPME, Agilent, USA) at 280 °C for 5 min to desorb the volatile compounds, a chromatographic column (Agilent J & WDB-5 ms, 30 m × 0.25 mm × 0.5 μm) was used to analyze the volatile compounds, and helium was used as the carrier gas with a flow rate of 1.3 mL min−1. The temperature-rising program used was as follows: the initial temperature of the column was 40 °C for 5 min, the temperature was raised to 140 °C for 1 min with a heating rate of 3 °C min−1, and then the heating rate was increased to 6 °C min−1 up to 280 °C and held for 5 min. The detector had a mass range of 33 to 300 and a scan rate of 2 times per second. Volatile components were identified based on the mass spectrometry database of Wiley and Nist with a match quality of 80%.2.12. Statistical analysis
Six replicates were used for texture analysis and three replicates were used for other indicators with three parallel tests. SPSS 8.0 (SPSS, Chicago, IL, USA) was used for statistical analysis. Univariate analysis of variance (ANOVA) was used to determine the statistical differences in the different treatment groups. Duncan's multiple range test was conducted to determine the significant differences between groups at a significant level at P < 0.05. The data were expressed as mean standard deviation (SD), and Origin 8.5 (OriginLab, Northampton, MA, USA) was used for plotting.3. Results and discussion
3.1. Cathepsin activity analysis
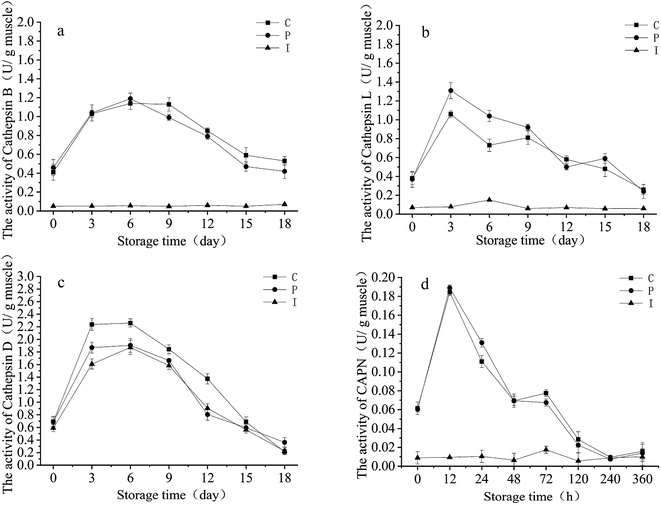
At present calpains and cathepsins are identified as two characterized proteolytic systems. Calpains are intracellular neutral cysteine proteases which have a calcium dependence, and cathepsin is an acid protease located in the lysosome. In the post-mortem fish, the pH decline contributes to the release of endogenous cathepsin from the lysosome into the cytosol, which leads to myofibrillar proteolysis.26 Changes in the cathepsin activity of all treatments are presented in Fig. 1.As shown in Fig. 1a–d, on day 3, the cathepsin B and L activities of the grouper fillets were 2.5 fold and 3.5 fold greater than that on day 0 in the P and C group, which dropped gradually at a late stage. Bahuaud found that the cathepsin B and L activity of Atlantic salmon increased significantly within 48 h and then decreased,27 this could be due to the different properties of cathepsin in different kinds of fish. The cathepsin B activity slightly increased by about 10% on day 18 and the cathepsin L activity increased by about 20% in the I group. Compared to the P and C group, the cathepsin B and L activities were inhibited by IAA obviously in the I group (p < 0.05). The slight increase in cathepsin B and the B ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) +
+ ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) L activities could be due to the sustained synthesis of cathepsin, studies have shown that the relevant genes which are responsible for cathepsin synthesis and cell apoptosis are continued to be expressed after the death of large yellow croakers (Pseudosciaena crocea).28 The difference between the cathepsin B and L activities was not significant between the P and C group during refrigerated storage, which showed that ProClin 300 had little impact on endogenous cathepsin. The decrease in the cathepsin B and L activities could be due to the pH value increase with the accumulation of amine compounds, which led to the values diverging from the optimum pH range for cathepsin.27
L activities could be due to the sustained synthesis of cathepsin, studies have shown that the relevant genes which are responsible for cathepsin synthesis and cell apoptosis are continued to be expressed after the death of large yellow croakers (Pseudosciaena crocea).28 The difference between the cathepsin B and L activities was not significant between the P and C group during refrigerated storage, which showed that ProClin 300 had little impact on endogenous cathepsin. The decrease in the cathepsin B and L activities could be due to the pH value increase with the accumulation of amine compounds, which led to the values diverging from the optimum pH range for cathepsin.27
Moreover, cathepsin D activity had no significant difference among three groups (p < 0.05), indicating that cathepsin D was unaffected by IAA. The reason for this was because the cathepsin D belongs to the aspartic acid enzyme, and IAA inhibits the activity of cysteine protease only.29 In addition, cathepsin D activity reduced gradually because of the changes in the pH value.
Calpain has maintained a low activity in the I group, and the calpain activity reached the maximum within 12 h in the P and I group and declined rapidly thereafter, it was below the initial value after 48 h. A similar result was found in a study of Caballero's observations,30 this indicated that calpain probably works on the grouper muscle early on in the refrigerated storage time. The inhibitive effects of IAA on calpain was proved by the experimental data.
