Open Access Article
Open Access ArticleDesign and in vitro/in vivo evaluations of a multiple-drug-containing gingiva disc for periodontotherapy†
Pooja Jaina,
Mohd. Aamir Mirza*a,
Sushama Talegaonkarb,
Shyamasree Nandyc,
Mridu Dudejac,
Nilima Sharmad,
Md. Khalid Anwere,
Saad M. Alshahranie and
Zeenat Iqbal *a
*a
aDepartment of Pharmaceutics, School of Pharmaceutical Education and Research, Jamia Hamdard, New Delhi 110062, India. E-mail: Zeenatiqbal@jamiahamdard.ac.in; aamir.jhu07@gmail.com; Tel: +91-9811733016 Tel: +91-9213378765
bDepartment of Pharmaceutics, Delhi Pharmaceutical Sciences and Research University, Govt. of NCT of Delhi, New Delhi, India
cDepartment of Microbiology, Hamdard Institute of Medical Sciences and Research, New Delhi, India
dDepartment of Dentistry, Hamdard Institute of Medical Sciences and Research & HAH Centenary Hospital, Jamia Hamdard, New Delhi, India
eDepartment of Pharmaceutics, College of Pharmacy, Prince Sattam Bin Abdulaziz University, Al-kharj, 11942, Saudi Arabia
First published on 27th February 2020
Abstract
In the current work, we set out to develop and evaluate a gingiva disc of cellulose acetate phthalate and poloxamer F-127 for the simultaneous delivery of multiple drugs, namely minocycline, celecoxib, doxycycline hyclate, and simvastatin, to abolish infection, impede inflammation, avert collagen destruction, and promote alveolar bone regeneration, respectively. In vitro release studies revealed the sustained release profiles of the drugs for 12 h and that they were active against Staphylococcus aureus, Escherichia coli and Streptococcus mutans. The in vivo bioactivity levels of these drugs were assessed by comparing the number of colony forming units during different phases of a study on Wistar rats, and the results showed a reduction in the number of bacterial colonies with the applied formulation. A mucosal irritation study conducted on Wistar rat gingiva confirmed the non-irritancy of the optimal gingiva disc. Hence, this customized, non-invasive polymeric gingiva disc displaying a sustained release of drugs can be a useful tool to treat acute to moderate stages of periodontitis.
Introduction
Periodontitis is the commonest and most challenging progressive dental ailment and exhibits a wide spectrum of symptoms such as gingival puffiness, inflammation, bleeding, and detachment of tooth bone often leading to pocket formation of 3–4 mm.1 The progression is often accompanied by a deposition of biofilm, leading to deeper pockets and edentulism if left untreated. The conventional dental therapeutics revolves around mechanical debridement of the periodontal pocket with plaque control measures to remove bacterial infection and biofilm deposits.2 This treatment strategy, however, is time consuming, involves consumption of a high dose of antibiotics, is expensive, and is often marred with significant patient non-compliance. Many novel drug delivery systems have been proposed for this issue, but most such systems are associated with various limitations as they do not address the complexity of periodontitis, which demands instead an all-encompassing therapeutic approach capable of mitigating all of the associated disease symptoms.3–6The success of any drug delivery system also relies upon the choice of the target site. An acute phase periodontitis involving relatively shallow pockets can be better treated with a system that could adhere to the gingival tissue and bathe the gum line with the drugs and arrest the disease. The gingival delivery of drugs involves immobilizing a drug delivery device, namely a polymeric disc, on a specific site for targeted release in the oral cavity, eliciting intimacy and a specific duration of contact.7 Thereby on the gingiva, when using the proposed disc, the risk is much lower than the benefit.
