Stretchable, self-healing and tissue-adhesive zwitterionic hydrogels as strain sensors for wireless monitoring of organ motions†
Abstract
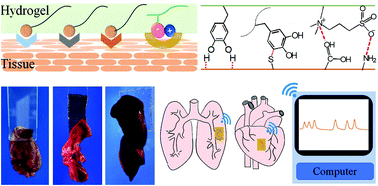
Skin-inspired sensors have great potential applications in wearable and implantable devices to monitor human motions. Robust tissue adhesion, fatigue resistance, and biocompatibility are desired for in situ signal capture and wireless transmission. Inspired by the mussel and zwitterionic adhesion mechanisms, we prepared novel stretchable, self-healing and polydopamine zwitterionic nanocomposite hydrogels that provide reversible and robust adhesion to tissues with a strength up to 19.4 kPa and a strain sensitivity of 4.3. The hydrogel sensors robustly adhere to organs like the heart, liver, and lungs to capture signals for remote monitoring and diagnostics through wireless transmission.
- This article is part of the themed collections: Materials Horizons 10th anniversary regional spotlight collection: China, 2020 Horizons Outstanding Paper Award Winners, Materials Horizons Lunar New Year collection 2021, 2020 Materials Horizons most popular articles and Horizons Community Board Collection: Biosensors


 Please wait while we load your content...
Please wait while we load your content...