3.2. Microorganism analysis
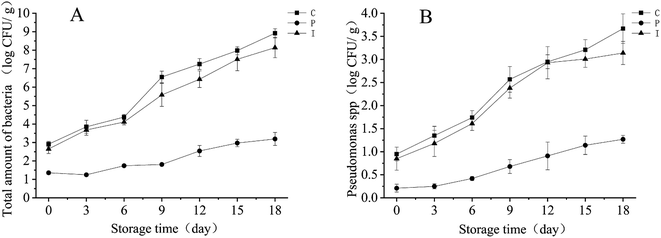
As a type of broad-spectrum antibacterial agent, ProClin 300 (Sigma-Aldrich) is a mixture which consists of 5-chloro-2-methyl-4-different thiazole moiety-3-ketone (CMIT) and 2-methyl-4-different thiazole moiety-3-ketone (MIT). The total amount of bacteria and Pseudomonas spp. in the different treatment groups are shown in Fig. 2.Compared to the I and C group, the P group fillets had a statistically significant lower total amount of bacteria and Pseudomonas spp. during refrigerated storage (P < 0.05). The total amount of bacteria and Pseudomonas spp. of the P group was restricted below 2 log CFU g−1 and 1 log CFU g−1 muscle during the entire storage period, which was restricted almost to below the initial level of the I and C group. However, the total amount of bacteria and Pseudomonas spp. of the I group was almost equal to the C group, this shows that IAA has a reduced effect on the growth of the microbiology. The growth of microorganisms was slow in the I group, as presented in Fig. 2, the total amount of bacteria and Pseudomonas spp. was almost 10% and 20% lower, separately, than that of the control group. The reason for the slight inhibition of the microorganisms by IAA might be because it inhibits the microbial metabolic enzymes including NADH oxidase and GPDH, which mainly contains sulfhydryl groups.31
3.3. Changes in the hardness
The hardness of the fish muscle can reflect the change in characteristics and quality during refrigerated storage. The fish fillets were softened with the decrease in the hardness, this means that the quality of fish has deteriorated.4 The changes in the hardness of different treatment groups are shown in Fig. 3.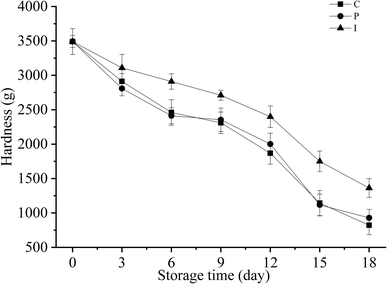 | ||
| Fig. 3 Changes in the hardness of grouper fillets with different pretreatments during refrigerated storage for 18 d at 4 °C. | ||
As presented in Fig. 3, the hardness of the grouper fillets consistently decreased with the storage time. The P and C groups experienced a striking decrease in the hardness which had dropped by 28% from day 0 to day 6, but there were no significant differences between the P and C group. However, the hardness of the I group fish fillets had dropped by only 8% from day 0 to day 6, and there was an obvious difference compared to the other groups (P < 0.05). The results indicated that IAA could effectively alleviate the decline in hardness of grouper fillets in cold storage. Through the above described analysis, endogenous protease was determined as being the main reason for the softening of the fish fillets, and it seems that the growth of microorganisms had no significant effect on the texture of the grouper fillets.
3.4. Changes in TCA soluble peptides
The TCA soluble peptide content can reflect the degree of degradation of the proteins. As shown in Fig. 4, the TCA soluble peptide content of all groups increased to some extent during storage.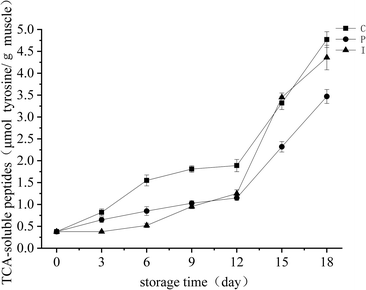 | ||
| Fig. 4 Changes in the TCA-soluble peptides of grouper fillets with different pretreatments during refrigerated storage for 18 d at 4 °C. | ||
Meanwhile, the order of the TCA soluble peptide content from days 3 to 6 was as follows: C group > P group > I group. It can be concluded that the endogenous proteases played a more important role in the increase in TCA soluble peptides than the microorganisms. Therefore, the endogenous proteases were a major factor in the increased TCA soluble peptide content, and similar results for grass carp (Ctenopharyngodon idellus) were reported by Yu [4]. The TCA soluble peptide content had a sudden increase after day 12 in the I group, which is consistent with the trends in the numbers of microorganisms, this shows that the primary cause of the TCA soluble peptide is the exogenous protease which is produced by the microorganisms during the latter part of the refrigerated period. Overall, this study found that endogenous cathepsin and microbial protease may act synergistically for protein hydrolysis, which promotes the accumulation of the TCA soluble peptide, the cathepsin plays the role of endopeptidase and the microbial protease primarily belongs to exopeptidase.
3.5. Changes in TVB-N
The volatile basic nitrogen is produced by the enzymatic degradation and microbial decomposition that occurs during the spoiling of animal-derived products, the content is an important indicator of the freshness of the fish and in the assessment of safety. TVB-N is composed primarily of ammonia, trimethylamine, dimethylamine and so on.32 TVB-N content in different treatment groups is shown in Fig. 5.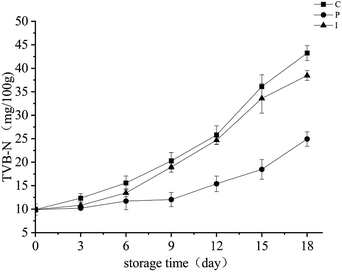 | ||
| Fig. 5 Changes in the TVB-N content of grouper fillets with different pretreatments during refrigerated storage for 18 d at 4 °C. | ||
Similarly, TVB-N content in the different treatment groups increased with the refrigerated preservation time. The TVB-N content of the C and I groups was significantly higher than that of the P group on day 9 (p < 0.05). The TVB-N content of the C group was slightly higher than that of the I group, but there was no significant difference between them. The results demonstrated that endogenous proteases showed no significant influence on the TVB-N content of the grouper fillets, the reason may be that the decarboxylase, which functions in the decarboxylation of the amino acid, was mainly from the microorganism. Moreover, the TVB-N content of the P group was still below the levels in the 30 mg per 100 g muscle on day 18.