A gingiva disc should simultaneously deliver antimicrobial, anti-inflammatory, anti-bone resorptive and osteogenic agents to achieve a complete reversal of acute phase periodontitis. The assemblage of drugs chosen in the current work included minocycline, an antibiotic against the plaque bacteria,8,9 celecoxib, a selective COX-II inhibitor for treating infection-associated inflammation,10 doxycycline hyclate as an inhibitor of matrix metalloproteinases (MMP-1, MMP-8) for circumvention of collagen destruction11–13 and simvastatin to promote the expression of bone morphogenic protein (BMP-2) and aid in alveolar bone regeneration.14,15
For the matrix of the gingiva disc, a system including an association of the polymers cellulose acetate phthalate and poloxamer F-127 (CAP-P) was selected, as it has been shown to release the drugs in a controlled manner. To impart mucoadhesion characteristics onto the gingiva disc, a thin coating of hydroxyl-ethyl cellulose solution was applied. The CAP-P system has been exploited in various studies to achieve a controlled and programmed drug delivery by altering the ratio of the amounts of the two polymers.16–19
The present study aimed to deliver antimicrobial, anti-inflammatory, anti-bone resorptive and osteogenic agents from the gingiva disc for the complete reversal of acute phase periodontitis.
Experimental
Materials
Cellulose acetate phthalate (CAP) and poloxamer F-127 were obtained from Jubilant Life Sciences (New Delhi, India) ex gratia. Hydroxyl-ethyl cellulose was procured from SRL India. Isotonic phosphate buffer of pH 6.8 was prepared according to USP XX (1980). Acetone and isopropyl alcohol were procured from E. Merck (India) Ltd (Mumbai, India). A Shimadzu (Japan) UV-1601 UV spectrophotometer was used for spectrophotometric analysis. A Shimadzu (Japan) LC-10 HPLC system was also used.Preparation of the polymeric gingiva disc
Each disc was prepared using the pressed pellet method.14 Various components and the four drugs minocycline, doxycycline, celecoxib and simvastatin in each formula were mixed by carrying out trituration in a glass pestle and mortar. All four drugs were present in a single disc in combined form. The mixture was then compressed using a 13 mm-diameter die on an infrared hydraulic press (Spectra Lab-SL-89, Mumbai) using a compression force of 5 tons and a compression time of 15 s. The prepared disc was then coated with a 2% (w/v) slurry of hydroxyl-ethyl cellulose in a mixture of acetone![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) isopropyl alcohol (65
isopropyl alcohol (65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35).20
35).20
Optimization of the gingiva disc
