Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Probing batch and continuous flow reactions in organic solvents: Granulicella tundricola hydroxynitrile lyase (GtHNL)†
José
Coloma
ab,
Yann
Guiavarc'h
ac,
Peter-Leon
Hagedoorn
 a and
Ulf
Hanefeld
a and
Ulf
Hanefeld
 *a
*a
aBiokatalyse, Afdeling Biotechnologie, Technische Universiteit Delft, Van der Maasweg 9, 2629 HZ Delft, The Netherlands. E-mail: u.hanefeld@tudelft.nl
bUniversidad Laica Eloy Alfaro de Manabí, Avenida Circunvalación S/N, P.O. Box 13-05-2732, Manta, Ecuador
cLaboratory Reactions and Process Engineering, CNRS, LRGP, University of Lorraine, F-54000 Nancy, France
First published on 14th May 2020
Abstract
Granulicella tundricola hydroxynitrile lyase (GtHNL) is a manganese dependent cupin which catalyses the enantioselective synthesis of (R)-cyanohydrins. The GtHNL triple variant A40H/V42T/Q110H, previously reported to exhibit a high activity and stability, was immobilised on Celite R-633 by adsorption. The synthesis of (R)-mandelonitrile catalysed by immobilised enzyme in a rotating bed reactor was compared to a continuous flow reactor. A batch reaction was used as reference system and organic solvent (MTBE) was used as reaction medium to suppress the chemical background reaction, ensuring the synthesis of enantiopure cyanohydrin. The rotating bed reactor, designed to boost conversion rates due to enhanced mass transfer, did not greatly enhance the reaction displaying a rate 1.7 times higher than the reference batch model. Moreover, similar conversion (96% after 4 hours) and recyclability were observed as compared to the reference system. The continuous flow reactor displayed rates 2 and 3 times higher than the rotating bed and the reference batch systems, respectively. Good conversions were achieved within minutes (97% conversion in 4 minutes at 0.1 mL min−1). The immobilised enzyme displayed excellent enantioselectivity and high operational stability under all evaluated conditions. Overall, GtHNL triple variant A40H/V42T/Q110H immobilised on Celite R-633 is an excellent catalyst for the synthesis of (R)-mandelonitrile with a great potential for continuous flow production of cyanohydrins.
Introduction
Enzyme catalysed carbon–carbon bond forming reactions are important in organic chemistry to produce chiral compounds.1,2 In plants, hydroxynitrile lyases (HNLs) catalyse the cleavage of cyanohydrins into aldehydes or ketones releasing toxic hydrogen cyanide (HCN). This mechanism is a defense system against the attack of predators (cyanogenesis) and a source of nitrogen for the biosynthesis of L-asparagine (nitrogen fixation).3,4 The reverse reaction is of great interest as it enables the synthesis of chiral α-cyanohydrins (Scheme 1).The importance of cyanohydrins as platform molecules lies in their two functional groups, the hydroxyl and nitrile moiety, which can be converted into a variety of valuable chiral products such as α-hydroxy acids, primary and secondary β-hydroxy amines, α-hydroxy aldehydes or ketones, etc. All these compounds are known as platform molecules for the production of pharmaceutical and fine chemical products.1,5–8
Recently, a new manganese-dependent bacterial HNL was discovered in the soil bacterium Granulicella tundricola (GtHNL). The gene was heterologously expressed in Escherichia coli and the crystal structure was solved revealing a cupin fold.9 The wild type GtHNL (GtHNL-WT) catalysed the synthesis of (R)-mandelonitrile with a promising yield and enantioselectivity of 80% and 90% respectively. Site-saturation mutagenesis of active site amino acids produced a triple variant GtHNL-A40H/V42T/Q110H (GtHNL-TV) with a remarkable 490-fold-increase in specific activity in comparison to the wild type enzyme.10 EPR spectroscopy revealed an unusually high Lewis acidity for the Mn2+ as essential metal.11 Moreover Mn2+ was bound more tightly in the triple variant than in the wild type enzyme, which resulted in higher stability and activity.