3.6. Changes to the microstructures
Myofibrils are the basic units of muscle contraction and relaxation, maintaining the structural integrity of the myofibril plays an important role in the quality control of the muscle. Myofibrils are composed of thick filaments and thin filaments. The composition of the thick filaments is myosin and the composition of the thin filaments is actin, supplemented by tropomyosin and troponin. The sarcomere consists of the dark band (A-band) and half of the light band of both sides (I-band).33 Transmission electron microscopy was used to observe the myofibril of fresh grouper fillets and samples from all groups on day 9. The microstructure provided intuitive evidence for the interpretation of the relationship between the myofibril protein degradation and grouper fillet softening.As shown in Fig. 6A, it can be seen that the fresh muscle myofibril had a good structural integrity on day 0, a clear distribution of the I and A bands was found upon observation, and the Z-disk and M-line aligned in a good manner. On the 9th day, it was found that the Z-disk and M-line gradually disappeared in the C group, meanwhile, the I and A bands became indistinct, which shows the integrity of the myofibril was destroyed. The Z-disk of the myofibril was composed of desmin and actin, and the M-line was partially dependent on titin. It can be deduced from Fig. 6B that desmin, actin, and titin were degraded seriously on day 9 in the I group. The I and A bands in the P group were similar to that in the C group. Although the M-line was observed to be quite blurry, and the junction of the Z-disk and I bands had become fragmented. However, the I band, A band, M-line, and Z-disk were observed clearly in the I group on day 9.
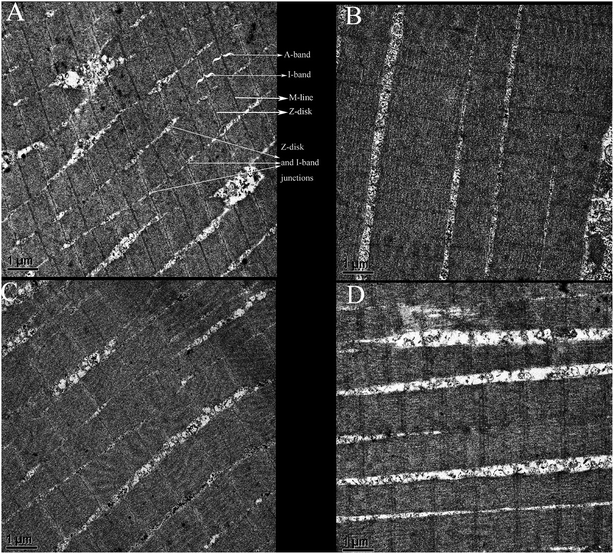 | ||
Fig. 6 Changes in the myofibrils of grouper fillets stored at 4 °C (transmission electron microscopy was used for observation, ×30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000): (A) fresh grouper fillets; (B) C9; (C) I9; and (D) P9. 000): (A) fresh grouper fillets; (B) C9; (C) I9; and (D) P9. | ||
The results showed that the endogenous protease plays a key role in the change in the microstructure and softening of the texture, which was caused by the degradation of myofibril proteins by endogenous protease.
3.7. Changes in the myofibril fragmentation
Myofibril fragmentation (MFI) is a phenomenon of the myofibril which is fragmented into shorter fragments at the Z-disk or close by,34 the main reason for this is the degradation of connectin and nebulin in the I bands. The value of MFI reflected the extent of damage to the internal structural integrity of the myofibril.35 The change in the MFI in different treatment groups is shown in Fig. 7.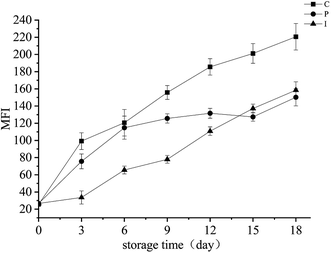 | ||
| Fig. 7 Changes in the MFI values of the grouper fillets with different pretreatments during refrigerated storage for 18 d at 4 °C. | ||
The MFI of the different treatment groups showed an upward trend with the prolonged refrigerated storage time. On the 9th day, the MFI of the C, P, and I groups reached 6.06, 4.89, and 2.99 times the initial values, respectively. The result shows that the IAA effectively inhibited the degradation of the myofibrillar protein, indicating that the endogenous protease was the main cause of the myofibril breakage and degradation during the early refrigerated storage time, which was consistent with that of the transmission electron microscopy (Fig. 6). However, the MFI values of both the I and P groups was significantly lower than that of the C group on day 18, which indicated that the proliferation of the microorganisms in the late stage of cold storage also had a significant role in promoting the increasing MFI.
3.8. Degradation of the myofibrils
Changes in the myofibril protein patterns of the grouper fillets with different pretreatments on days 0, 6, 12 and 18 were investigated using SDS-PAGE, the results are shown in Fig. 8.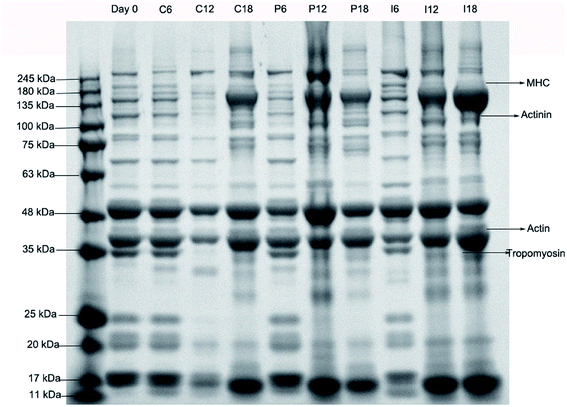 | ||
| Fig. 8 Changes in the myofibril protein patterns of grouper fillets with different pretreatments during refrigerated storage at 4 °C. | ||
Coomassie staining can indicate degradation of different levels of MHC (220 kDa) in different pretreatments groups compared to that of day 0. The optical density of MHC was 560, 585 and 765 in C6, P6 and I6, separately, which was analyzed using the optical density by Quantity One 4.0, indicating that the structural completeness of MHC remained in the I group. The degradation of MHC was one of the main causes of texture softening in the fish muscle. In a previous study by Wang,36 it was found that the degradation of MHC could be due to the function of endogenous protease.
Similar changes in the protein bands were seen in the cytoskeletal protein containing actin (42 kDa), actinin (105 kDa), and tropomyosin (34 kDa). The degradation of the cytoskeletal protein, even a small amount, could lead to inferior changes in the structure of the muscle, thus this might degrade the quality of the meat.37 There had been a significant slowdown in the degradation of myofibril in the I group on day 6, compared to the other groups, this indicated that the degradation of myofibril occurred by the action of protease containing cathepsin and calpain. The optical density of MHC and actin in the P group was higher than that of the C group on day 12, this showed that the exogenous enzymes that came from the microorganisms played a certain role in the degradation of the myofibril. The conclusions were consistent with that of the transmission electron microscopy, as well as the changes in the MFI.