Discs were prepared using the pressed pellet method and optimized using Design Expert® version 10 software.21 A 23 factorial design was applied to study the effect of two independent factors, namely CAP concentration [A] and poloxamer concentration [B], in three levels (coded as −1, 0, +1). Four drugs were incorporated in the formulation and the amount of each drug was fixed at 6 mg. A total of eleven formulation batches were prepared in which 2% hydroxy-ethyl cellulose was used as a coating polymer. All eleven formulations were tested to determine mucoadhesion time, tensile strength, bioadhesive force, and in vitro drug release, and these results were compared to each other.Evaluation of the polymeric gingiva disc
| S.I. = [(M2 − M1) × 100]/M1, |
| Bioadhesive force (N) = bioadhesive strength (g) × 9.81 ÷ 1000. |
A similar method was used to assess the tensile strength of the disc, and the force at disc break was measured. Results here are reported as the mean (±SD) of three replicates (n = 3). The tensile strength was calculated using the formula
| TS (kg mm−2) = force at break (kg)/initial cross-sectional area of sample (mm−2). |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 10 min, 4 °C). A volume of 2 ml of the resulting supernatant organic layer was dried and reconstituted in 100 μL of mobile phase, and a volume of 20 μL of the resulting reconstituted sample was injected into the HPLC system for analysis.14
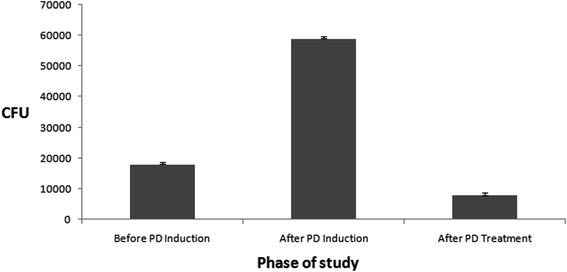
000 rpm, 10 min, 4 °C). A volume of 2 ml of the resulting supernatant organic layer was dried and reconstituted in 100 μL of mobile phase, and a volume of 20 μL of the resulting reconstituted sample was injected into the HPLC system for analysis.14For eight weeks, all experimental periodontitis-induced rats were fed with a 10% w/v sucrose solution. When the disease developed, the group 3 animals were treated with the gingiva disc. After eleven days of treatment, the different groups were examined for microbiological studies. In vivo antimicrobial activity of the disc was studied by comparing colony forming units (CFUs) on the blood agar media. Before disease induction, after disease development and after disease treatment with gingiva disc, mouth swabs were taken of the rats and applied onto the surface of blood agar media by using the streaking method. The plates were then incubated at 37 ± 0.5 °C for 24 h, and then CFUs were counted and compared.24 All of the studies were performed in triplicate (n = 3) and the results are presented as the mean of the three (mean ± SD).
Results & discussion
Preparation and optimization of the gingiva disc
Various discs were prepared using different amounts of the polymers cellulose acetate phthalate and poloxamer F-127, and a fixed amount of hydroxyl-ethyl cellulose as given in Table 1. A schematic representation of the disc is shown in Fig. 1. Furthermore, Design Expert® version 10 software was used to optimize the disc on the basis of tensile strength and maximum percentage drug release.| Formulation code | Total polymer (mg) | CAP (mg) | Poloxamer F-127 (mg) | Each drug (mg) |