In this study, we describe the immobilisation of GtHNL-TV on Celite R-633, the silicate skeletons of diatoms,12 for the synthesis of (R)-mandelonitrile in batch and continuous flow systems. Enzyme immobilisation plays an important role enhancing the enzyme stability toward harsh conditions such as extreme pH values, organic solvents, high ionic strengths, etc. Additionally, it allows a straightforward enzyme separation from the reaction mixture as well as the operation in continuous flow processes while minimizing the product contamination with enzymes.12–14 Celite was used as a carrier for enzyme immobilisation as it is an environmentally friendly material that has been successfully employed for the immobilisation of several HNLs enabling the production of (R)- and (S)-cyanohydrins with good yield, enantioselectivity and recyclability.5,15–17
Currently the vast majority of enzyme-catalysed conversions are performed in stirred tank reactors.2 To achieve full conversion extended reaction times are often required (affecting the productivity). Rapid stirring is required to avoid diffusion limitations. Especially at industrial scales, this induces shear forces that affect enzyme stability.18 To overcome these limitations, synthesis in a rotating bed reactor (RBR) and continuous flow reactor (CFR) are gaining attention. RBR enables efficient stirring and percolation of the substrates through the immobilised enzyme bed. This is suggested to result in improved mass transfer without mechanical enzyme attrition.19,20
Biosynthesis in continuous flow is also becoming an attractive way to increase productivity, reduce enzyme inhibition and facilitate downstream processing.21–26 Additionally reaction volumes are reduced, increasing safety, in particular for toxic compounds such as cyanide.27 Several enzymes have been tested in continuous flow systems such as HNLs,21,25 transaminases,26,28–30 oxidoreductases,31–33 and aldolases.34,35
The aim of this work was to evaluate whether continuous flow reactions facilitate process intensification compared to a rotating bed reactor, reducing shear forces, improving stability and activity of the enzyme. For this purpose, GtHNL-TV was immobilised on Celite R-633 and its catalytic performance and stability were evaluated in RBR and CFR and compared to a batch reaction under the same reaction conditions.
Results and discussion
Celite is an environmentally benign siliceous carrier material, produced by diatoms, a type of microalgae.38 Several HNLs were immobilised on this environmentally friendly material and performed better than on other carriers. Prunus amygdalus HNL (PaHNL) immobilised on Celite was compared to Avicel,39 controlled pore glass and Sephadex,17 in all cases Celite was the best carrier in terms of enzymatic activity. Hevea brasiliensis HNL (HbHNL) immobilised on Celite gave rise to better enantioselectivity compared to Avicel and EP-700 (hydrophobic polyamide),40 and the very acid sensitive Arabidopsis thaliana HNL (AtHNL) had enhanced stability towards acidic pH values and organic solvents when it was immobilised on Celite R-633.16 The ability of Celite to bind water, enabling a local environment surrounding the enzyme with organic solvents, might explain these results.38,41 Because of these favourable results and to ensure comparability with previous studies Celite R-633 was utilised as a carrier material.Batch reactions
Both, purified GtHNL-WT and GtHNL-TV were immobilised on Celite R-633. All batch reactions were performed at 5 °C since it was reported earlier9 that a significantly higher enantiomeric excess can be obtained under this condition compared to the reaction at 15 °C. After immobilisation, the GtHNL-TV showed considerably higher activity and selectivity compared to GtHNL-WT (Fig. 1), which is in line with earlier results obtained for the enzyme in solution.10The specific activity of GtHNL-TV was 56.5 ± 18 U mg−1 which is 63 times higher compared to the wild type enzyme under the same reaction conditions. This can be ascribed to the additional histidines introduced at positions 40 and 110, improving the deprotonation of the hydrogen cyanide and giving rise to enhanced conversion and enantioselectivity.10,11 At the same time these mutations greatly improve the binding of the metal to the active site, indeed metal removal was very difficult.11 Since GtHNL-TV proved to be a better catalyst than the wild type enzyme, only the variant enzyme henceforth was tested for the synthesis of (R)-mandelonitrile.