3.9. Relationship between the activity of endogenous protease and the hardness
Correlational analyses of the relationship between the activity of endogenous protease and the hardness of the grouper muscle were performed according to the previously published study by Wickings.38 A first-order exponential decay model was adopted to form the functions as follows: Y = A × e−kx + B.In which X is the integral of the activity of endogenous proteases with time, Y is the hardness of the grouper fillets, K is the decrease in the enzyme activity of the unit time, and A and B are the coefficients of function.
The K value represents the contribution to the drop in the hardness of the endogenous proteases, the larger K value indicated that the proteases play a more significant role in muscle softening. Meanwhile, the correlation between the MFI and the enzyme activity was analyzed using SPSS software, it was expressed as r. The K and r values of the different endogenous proteases are presented in Table 1.
| Cathepsin B | Cathepsin L | Cathepsin D | Calpain | |
|---|---|---|---|---|
| K/R2 | 0.11/0.97 | 0.21/0.96 | 0.06/0.97 | 0.04/0.98 |
| R | 0.77 | 0.81 | 0.59 | 0.36 |
As shown in Table 1, the fitting degree of the first-order exponential decay model between the integral of several endogenous protease activities with the storage time and hardness was more than 0.95 with a good fitting effect. The K value of cathepsin L reached 0.21, and the K values of cathepsin B and D were 0.11 and 0.06, respectively; the K value of calpain was only 0.04. Several kinds of endogenous proteases contributed to different degrees to decrease in the hardness of the grouper fillets. The order of contribution among the endogenous proteases was cathepsin L > cathepsin B > cathepsin D > calpain. The correlation analysis between the enzyme activity and hardness also showed that cathepsin L was the most important protease for the texture deterioration of the grouper, and calpain protease activity had a lower correlation with hardness.
3.10. Changes to the FAA
The free amino acid (FAA) content values of different treatment groups are shown in Table 2.| FAA | Day 0 | C9 | C18 | P9 | P18 | I9 | I18 |
|---|---|---|---|---|---|---|---|
| a Results are expressed as mean in mg per 100 g of sample with standard error. Different lower case letters in different groups from the same day indicate a significant difference (p < 0.05). | |||||||
| Asp | 6.81 ± 0.18a | 4.57 ± 0.15b | 2.56 ± 0.09c | 3.86 ± 0.15c | 3.02 ± 0.12c | 4.45 ± 0.22b | 2.92 ± 0.07c |
| Thr | 15.24 ± 0.51c | 35.01 ± 1.05a | 17.39 ± 1.18bc | 20.13 ± 0.25a | 9.81 ± 0.15b | 32.73 ± 1.51a | 19.44 ± 2.18b |
| Ser | 20.25 ± 0.65b | 30.823 ± 1.15a | 5.82 ± 0.71d | 13.32 ± 0.17c | 6.65 ± 0.25d | 16.75 ± 1.57bc | 6.55 ± 0.75d |
| Glu | 9.72 ± 0.39d | 54.52 ± 3.09a | 26.87 ± 3.35c | 25.63 ± 1.18c | 30.51 ± 0.11bc | 53.87 ± 1.36b | 30.09 ± 3.37bc |
| Gly | 95.14 ± 2.68c | 331.85 ± 5.58a | 148.67 ± 3.05bc | 104.17 ± 1.75b | 88.73 ± 0.21bc | 268.79 ± 8.11b | 86.16 ± 3.85b |
| Ala | 24.54 ± 0.96bc | 113.61 ± 2.04a | 68.56 ± 1.85bc | 68.01 ± 0.59b | 37.53 ± 0.22bc | 111.11 ± 1.89bc | 76.26 ± 1.74bc |
| Val | 4.71 ± 0.18c | 18.62 ± 2.15ab | 5.596 ± 0.95c | 14.69 ± 0.14b | 6.25 ± 0.14c | 20.49 ± 0.36a | 6.17 ± 0.55c |
| Met | 1.75 ± 0.16d | 9.09 ± 0.26c | 4.32 ± 0.25d | 13.19 ± 0.16a | 4.64 ± 0.15d | 16.82 ± 0.15bc | 4.56 ± 0.14d |
| Ile | 2.65 ± 0.15c | 14.09 ± 1.52a | 3.33 ± 0.18c | 9.45 ± 0.85b | 3.64 ± 0.16c | 15.81 ± 0.84a | 3.59 ± 0.16c |
| Leu | 5.18 ± 0.13c | 22.26 ± 0.89a | 4.79 ± 0.26c | 13.82 ± 0.25b | 5.32 ± 0.02c | 23.22 ± 0.85a | 5.24 ± 0.28c |
| Tyr | 1.65 ± 0.28d | 8.83 ± 0.86b | 3.12 ± 0.43c | 9.18 ± 0.85b | 3.51 ± 0.17c | 15.16 ± 0.3a | 3.39 ± 0.41c |
| Phe | 5.15 ± 0.19c | 6.44 ± 0.25c | 3.76 ± 0.16c | 11.01 ± 0.44b | 4.43 ± 0.35c | 16.26 ± 0.98a | 4.29 ± 0.15c |
| Lys | 38.06 ± 1.54d | 95.44 ± 1.25b | 20.55 ± 0.48e | 65.07 ± 0.17c | 23.62 ± 0.22e | 106.85 ± 3.97a | 23.32 ± 0.43e |
| His | 4.39 ± 0.28c | 9.83 ± 0.58ab | 4.06 ± 0.82c | 8.61 ± 0.41b | 4.71 ± 0.13c | 10.16 ± 0.58a | 4.89 ± 0.89c |
| Arg | 4.87 ± 0.02c | 13.65 ± 0.88a | 1.17 ± 0.15d | 6.01 ± 0.35b | 1.31 ± 0.04d | 7.32 ± 0.19b | 1.39 ± 0.05d |
| Pro | 8.55 ± 0.18d | 32.89 ± 2.18a | 14.83 ± 0.45b | 5.89 ± 0.23d | 16.44 ± 0.16b | 8.71 ± 1.05d | 16.28 ± 0.52b |
| Total | 248.66 ± 19.61e | 801.55 ± 14.49a | 335.45 ± 8.52c | 392.03 ± 16.14b | 250.11 ± 10.31d | 728.55 ± 18.56a | 363.93 ± 13.73bc |
The accumulation of the FAA was closely related to the degradation of the proteins, which showed a trend of rising first and then decreasing, this was consistent with our previous research.21 The reduction in the total FAA content in the later period might be due to the decarboxylase enzymes produced by the rapid growth of microorganisms which convert the FAA into biogenic amines. It can be seen from Table 2 that on the 9th day, the content of FAA in the C, P, and I groups was 3.23, 1.28, and 2.93 times that of the initial FAA contents, respectively. The total amount of FAA in the P group was significantly lower than that in the C and I groups (p < 0.05), which indicated that endogenous cathepsins were mainly endopeptidases. The increase in FAA was mainly produced by the protease secreted by microorganisms, which is also consistent with the conclusions drawn by Nie et al.39 Aspartic acid, glutamic acid, and glycine are important flavor substances in fish muscles.40 A balanced amount of FAA provides a good taste to foodstuffs. However, the reduction of FAA in the later period might be caused by the degradation by microorganisms, thus, the FAA was turned into biogenic amines which led to the degradation of the quality of fish, such as tyrosine, lysine, and arginine.41 In summary, the production of free amino acids and biogenic amines were mainly caused by microorganisms during refrigerated storage. Although the flavor of the fish was increased by the production of the initial FAA, overall, it was detrimental to the quality of the grouper fillets during refrigerated storage.