|---|---|---|---|---|
| F01 | 110 | 70 | 40 | 6 |
| F02 | 100 | 65 | 35 | 6 |
| F03 | 110 | 60 | 40 | 6 |
| F04 | 95 | 65 | 30 | 6 |
| F05 | 90 | 60 | 30 | 6 |
| F06 | 100 | 70 | 30 | 6 |
| F07 | 95 | 60 | 35 | 6 |
| F08 | 105 | 65 | 40 | 6 |
| F09 | 105 | 70 | 35 | 6 |
| F10 | 100 | 65 | 35 | 6 |
| F11 | 100 | 65 | 35 | 6 |
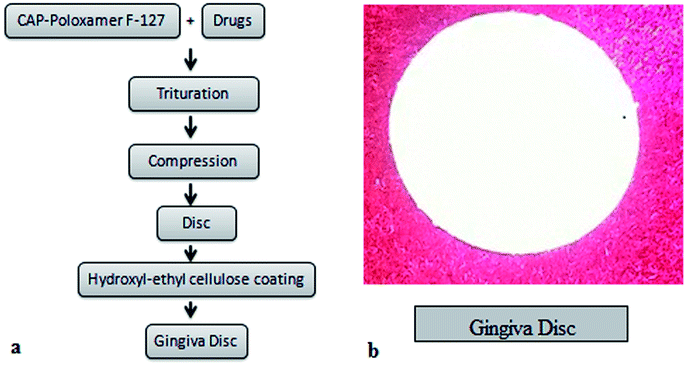 | ||
| Fig. 1 A schematic presentation of the design and development of the gingiva disc (a). A pictorial presentation of the developed gingiva disc (b). | ||
Weight and weight uniformity
Weights of ten discs of each formulation were measured and were found to be in the range 116.82–136.32 mg. Table 2 shows the means (±SD) of the obtained weight values. The low SD values reflected the weight uniformity amongst the various formulations of each batch.| Formulation code | Weight (mg) (mean ± SD) | Thickness (mm) (mean ± SD) | Surface pH (mean ± SD) | Swelling index (mean ± SD) | Bioadhesive force (N) (mean ± SD) | Mucoadhesion time (h) (mean ± SD) |
|---|---|---|---|---|---|---|
| F01 | 136.20 ± 0.57 | 0.86 ± 0.011 | 7.05 ± 0.58 | 45.28 ± 1.16 | 2.738 ± 0.51 | 11.00 ± 0.15 |
| F02 | 126.04 ± 0.57 | 0.81 ± 0.005 | 7.23 ± 0.58 | 38.67 ± 1.78 | 2.543 ± 0.81 | 10.80 ± 1.10 |
| F03 | 136.30 ± 0.58 | 0.75 ± 0.003 | 7.19 ± 0.57 | 29.70 ± 1.18 | 2.198 ± 0.93 | 09.00 ± 0.78 |
| F04 | 121.73 ± 0.57 | 0.84 ± 0.004 | 7.03 ± 0.58 | 41.12 ± 1.82 | 2.621 ± 0.71 | 11.15 ± 0.84 |
| F05 | 116.82 ± 0.57 | 0.79 ± 0.003 | 7.20 ± 0.58 | 34.57 ± 1.32 | 2.423 ± 0.82 | 10.02 ± 0.18 |
| F06 | 126.52 ± 0.57 | 0.83 ± 0.003 | 7.16 ± 0.57 | 38.89 ± 1.40 | 2.557 ± 0.93 | 12.50 ± 0.95 |
| F07 | 121.52 ± 0.57 | 0.80 ± 0.005 | 7.02 ± 0.57 | 35.78 ± 1.22 | 2.501 ± 0.81 | 10.25 ± 0.77 |
| F08 | 131.23 ± 0.58 | 0.85 ± 0.004 | 7.08 ± 0.57 | 42.09 ± 1.72 | 2.672 ± 0.22 | 12.25 ± 0.52 |
| F09 | 131.53 ± 0.58 | 0.77 ± 0.003 | 7.13 ± 0.57 | 33.32 ± 1.57 | 2.392 ± 0.73 | 09.50 ± 0.31 |
| F10 | 126.23 ± 0.03 | 0.80 ± 0.025 | 7.21 ± 0.41 | 37.46 ± 1.23 | 2.538 ± 0.21 | 10.50 ± 1.90 |
| F11 | 126.78 ± 0.21 | 0.82 ± 0.001 | 7.33 ± 0.81 | 38.07 ± 1.08 | 2.563 ± 0.71 | 10.70 ± 0.23 |
Thickness and thickness uniformity
The thicknesses of ten discs of each type of formulation were measured and were found to be in the range 0.75–0.86 mm. Table 2 shows the means (±SD) of the obtained thickness values. The low SD values reflected the uniform thickness.Mean tensile strengths of all eleven formulations are shown in Table 3. These strength values were all more than 7 kg mm−1,2 indicative of good mechanical strength. Moreover, the tensile strength increased with increasing poloxamer mass. Using design expert software, a quadratic model was derived and an F-value of 62.02 (p < 0.0500) was obtained, which implied that the model was significant. The relationship between tensile strength and the independent factors was determined to follow the equation
