Integrated nano-architectured photocatalysts for photochemical CO2 reduction
Abstract
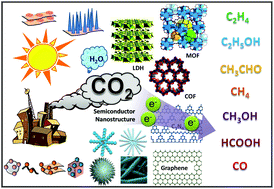
Recent advances in nanotechnology, especially the development of integrated nanostructured materials, have offered unprecedented opportunities for photocatalytic CO2 reduction. Compared to bulk semiconductor photocatalysts, most of these nanostructured photocatalysts offer at least one advantage in areas such as photogenerated carrier kinetics, light absorption, and active surface area, supporting improved photochemical reaction efficiencies. In this review, we briefly cover the cutting-edge research activities in the area of integrated nanostructured catalysts for photochemical CO2 reduction, including aqueous and gas-phase reactions. Primarily explored are the basic principles of tailor-made nanostructured composite photocatalysts and how nanostructuring influences photochemical performance. Specifically, we summarize the recent developments related to integrated nanostructured materials for photocatalytic CO2 reduction, mainly in the following five categories: carbon-based nano-architectures, metal–organic frameworks, covalent-organic frameworks, conjugated porous polymers, and layered double hydroxide-based inorganic hybrids. Besides the technical aspects of nanostructure-enhanced catalytic performance in photochemical CO2 reduction, some future research trends and promising strategies are addressed.
- This article is part of the themed collections: Nanoscale 2021 Lunar New Year Collection, Recent Review Articles and Nanoscale Most Popular 2020 Articles


 Please wait while we load your content...
Please wait while we load your content...