3.11. Volatile substance analysis
The results of volatile substances in different treatment groups are shown in Table 3| Compound | 0 d | CON9 | I9 | P9 | CON18 | I18 | P18 |
|---|---|---|---|---|---|---|---|
| Alcohols | |||||||
| 1-Penten-3-ol | 5.37 ± 0.32 | 9.97 ± 0.15 | 5.05 ± 0.3 | 5.84 ± 0.18 | 1.63 ± 0.37 | 10.17 ± 0.14 | 5.26 ± 0.12 |
| 1-Octen-3-ol | 40.49 ± 3.15 | 19.79 ± 1.16 | 22.67 ± 1.46 | 35.68 ± 2.22 | 9.69 ± 1.51 | 9.91 ± 0.35 | 18.28 ± 1.53 |
| 1-Pentanol | 39.70 ± 1.87 | 25.35 ± 3.08 | 12.07 ± 0.45 | 10.07 ± 0.25 | — | 2.69 ± 0.12 | 24.73 ± 0.95 |
| 2-Ethylhexanol | 44.51 ± 0.25 | 33.32 ± 2.28 | 22.82 ± 0.52 | 11.46 ± 0.13 | 21.68 ± 1.12 | 18.48 ± 1.25 | 8.99 ± 0.18 |
| 3,7,11-Trimethyldodecan-1-ol | — | 0.85 ± 0.13 | 1.45 ± 0.44 | 0.93 ± 0.04 | — | 0.44 ± 0.63 | 0.33 ± 0.04 |
| 1-Hexanol | 1.61 ± 0.27 | 6.21 ± 0.94 | 6.09 ± 0.27 | 2.78 ± 0.56 | 3.33 ± 0.74 | 3.48 ± 0.36 | 6.41 ± 0.56 |
| 1-Heptanol | — | 1.65 ± 0.15 | 0.53 ± 0.03 | 0.86 ± 0.03 | — | — | 0.26 ± 0.02 |
| (E)-Oct-2-en-1-ol | — | 1.21 ± 0.53 | 1.86 ± 0.23 | 1.61 ± 0.15 | 1.04 ± 0.37 | 1.08 ± 0.14 | 1.35 ± 1.25 |
| 2-Hexadecanol | 1.71 ± 0.82 | 0.62 ± 0.03 | 0.42 ± 0.03 | 1.59 ± 0.14 | — | 0.11 ± 0.15 | 0.17 ± 0.02 |
| 2-Nonanol | — | 4.08 ± 0.66 | 4.11 ± 0.26 | 2.02 ± 0.15 | — | — | — |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Aldehydes | |||||||
| Hexanal | 13.27 ± 2.23 | 26.28 ± 1.8 | 21.51 ± 1.04 | 20.63 ± 3.02 | 19.28 ± 0.85 | 19.69 ± 2.78 | 16.49 ± 1.34 |
| (Z)-4-Heptenal | — | 5.02 ± 0.28 | 5.59 ± 1.86 | 5.13 ± 0.27 | 3.18 ± 0.82 | 3.58 ± 0.36 | 3.69 ± 0.88 |
| Heptanal | 11.21 ± 1.62 | 6.37 ± 1.36 | 6.37 ± 0.38 | 2.64 ± 0.75 | 0.58 ± 0.05 | 0.36 ± 0.29 | 0.33 ± 0.56 |
| Benzaldehyde | 15.11 ± 2.05 | 5.24 ± 0.49 | 10.36 ± 1.23 | 9.69 ± 1.56 | 2.45 ± 0.69 | 2.73 ± 0.62 | 4.38 ± 0.15 |
| (Z)-2-Decenal | 5.01 ± 0.35 | 2.39 ± 0.25 | 3.85 ± 0.28 | 5.03 ± 0.47 | — | 0.56 ± 0.22 | 2.03 ± 0.15 |
| 2-Methylbutyraldehyde | 5.45 ± 1.3 | 7.75 ± 0.56 | 3.18 ± 0.81 | 9.48 ± 0.54 | 2.60 ± 0.22 | 2.05 ± 0.05 | 6.02 ± 0.48 |
| (E)-2-Octenal | 3.73 ± 0.23 | 4.75 ± 0.20 | 8.53 ± 0.35 | 5.73 ± 0.33 | 11.93 ± 1.65 | 11.01 ± 0.79 | 5.18 ± 0.83 |
| Nonanal | 9.96 ± 3.35 | 17.58 ± 1.48 | 24.03 ± 2.68 | 27.76 ± 2.58 | 12.03 ± 1.65 | 12.77 ± 1.02 | 15.72 ± 3.04 |
| 3-Methylbutanal | — | 2.92 ± 0.13 | 2.64 ± 0.23 | 2.42 ± 0.28 | — | — | 1.01 ± 0.20 |
| Decanal | 3.35 ± 0.98 | 0.92 ± 0.17 | 1.86 ± 0.18 | 2.57 ± 0.27 | — | — | — |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Ketones | |||||||
| 2-Methyl-3-pentanone | — | 6.19 ± 0.40 | 5.15 ± 0.25 | 3.19 ± 0.43 | — | — | — |
| 2,3-Octanedione | 2.08 ± 0.36 | 5.93 ± 0.25 | 3.75 ± 0.23 | 4.75 ± 0.26 | 18.53 ± 2.32 | 18.39 ± 1.78 | 12.27 ± 2.85 |
| 2-Nonanone | — | 18.08 ± 2.49 | 15.25 ± 2.26 | 12.28 ± 1.28 | — | — | — |
| Acetophenone | 1.34 ± 0.86 | — | — | — | — | — | — |
| 2-Undecanone | 1.59 ± 0.26 | 1.89 ± 0.79 | 1.57 ± 0.25 | 2.63 ± 0.32 | — | — | 0.52 ± 0.23 |
| 3,5-Octadien-2-one | 1.52 ± 0.16 | 0.54 ± 0.06 | — | — | — | — | — |