| Tensile strength = +14.68 + 1.56A + 2.92B − 0.84AB + 0.69A2 + 1.21B2. |
| Formulation code | Factor 1: polymer mass (mg) | Factor 2: poloxamer mass (mg) | Response 1: tensile strength (kg mm−2) (mean ± SD) (n = 3) | Response 2: maximum drug release (%) (mean ± SD) (n = 3) |
|---|---|---|---|---|
| a F06 was selected as the optimal formulation and used as the subject for further studies. | ||||
| F01 | 1 | 1 | 20.23 ± 1.86 | 98.62 ± 0.36 |
| F02 | 0 | 0 | 15.94 ± 0.04 | 97.50 ± 1.20 |
| F03 | −1 | 1 | 13.68 ± 0.02 | 95.01 ± 0.44 |
| F04 | 0 | −1 | 18.89 ± 0.09 | 98.04 ± 0.29 |
| F05 | −1 | −1 | 14.57 ± 1.01 | 96.89 ± 0.71 |
| F06a | 1 | −1 | 17.87 ± 0.32 | 97.60 ± 0.21 |
| F07 | −1 | 0 | 15.10 ± 0.51 | 97.41 ± 0.38 |
| F08 | 0 | 1 | 19.23 ± 0.26 | 98.26 ± 0.31 |
| F09 | 1 | 0 | 14.01 ± 0.71 | 96.20 ± 0.82 |
| F10 | 0 | 0 | 15.72 ± 0.23 | 97.00 ± 0.20 |
| F11 | 0 | 0 | 15.54 ± 0.01 | 96.90 ± 0.80 |
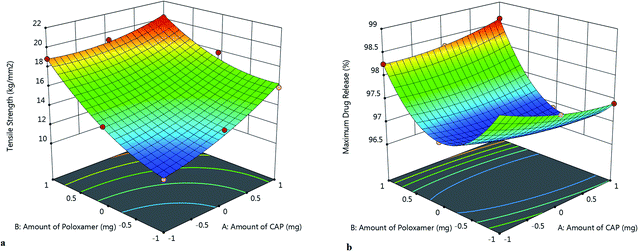
ANOVA results showed A, B, AB, A2, B2 to be significant model terms with p < 0.05. Factors A (CAP mass) and B (poloxamer mass) were concluded from this equation to have positive effects on tensile strength. Interaction terms (AB) showed negative effects on tensile strength whereas higher-order terms (A2 and B2) showed positive effects on tensile strength. Also the effects of the two independent variables on the tensile strength were studied by producing a 3D response surface plot that is shown in Fig. 2a.
| Maximum% drug release = +96.85 − 0.0683A + 0.3567B + 0.2475AB + 0.1161A2 + 1.11B2. |
ANOVA results revealed A, B, A,2 B2 and AB to be significant model terms. Here we observed that factor A (mass of CAP) produced a significant negative effect on maximum drug release whereas factor B (mass of poloxamer), interaction terms, and higher-order terms produced positive effects on the maximum drug release.
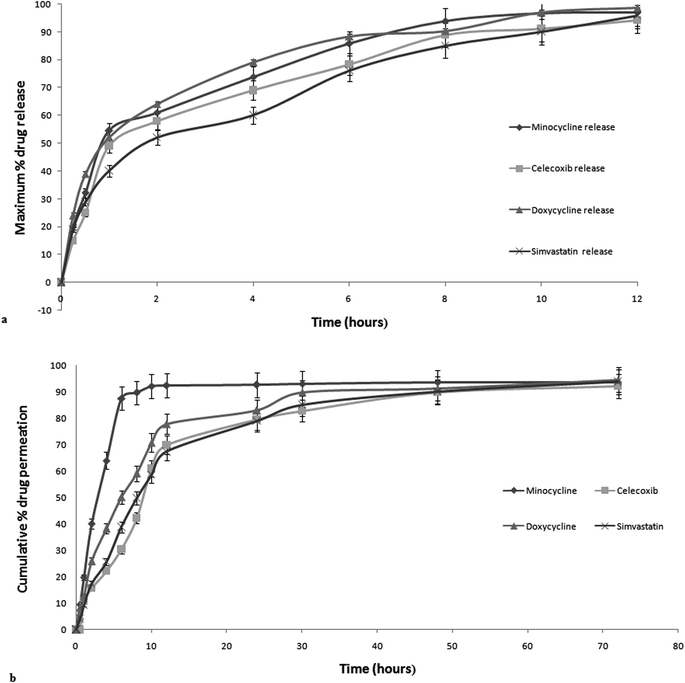
To study the effects of the two independent variables on the in vitro drug release, a 3D-response surface plot was constructed and is shown in Fig. 2b. Inspection of the plot indicated that as the masses of CAP and poloxamer were increased, the percentage of the drug released from the formulation first decreased and then increased. This behavior might have been the outcome of an association of the two polymers. By comparing the values of tensile strength and in vitro release, F06 (70 mg of CAP and 30 mg of poloxamer) was selected as the optimal formulation and further studies were performed on it. The maximum percentage drug releases of minocycline, celecoxib, doxycycline and simvastatin from this formulation are shown in Fig. 3a and were found to be in the range 94–98%. The differences between the percent releases of the four drugs may have been due to their respective different solubilities in the simulated isotonic phosphate buffer.
