Total synthesis of isatindigotindoline C†
Abstract
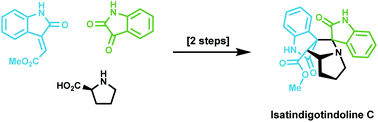
Total synthesis of isatindigotindoline C, a 3,3′-spiropyrrolidine oxindole alkaloid, is achieved in two steps using an exo-selective decarboxylative 1,3-dipolar cycloaddition as the key step. The synthesis verifies the originally assigned relative anti-stereochemistry for the bis-oxindole core of isatindigotindoline C.
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...