| 3-Octanone | — | 28.71 ± 2.26 | — | 5.06 ± 0.49 | — | — | — |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Esters | |||||||
| Pyruvic acid methyl ester | — | 0.67 ± 0.26 | 0.74 ± 0.31 | — | — | — | — |
| Ethyl tetradecanoate | — | 0.53 ± 0.08 | 0.25 ± 0.11 | — | — | — | — |
| Butanoic acid, ethyl ester | — | 1.95 ± 0.42 | 1.71 ± 0.19 | — | — | — | — |
| Ethyl caprylate | — | 1.51 ± 0.36 | — | — | — | — | — |
| Ethyl palmitate | 1.69 ± 0.95 | 2.43 ± 0.58 | 3.15 ± 0.42 | — | 0.17 ± 0.01 | — | 0.48 ± 0.02 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Hydrocarbons | |||||||
| 1,3,5-Cycloheptatriene | — | 6.13 ± 0.58 | 5.25 ± 0.25 | — | 3.14 ± 0.88 | — | — |
| 2,6-Dimethyloctane | 2.51 ± 0.08 | 4.82 ± 0.24 | 10.76 ± 0.45 | 3.36 ± 0.93 | 1.85 ± 0.26 | 1.96 ± 0.53 | 1.07 ± 0.18 |
| Nonane | 2.32 ± 0.21 | 4.31 ± 0.79 | 5.02 ± 0.34 | 4.85 ± 0.31 | 10.09 ± 0.33 | 9.19 ± 0.36 | 8.75 ± 0.55 |
| Decane | — | 22.42 ± 3.56 | 24.53 ± 2.66 | 31.17 ± 4.19 | 27.52 ± 1.27 | 21.56 ± 2.56 | 26.01 ± 2.42 |
| Undecane | 1.99 ± 0.33 | 2.14 ± 0.23 | 1.62 ± 0.21 | 2.25 ± 0.32 | 1.65 ± 0.61 | 1.52 ± 0.84 | 1.29 ± 0.09 |
| Dodecane | 2.48 ± 0.93 | 3.03 ± 0.99 | 5.81 ± 0.83 | 6.93 ± 0.55 | 4.15 ± 0.77 | 5.19 ± 0.22 | 4.11 ± 0.24 |
| Tridecane | 2.36 ± 0.33 | 3.34 ± 0.44 | 6.43 ± 0.91 | 10.78 ± 1.61 | 4.34 ± 0.71 | 6.48 ± 0.23 | 4.41 ± 0.36 |
| Tetradecane | 3.39 ± 0.45 | 5.26 ± 0.21 | 6.35 ± 0.32 | 6.54 ± 1.06 | 4.14 ± 0.28 | 4.44 ± 0.87 | 5.21 ± 0.62 |
| Pentadecane | 14.86 ± 1.89 | 18.74 ± 1.03 | 21.36 ± 1.14 | 33.99 ± 2.28 | 11.41 ± 0.75 | 11.08 ± 0.49 | 7.07 ± 0.71 |
| Heptadecane | 1.51 ± 0.21 | 10.48 ± 1.81 | 23.32 ± 2.96 | 17.24 ± 2.23 | 7.85 ± 0.36 | 7.22 ± 3.23 | 3.68 ± 0.81 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Aromatics | |||||||
| p-Xylene | 19.56 ± 1.64 | 5.49 ± 0.49 | 9.01 ± 0.92 | 6.95 ± 0.44 | — | — | 0.29 ± 0.09 |
| Ethylbenzene | 4.25 ± 0.45 | 4.82 ± 0.26 | 2.32 ± 0.24 | 3.77 ± 0.17 | — | — | — |
| 1,3-Dimethylbenzene | 2.37 ± 0.25 | — | 8.48 ± 1.41 | 9.34 ± 0.34 | — | — | — |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Amines | |||||||
| (E)-Octadec-9-enamide | — | — | — | — | 3.96 ± 0.85 | 3.81 ± 0.21 | 4.51 ± 0.79 |
| 2,2 Dimethyl-propanamide | — | 1.81 ± 0.43 | — | 2.39 ± 0.60 | — | — | — |
| Trimethylamine | — | 24.69 ± 5.31 | — | — | 30.83 ± 2.39 | 19.11 ± 2.27 | — |
There were 52 kinds of volatile flavor substances detected in the fresh grouper muscle, including 6 alcohols, 8 aldehydes, 4 ketones, 5 esters, 10 hydrocarbons, 3 aromatics, 3 amines, and 4 other substances. As important flavor substances, aldehydes, ketones, and alcohols are mainly produced by the oxidative hydrolysis of lipids.42 On day 9, the total number of aldehydes, ketones, and alcohols was 10, 10, and 6 in the C, P, and I groups, respectively. The numbers of volatile substances increased under the action of endogenous enzymes and microorganisms with prolonged refrigerated storage time. However, some volatile substances disappeared on day 18, the reason for this may be that the substances changed into other substances or the concentrations were too low to be detected.