| Time of sampling (min) | Diameter of zone of inhibition (mm) | ||
|---|---|---|---|
| E. coli (CC25922) (mean ± SD) (n = 3) | S. aureus (CC25923) (mean ± SD) (n = 3) | Streptococcus mutans (CC25175) (mean ± SD) (n = 3) | |
| 15 | 28 ± 1.0 | 29 ± 1.3 | 28 ± 1.24 |
| 30 | 29 ± 1.3 | 30 ± 2.4 | 28 ± 0.89 |
| 60 | 30 ± 2.1 | 32 ± 1.7 | 30 ± 2.0 |
| 120 | 31 ± 1.5 | 32 ± 0.9 | 33 ± 1.03 |
| 240 | 32 ± 1.2 | 34 ± 1.23 | 35 ± 0.34 |
| 480 | 34 ± 2.2 | 35 ± 0.73 | 37 ± 0.75 |
| 720 | 36 ± 0.8 | 37 ± 2.1 | 38 ± 0.31 |
Conclusions
A polymeric gingiva disc displaying muco-retention, the controlled release of embedded drugs, and bioerosion was successfully designed and developed. When subjected to in vitro and in vivo characterization, it revealed its suitability for completely treating acute-phase periodontitis. The developed formulation is proposed to be an all-encompassing treatment modality for acute phase periodontitis.Abbreviations
| CAP | Cellulose acetate phthalate |
| MMP | Matrix metalloproteinase |
| BMP | Bone morphogenic protein |
| CFU | Colony forming unit |
| PDL | Periodontal ligament |
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors thank the Department of Pharmaceutics, School of Pharmaceutical Education and Research, Jamia Hamdard, New Delhi, India, for providing essential facilities for this research. The authors are also thankful to the Department of Microbiology and Department of Dentistry HIMSR for providing guidance in the research.References
- G. C. Armitage, Periodontol, 2004, 34, 9–21, DOI:10.1046/j.0906-6713.2002.003421.x.
- S. Kothari, G. Gnanaranjan and P. Kothiyal, Int. J. Drug. Res.Technol., 2012, 2, 411–421 Search PubMed.
- M. A. da Silva, R. N. Oliveira, R. H. Mendonca, T. G. Lourenco, A. P. V. Colombo, M. N. Tanaka, E. M. Tude, M. F. da Costa and R. M. S. Thire, J. Biomed. Mater. Res., Part B, 2016, 104, 106–115, DOI:10.1002/jbm.b.33357.
- S. Pragati, S. Ashok and S. Kuldeep, Int. J. Drug Delivery, 2009, 1, 1–14 CrossRef CAS.
- S. C. Sundararaj, M. V. Thomas, R. Peyyala, T. D. Dziubla and D. A. Puleo, Biomaterials, 2013, 34, 8835–8842, DOI:10.1016/j.biomaterials.2013.07.093.
- S. Vyas, V. Sihorkar and V. Mishra, J. Clin. Pharm. Ther., 2000, 25, 21–42, DOI:10.1046/j.1365-2710.2000.00261.x.
- A. Puratchikody, V. Prasanth, S. T. Mathew and A. Kumar, Int. J. Drug Delivery, 2011, 3, 171–184 CAS.