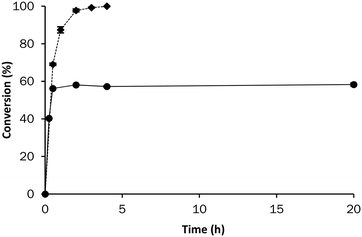
Having established Celite R-633 as suitable carrier on which the enzyme displayed similar activity as in solution, a leaching test was performed (Fig. 2). In earlier studies the structurally unrelated PaHNL and AtHNL were found not to leach from Celite R-633.15,16 As was earlier shown for AtHNL16 the GtHNL-TV was found to be active in organic solvents and at low pH without immobilisation (Fig. S2†). However, it precipitated during the reaction making reuse impossible. In the leaching experiment, the immobilised enzyme was removed from the reaction medium after 30 minutes of enzyme catalysed conversion. A high enzyme-support ratio (4 U mg−1) was used intentionally to clearly see any enzyme leaching to the reaction medium. After removal of the enzyme, the reaction did not proceed anymore, demonstrating that no active GtHNL-TV leached from the carrier into the reaction medium (Fig. 2). The hydrophilic characteristics of the enzyme – carrier and the insolubility of the enzyme in organic solvents explain this result.12
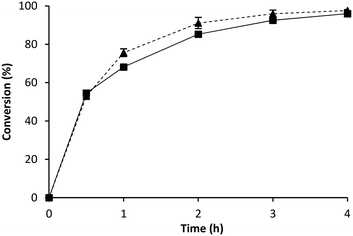
Having firmly established that GtHNL-TV was successfully immobilised on Celite R-633, the enzyme loading for the synthesis of (R)-mandelonitrile in batch reactions (BR) was studied. As described earlier for PaHNL the immobilised enzyme was placed tightly packed inside tea bags15 (Fig. S7, Table S1†). Nearly complete conversion and excellent enantioselectivity (ee > 99%) were achieved after 4 hours of reaction time, regardless of the enzyme loading (Fig. 3). Interestingly, higher enzyme loadings did not show faster conversion, indicating that the reaction is mass transfer limited at high enzyme loading.
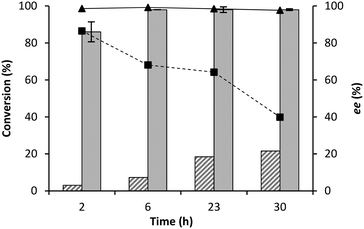
A recycling study was performed utilising 1 U mg−1GtHNL-TV immobilised on Celite R-633. With this low catalyst loading any loss of activity will be observed directly while higher catalysts loading might mask an initial activity loss.5,15 The biocatalyst exhibited good recyclability, conversions gradually dropped to >70% over all cycles but remarkable high enantioselectivity (>99%) was observed during all 8 cycles (Table 1).
| Cycle | Conversion (%) | ee (R)-mandelonitrile (%) |
|---|---|---|
Conditions: ratio benzaldehyde![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HCN in buffered MTBE, pH 4, 1 HCN in buffered MTBE, pH 4, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 100 μL benzaldehyde (1 mmol), 2 ml HCN solution in acetate buffered MTBE (1.5–2 M) pH 4, 27.5 μL 1,3,5-tri-isopropylbenzene (0.1 mmol, internal standard), a tea bag filled with GtHNL-TV immobilised on 50 mg Celite R-633 (1 U mg−1 = 50 U). The reaction was stirred at 700 rpm at 5 °C; reaction time: 4 h. The enzyme was washed for 1 minute with 100 mM acetate buffer saturated MTBE, pH 4, after each cycle. 4, 100 μL benzaldehyde (1 mmol), 2 ml HCN solution in acetate buffered MTBE (1.5–2 M) pH 4, 27.5 μL 1,3,5-tri-isopropylbenzene (0.1 mmol, internal standard), a tea bag filled with GtHNL-TV immobilised on 50 mg Celite R-633 (1 U mg−1 = 50 U). The reaction was stirred at 700 rpm at 5 °C; reaction time: 4 h. The enzyme was washed for 1 minute with 100 mM acetate buffer saturated MTBE, pH 4, after each cycle.