1-Octen-3-ol is often used as a symbol of freshness among alcohols,43 as it plays an important role in the formation of sea fish flavor owing to its low odor threshold.44 Fishy taste substances are mainly composed of hexanal, heptaldehyde, and decaldehyde.45 On day 9, the contents of fishy taste in the P and I groups were lower than those in the C group and the fishy substances in the P group were lower than those in the I group. Furthermore, the largest content of 1-octen-3-ol was retained in the P group among all of the groups, this showed that inhibiting the growth of microorganisms was conducive to maintaining the good flavor of grouper fillets. The contribution to flavor was basically negligible owing to the high threshold of hydrocarbons.43 It can be seen from Table 2 that the production of esters in group P was significantly less than the other two groups, it was reported that the production of ethyl esters was mainly caused by the effect of the esterase activity of the lactic acid bacteria and the catabolism of amino acids by Pseudomonas.46 Trimethylamine (TMA) is one of the representative volatile compounds that characterize the freshness of fish. The accumulation of TMA is due to the action of bacteria or enzymes which convert trimethylamine oxide (TMA-O) to TMA.47 TMA was detected on day 9 in the C group and day 18 in the I group, and TMA was not detected in the P group during the entire refrigeration cycle.
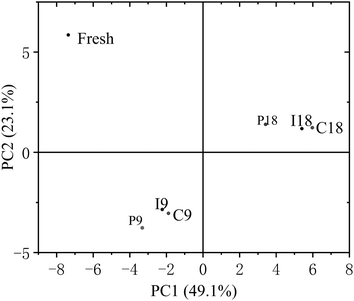
To simplify the correlation between the volatile components and pretreatment conditions, principal component analysis (PCA) was performed on the volatile compounds identified in the different pretreatment groups, the spatial distribution point of PCA was used to reflect the differences among samples. As shown in Fig. 9, the flavor of the grouper fillets in the I group was closer to that of the C group. By combining this with the results shown in Table 2, it can be concluded that the contribution of microorganisms to the change in volatile components exceeds that of endogenous proteases in refrigerated grouper fillets.
4. Conclusions
Iodoacetic acid solution can effectively inhibit cathepsin B, L, and calpain activity in grouper fillets, but has a poor inhibitory effect on cathepsin D. The number of microorganisms was reduced significantly upon treatment with ProClin 300 in grouper fillets during refrigerated storage. The degree of texture deterioration of the I group was significantly lower than those of the C and P groups. Combined with indicators such as the results of transmission electron microscopy and MFI, it was concluded that changes in the texture of refrigerated grouper fillets was mainly caused by endogenous protease, and cathepsin L played the most important role in the degradation of grouper myofibrillar proteins. There was little correlation between the flavor changes in refrigerated grouper fillets and endogenous proteases, according to comprehensive analysis of TVB-N, free amino acids, and volatile substances. Compared with the role of endogenous proteases, exogenous proteases secreted by microorganisms may play a greater role in changes in flavor substances in grouper fillets.Conflicts of interest
The authors declare no conflicts of interest.Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (grant number: 31972142, 31571914), the China Agriculture Research System (CARS-47), the Construction Project of Public Service Platform for Shanghai Municipal Science and Technology Commission (17DZ2293400), and the Shanghai Municipal Science and Technology Project to enhance the capabilities of the platform (grant number: 19DZ2284000).References
- S. Li, Z. Li, N. Chen, P. Jin and J. Zhang, Aquaculture, 2019, 498, 568–577 CrossRef CAS.
- H. Zhao, S. Liu, C. Tian, G. Yan and D. Wang, Int. J. Refrig., 2018, 88, 483–495 CrossRef CAS.
- D. Liu, L. Liang, W. Xia, J. M. Regenstein and P. Zhou, Food Chem., 2013, 140, 105–114 CrossRef CAS PubMed.
- D. Yu, J. M. Regenstein, J. Zang, W. Xia, Y. Xu, Q. Jiang and F. Yang, Food Chem., 2018, 262, 1–6 CrossRef CAS PubMed.
- S. Sharifian, E. Alizadeh, M. S. Mortazavi and M. M. Shahriari, J. Food Sci. Technol., 2014, 51, 929–935 CrossRef CAS PubMed.
- L. Iacumin, E. Tirloni, M. Manzano and G. Comi, Turk. J. Fish. Aquat. Sci., 2017, 17, 1279–1285 Search PubMed.
- L. Pripis-Nicolau, G. D. Revel, A. Bertrand and A. Maujean, J. Agric. Food Chem., 2000, 48, 3761–3766 CrossRef CAS PubMed.
- P. Li, Y. Peng, J. Mei and J. Xie, LWT--Food Sci. Technol., 2020, 118, 108831 CrossRef.
- Y. Xu, L. Li, J. M. Regenstein, P. Gao, J. Zang, W. Xia and Q. Jiang, Food Chem., 2018, 256, 259–267 CrossRef CAS PubMed.
- L. Zhang, Y. Zhang, S. Jia, Y. Li, Q. Li, K. Li, H. Hong and Y. Luo, Food Chem., 2019, 295, 520–529 CrossRef CAS PubMed.
- L. Ge, Y. Xu and W. Xia, Int. J. Food Sci. Technol., 2014, 50, 797–803 CrossRef.
- P. A. Wang, B. Vang, A. M. Pedersen, I. Martinez and R. L. Olsen, Food Chem., 2011, 124(3), 1090–1095 CrossRef CAS.
- Z. Ahmed, O. Donkor, W. A. Street and T. Vasiljevic, Trends Food Sci. Technol., 2015, 45, 130–146 CrossRef CAS.