- S. G. Ciancio, M. L. Mather and J. A. McMullen, J. Periodontol., 1980, 51, 530–534, DOI:10.1902/jop.1980.51.9.530.
- B. O’connor, H. Newman and M. Wilson, J. Periodontol., 1990, 61, 228–233, DOI:10.1902/jop.1990.61.4.228.
- M. Pinho, N. de, L. B. Pereira, S. L. S. Souza, D. B. de Palioto, M. F. Grisi, M. de, A. B. Novaes Jr and M. Taba Jr, Braz. Dent. J., 2008, 19, 323–328, DOI:10.1590/s0103-64402008000400007.
- G. Emingil, G. Atilla, T. Sorsa, H. Luoto, L. Kirilmaz and H. Baylas, J. Periodontol., 2004, 75, 106–115, DOI:10.1902/jop.2004.75.1.106.
- T. Larsen, Oral Microbiol. Immunol., 2002, 17, 267–271, DOI:10.1034/j.1399-302x.2002.170501.x.
- G. Mundy, R. Garrett, S. Harris, J. Chan, D. Chen, G. Rossini, B. Boyce, M. Zhao and G. Gutierrez, Science, 1999, 286, 1946–1949, DOI:10.1126/science.286.5446.1946.
- J. Ali, R. Khar, A. Ahuja and R. Kalra, Int. J. Pharm., 2002, 238, 93–103, DOI:10.1016/s0378-5173(02)00059-5.
- M. Sugiyama, T. Kodama, K. Konishi, K. Abe, S. Asami and S. Oikawa, Biochem. Biophys. Res. Commun., 2000, 271, 688–692, DOI:10.1006/bbrc.2000.2697.
- J. H. Jeon, W. T. Piepgrass, Y. L. Lin, M. V. Thomas and D. A. Puleo, J. Periodontol., 2008, 79, 1457–1464, DOI:10.1902/jop.2008.080004.
- J. H. Jeon, M. V. Thomas and D. A. Puleo, Int. J. Pharm., 2007, 340, 6–12, DOI:10.1016/j.ijpharm.2007.03.007.
- A. T. Raiche and D. A. Puleo, IEEE Eng. Med. Biol. Mag., 2003, 22, 35–41, DOI:10.1109/memb.2003.1256270.
- X. Xu and P. I. Lee, Pharm. Res., 1993, 10, 1144–1152, DOI:10.1023/a:1018960016756.
- B. Satishbabu and B. Srinivasan, Indian J. Pharmaceut. Sci., 2008, 70, 175, DOI:10.4103/0250-474X.41451.
- S. Singh, D. Verma, M. A. Mirza, A. K. Das, M. Dudeja, M. K. Anwer, S. Talegaonkar and Z. Iqbal, J. Drug. Deliv. Sci. Technol., 2017, 39, 95–103, DOI:10.1016/j.jddst.2017.03.007.
- R. A. Shellis, Arch. Oral Biol., 1978, 23, 485–489, DOI:10.1016/0003-9969(78)90081-X.
- M. Srivastava, Y. R. Neupane, P. Kumar and K. Kohli, Drug. Deliv., 2016, 23, 2228–2234, DOI:10.3109/10717544.2014.958625.
- A. Györfi, A. Fazekas, Z. Suba, F. Ender and L. Rosivall, J. Clin. Periodontol., 1994, 21, 601–605, DOI:10.1111/j.1600-051x.1994.tb00750.x.
- Y. Xu and W. Wei, Arch. Oral Biol., 2006, 51, 794–803, DOI:10.1016/j.archoralbio.2006.03.018.
- M. A. Botelho, J. G. Martins, R. S. Ruela, D. B. Queiroz and W. S. Ruela, J. Appl. Oral Sci., 2010, 18, 335–342, DOI:10.1590/s1678-77572010000400003.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c9ra09569a |
| This journal is © The Royal Society of Chemistry 2020 |