|
||
| 1 | 98.0 ± 0.2 | >99 |
| 2 | 90.0 ± 0.3 | >99 |
| 3 | 88.0 ± 0.9 | 98.7 |
| 4 | 88.0 ± 0.1 | >99 |
| 5 | 87.0 ± 0.1 | >99 |
| 6 | 77.0 ± 0.7 | >99 |
| 7 | 74.0 ± 1.0 | >99 |
| 8 | 73.0 ± 0.4 | >99 |
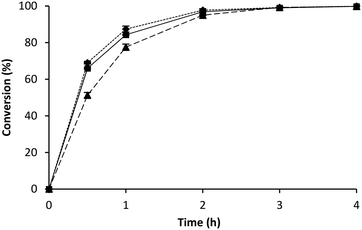
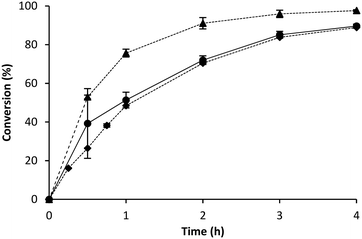
With the BR as the reference point, the comparison to the RBR could be performed. The reaction volume was scaled up circa 40 times to evaluate the mass transfer influence on the kinetics of the reaction in a RBR. This device has been designed to improve mass transfer, combining the advantages of fixed bed and stirred tank reactors.42 At the same time it also displays the typical safety disadvantage of batch reactions; a large scale requires a large amount of a toxic compound in a vessel.27 A first comparison between BR and RBR showed higher reaction rates for the BR (Fig. 4). Surprisingly, when the same immobilised enzyme was placed tightly packed in the above mentioned tea bags into the RBR the conversions and enantioselectivities were enhanced along the reaction times, displaying a similar feature to the batch reaction (Fig. 5).
These results are unexpected since the RBR has been designed to boost the efficiency in biocatalytic reactions by reducing diffusion limitations. However, in an earlier study comparing a RBR and a stirred tank reactor, i.e. a BR; similar conversions were found in both cases. The transaminase and lipase catalysed kinetic resolution of (R,S)-1-phenylethylamine and (R,S)-1-phenylethanol respectively were utilised for that comparison.20
Tables 2 and 3 show a clear effect of the packing on the GtHNL-TV recyclability in the RBR. After the first cycle without bag (Table 3), the immobilised enzyme was placed tightly packed into tea bags. Tightly packed enzymes were more stable than loosely packed enzymes over 4 cycles. A possible explanation might be higher shear forces exerted on the GtHNL-TV immobilised on Celite freely placed or loosely packed into the RBR, when compared to tightly packed biocatalyst. Shear forces might result in breaking or stretching molecular bonds. Recovery of the enzyme can occur when the shear force is removed.43 A tightly packed enzyme is better protected against shear forces. The decrease in enantiomeric excess during the first cycle (Table 3, cycle 1), can be explained by a more pronounced chemical background reaction when the immobilised enzyme is placed freely inside the RBR.15
| Cycle | Conversion (%) | ee (R)-mandelonitrile (%) |
|---|---|---|
| Conditions: 85 mL HCN (1.5–2 M) in 100 mM acetate buffered MTBE, pH 4, 4.25 mL (42 mmol) benzaldehyde, 1.16 mL (4.2 mmol) 1,3,5 tri-isopropylbenzene as internal standard (I.S.), immobilised enzyme on 773 mg Celite (1 U mg−1 = 773 U), 700 rpm, 5 °C. The enzyme was washed for 1 minute with acetate buffer saturated MTBE, pH 4, after each cycle. | ||
| 1 | 88.9 ± 0.2 | >99 |
| 2 | 79.0 ± 0.6 | >99 |