- L. Hultmann, T. M. Phu, T. Tobiassen, Ã. Aas-Hansen and T. Rustad, Food Chem., 2012, 134, 1399–1408 CrossRef CAS PubMed.
- O. Hagen, C. Solberg and I. A. Johnston, J. Agric. Food Chem., 2008, 56, 5953–5959 CrossRef CAS PubMed.
- O. H. Lowry, N. J. Rosebrough, A. L. Farr and R. J. Randall, J. Biol. Chem., 1951, 193, 265–275 CAS.
- N. Li, W. Liu, Y. Shen, J. Mei and J. Xie, Coatings, 2019, 9(4), 273 CrossRef CAS.
- C. Sriket, S. Benjakul, W. Visessanguan, K. Hara and A. Yoshida, Food Chem., 2012, 135, 571–579 CrossRef CAS PubMed.
- C. N. Aroeira, R. A. Torres Filho, P. R. Fontes, A. L. S. Ramos, C. J. Contreras Castillo, D. L. Hopkins and E. M. Ramos, Meat Sci., 2020, 160, 107955 CrossRef PubMed.
- D. Yu, Y. Xu, J. M. Regenstein, W. Xia, F. Yang, Q. Jiang and B. Wang, Food Chem., 2018, 242, 412–420 CrossRef CAS PubMed.
- X. Zhang, W. Huang and J. Xie, Foods, 2019, 8, 325 CrossRef PubMed.
- M. Ogawa, S. Nakamura, Y. Horimoto, H. An, T. Tsuchiya and S. Nakai, J. Agric. Food Chem., 1999, 47, 3309 CrossRef CAS PubMed.
- V. Verrez-Bagnis, C. Ladrat, M. Morzel, J. Noël and J. Fleurence, Electrophoresis, 2015, 22, 1539–1544 CrossRef.
- O. J. Castejón and A. J. Caraballo, Scanning Electron Microsc., 1980, IV, 197–207 Search PubMed.
- E. Jaffrès, V. Lalanne, S. Macé, J. Cornet, M. Cardinal, T. Sérot, X. Dousset and J.-J. Joffraud, Int. J. Food Microbiol., 2011, 147, 195–202 CrossRef PubMed.
- R. Chéret, C. Delbarre-Ladrat, M. d. Lamballerie-Anton and V. Verrez-Bagnis, Food Chem., 2007, 101, 1474–1479 CrossRef.
- D. Bahuaud, T. Mørkøre, Ø. Langsrud, K. Sinnes, E. Veiseth, R. Ofstad and M. S. Thomassen, Food Chem., 2008, 111, 329–339 CrossRef CAS PubMed.
- J. Zhao, J. R. Li, J. L. Wang and W. J. Lv, J. Agric. Food Chem., 2012, 60, 11387–11394 CrossRef CAS PubMed.
- D. Martínez-Alarcón, R. Saborowski, L. Rojo-Arreola and F. García-Carreño, Comp. Biochem. Physiol., Part B: Biochem. Mol. Biol., 2018, 215, 31–38 CrossRef PubMed.
- M. J. Caballero, M. Betancor, J. C. Escrig, D. Montero, A. Espinosa de los Monteros, P. Castro, R. Ginés and M. Izquierdo, Aquaculture, 2009, 291, 210–216 CrossRef CAS.
- S. Kawasaki, J. Ishikura, D. Chiba, T. Nishino and Y. Niimura, Arch. Microbiol., 2004, 181, 324–330 CrossRef CAS PubMed.
- Y. Li, X. Tang, Z. Shen and J. Dong, Food Chem., 2019, 287, 126–132 CrossRef CAS PubMed.
- R. Hamm, Functional Properties of the Myofibrillar System and Their Measurements, 1986 Search PubMed.
- D. Wang, M. Zhang, S. Deng, W. Xu, Y. Liu, Z. Geng, C. Sun, H. Bian and F. Liu, Food Chem., 2016, 197, 340–344 CrossRef CAS PubMed.
- R. G. Taylor, G. H. Geesink, V. F. Thompson, M. Koohmaraie and D. E. Goll, J. Anim. Sci., 1995, 73, 1351 CrossRef CAS PubMed.
- P. A. Wang, I. Martinez and R. L. Olsen, Food Chem., 2009, 115, 1228–1233 CrossRef CAS.
- Y. Bao, K. Wang, H. Yang, J. M. Regenstein, P. Ertbjerg and P. Zhou, Food Chem., 2020, 308, 125576 CrossRef CAS PubMed.
- K. Wickings, A. S. Grandy, S. C. Reed and C. C. Cleveland, Ecol. Lett., 2012, 15, 1180–1188 CrossRef PubMed.
- X. Nie, S. Lin and Q. Zhang, Food Chem., 2014, 145, 840–844 CrossRef CAS PubMed.
- Ö. Özden, J. Sci. Food Agric., 2010, 85, 2015–2020 CrossRef.
- J. Calanche, A. Tomas, S. Martinez, M. Jover, V. Alonso, P. Roncalés and J. A. Beltrán, Food Res. Int., 2019, 119, 126–134 CrossRef CAS PubMed.
- K. H. S. Farvin, H. D. Grejsen and C. Jacobsen, Food Chem., 2012, 131, 843–851 CrossRef.
- L. G. Fidalgo, M. M. Q. Simões, S. Casal, J. A. Lopes-da-Silva, A. M. S. Carta, I. Delgadillo and J. A. Saraiva, Food Res. Int., 2020, 127, 108740 CrossRef PubMed.
- T. Sae-leaw and S. Benjakul, Eur. J. Lipid Sci. Technol., 2014, 116, 885–894 CAS.
- W. Guan, X. Ren, Y. Li and L. Mao, LWT--Food Sci. Technol., 2019, 101, 25–31 CrossRef CAS.
- A. Casaburi, P. Piombino, G. J. Nychas, F. Villani and D. Ercolini, Food Microbiol., 2015, 45, 83–102 CrossRef CAS PubMed.
- F. Pena-Pereira, I. Lavilla and C. Bendicho, Food Chem., 2010, 119, 402–407 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2020 |