High-heat treatment enhanced catalytic activity of CuO/CeO2 catalysts with low CuO content for CO oxidation
Abstract
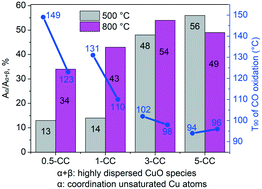
A series of CuO/CeO2 catalysts with different CuO loadings (0.5, 1, 3, and 5 wt%) were prepared by an impregnation method, and tested for the total oxidation of CO. The samples were characterized by various structural and textural detection techniques including XRD, N2 sorption, TEM-EDS, H2-TPR, XPS, and in situ DRIFTS. Unexpectedly, the low CuO loading (0.5–3 wt%) samples calcined at high temperature (800 °C) exhibited better catalytic performance than those calcined at 500 °C, and the catalytic activity gap between the catalysts calcined at 500 °C and 800 °C shrunk with the increase of copper content. In contrast, when the loading amount of CuO reached a normal value (5 wt%), the CuO/CeO2 catalyst with low temperature calcination (500 °C) obtained a slightly better activity than that calcined at 800 °C. The results showed that, for the catalysts with low CuO content (0.5–3 wt%), the higher temperature calcination (800 °C) can promote the formation of the coordinatively unsaturated copper atoms and increase the content of active Cu species, resulting in a higher activity than those calcined at 500 °C. But this effect was suppressed when the CuO content reached 5 wt%. The coordinatively unsaturated copper atoms, as well as Cu+, were confirmed to be the main active sites of the CuO/CeO2 catalysts.


 Please wait while we load your content...
Please wait while we load your content...