| 3 | 82.9 ± 0.3 | >99 |
| 4 | 60.7 ± 0.2 | >99 |
| Cycle | Conversion (%) | ee (R)-mandelonitrile (%) |
|---|---|---|
| Conditions: 85 mL HCN (1.5–2 M) in acetate buffered MTBE, 4.25 mL (42 mmol) benzaldehyde, 1.16 mL (4.2 mmol) 1,3,5 tri-isopropylbenzene as I.S., immobilised enzyme on 773 mg Celite (1 U mg−1 = 773 U), 700 rpm, 5 °C. The enzyme was washed for 1 minute with acetate buffer saturated MTBE, pH 4, after each cycle. a Immobilised GtHNL-TV was used without tea bags. b Immobilised GtHNL-TV was placed in tightly packed tea bags. | ||
| 1a | 90.3 ± 0.3 | 85.4 |
| 2b | 96.0 ± 0.2 | 99.3 |
| 3b | 93.5 ± 0.3 | 96.3 |
| 4b | 84.8 ± 0.2 | 99.4 |
For PaHNL immobilised on Celite this influence of the packing was observed, too.15 A faster racemic background reaction for loosely packed enzyme was observed in that case as well. Substrate inhibition affecting the RBR reaction by blocking the enzyme active site due to local high concentrations of benzaldehyde or HCN was ruled out by kinetic measurements (Fig. S3†). These results (Tables 1–3) show that the recyclability of the enzyme is similar in both batch systems (BR and RBR) when using tightly packed, immobilised enzyme.
Continuous flow reactions
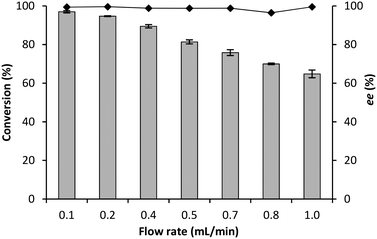
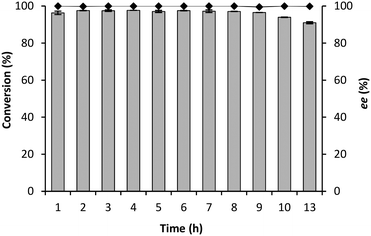
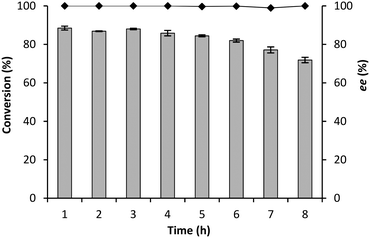
To maximally exploit the potential safety advantage of the flow chemistry, the synthesis of (R)-mandelonitrile was evaluated at different flow rates in a CFR of just 1 mL. As expected, a decrease in conversion from 97% to 63% (Fig. 6) was observed by increasing the flow rate from 0.1 mL min−1 to 1.0 mL min−1 (residence time: 240 s to 24 s).21,40 More remarkably the enantioselectivity was not influenced although all these experiments were performed at room-temperature, while cooling to 5 °C had been necessary to achieve good enantioselectivity in the BR and RBR.The stability of GtHNL-TV was evaluated at 0.1 and 0.2 mL min−1, conditions under which complete conversion was (just) observed. Any weaknesses of the system will immediately be revealed at these flow rates. High stability was observed during 13 and 8 hours respectively (Fig. 7 and 8). Remarkably the enantioselectivity remained excellent even when the conversion dropped due to enzyme deactivation. In the case of Manihot esculenta HNL (MeHNL) and Hevea brasiliensis HNL (HbHNL), immobilised on siliceous monoliths, this was not the case, as loss of activity was accompanied by loss of enantioselectivity.21 The biocatalytic synthesis of (R)-mandelonitrile in continuous flow using AtHNL immobilised on Celite R-633 has been reported previously.25 With a packed bed reactor (microbore column 3 mm/50 mm), the best conversion (85%) and enantioselectivity (96%) were achieved with 25 mg of pure AtHNL on 100 mg of Celite at a residence time of 35.3 min. Clearly, the conversions reported here (Fig. 6) are a step forward.
Comparison of the reactors
The different reactors can best be compared via specific rates and productivity, expressed as space–time yield (STY). In batch reactions, the RBR showed a specific rate 1.7 times higher compared to the BR, whereas the CFR proved to be 3 and 2 times faster than BR and RBR respectively (Table 4). Importantly, almost full conversion and excellent enantioselectivities were obtained within minutes instead of 4 hours (batch reactions). In addition to this much higher rate, the substantially lower reaction volume in the CFR constitutes a significant improvement of safety.27The increase in productivity of the CFR can also be explained by the apparent turnover number (kapp) observed. BR and RBR displayed kapp from 0.77 s−1 and 1.32 s−1 respectively. The CFR exhibited 1.4 s−1 (0.1 mL min−1) to 9.2 s−1 (1 mL min−1) (Fig. 6) without reaching the maximum kapp, thus the enzyme is capable of converting even more substrate. In spite of the large macropores of Celite R-633 (6.5 μm average diameter),44 which are favourable to internal mass transfer, differences between the reactor types become apparent. In all reactors with heterogeneous processes such as with these mesoporous materials, some boundary layer limiting substrate and product transfer occurs. This contributes to mass transfer limitations and consequently, turnover rate limitations. Increased flow rates improve the kapp due to a reduction and almost depletion of this boundary layer, enabling more substrate to be exposed to the enzyme active site, explaining the advantage of CFR over other reactors.45–47
The STY, a parameter frequently used to evaluate the productivity of different systems normalized to a volume of 1 L, shows that the use of the continuous flow system resulted in a prominent increase in (R)-mandelonitrile synthesized (gproduct h−1 L−1). In steady state conditions, both batch reactions (BR and RBR) achieved circa 12 g h−1 L−1 whereas CFR at 0.1 mL min−1 reached 784 g h−1 L−1. This represents 65 times more product in total. Importantly, increasing the flow rate enables higher specific rates and therefore higher STY without significantly affecting the enzyme stability (Fig. 7 and 8). However, it is worthy to point out that higher flow rates lead to unreacted substrate, which may make downstream processing more difficult. Taking into account the amount of enzyme used for the reaction, the STY was 23 gproduct h−1 L−1 mgenz−1 at 0.1 mL min−1 up to 156 gproduct h−1 L−1 mgenz−1 at 1 mL min−1, which shows excellent productivity with a low enzyme loading.
Recently, the performance of MeHNL and HbHNL immobilised on porous, monolithic silica supports has been reported in a continuous flow microreactor. Full conversion and high enantioselectivity were achieved within minutes, but the enzyme stability diminished after 7 and 3 hours operation, respectively. Furthermore, a drastic improvement of the catalytic performance was observed as compared with the batch system, with a 8-fold increase of the specific reaction rate.21
Conclusion
GtHNL-TV showed a better catalytic performance for the production of (R)-mandelonitrile compared to the wild type enzyme. Nearly complete conversion and high enantioselectivity were achieved in both BR and RBR systems with tightly packed enzyme on a readily available and environmentally benign carrier, Celite R-633. The RBR did not greatly enhance the reaction rate and showed only a 1.7-fold increase in specific rate at 54% conversion but similar STY (gproduct h−1 L−1). By switching to a CFR, full conversions and excellent enantioselectivity were obtained within minutes. Furthermore, continuous flow enabled to operate at higher kapp which resulted in a tremendous increase in STY compared to both batch systems evaluated in this study. Additionally, the much smaller reaction volume improves safety at the same time. The high activity and enantioselectivity of immobilised GtHNL-TV together with the enhanced stability in batch and continuous flow systems outperform what has been reported for other HNLs and makes this enzyme a new competitor for the production of chiral cyanohydrins.Experimental section
Chemicals
All chemicals were bought from Sigma Aldrich (Schnelldorf, Germany) unless reported otherwise. Isopropanol and heptane were of HPLC grade (≥99%) and used as HPLC solvents. 1,3,5-triisopropylbenzene (97%) was from Fluka Chemie (Buchs, Switzerland). Potassium cyanide (KCN, 97%) from J.T. Baker (Deventer, The Netherlands) was used as cyanide source in the HCN solution. (±)-Mandelonitrile from Across Organics (New Jersey, USA) was purified by flash chromatography (PE/MTBE 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1/3
1/3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7).
7).
Heterologous production of wild type GtHNL (GtHNL-WT)
The pET-28a-GtHNL expression plasmid containing the GtHNL gene codon optimized for E. coli (ESI† A) was obtained from Bio Basic INC (Canada). E. coli BL21(DE3) was transformed with the expression plasmid. The production of GtHNL-WT was performed according to literature.10 A preculture was prepared by inoculating one single colony of E. coli BL21(DE3)–pET28aGtHNL in 10 mL of LB medium with kanamycin (40 μg mL−1) and incubated overnight (New Brunswick Scientific Incubator Shaker Excella E24 Series) at 37 °C, 180 rpm. Then, this preculture was used for the inoculation of 1 L of LB medium containing kanamycin (40 μg mL−1) and incubated at 37 °C, 120 rpm. When the OD600 reached 0.7–0.9 the gene expression was induced by adding 1 mL of 0.1 M isopropyl β-D-thiogalactoside (IPTG) per liter of culture (0.1 mM IPTG final concentration) and cultivation was continued at 25 °C, 120 rpm for 22 hours. Moreover, 100 μL of 1 M MnCl2 was added per liter of culture at the induction time (0.1 mM Mn2+ final concentration). Cells were harvested at 4 °C, 3600 rpm during 20 minutes (Sorvall RC6, Thermo Scientific). The supernatant was discarded and the pellet was washed with 20 mL of 10 mM sodium phosphate buffer, pH 7, and stored at −80 °C.Cloning and expression of triple variant GtHNL-A40H/V42T/Q110H (GtHNL-TV)
The pUC57 shuttle vector containing the gene encoding GtHNL-A40H/V42T/Q110H, codon optimised for E. coli (ESI† A) was obtained from Bio Basic INC (Canada) and used to transform E. coli Top 10. The gene encoding GtHNL-A40H/V42T/Q110H gene was cloned into pET28a expression vector using NcoI and HindIII restriction enzymes. The resulting pET28a-GtHNL-A40H/V42T/Q110H expression vector was cloned into E. coli TOP 10 to obtain a stable host for plasmid DNA. Finally, pET28a-GtHNLA-40H/V42T/Q110H was used to transform the expression host E. coli BL21(DE3). The cultivation of the expression strain was performed in TB (terrific broth) medium following the same procedure described before for the GtHNL wild type.Purification of GtHNL-WT and GtHNL-TV
GtHNL-WT was purified according to the literature10 with slight modifications. The pellets were resuspended in lysis buffer A (50 mM bis-Tris buffer + 30 mM NaCl + DNAse) pH 6.8, respectively, and lysed in a cell disruptor (Constant Systems Ltd., United Kingdom) at 1.5 kBar and 4 °C to avoid protein denaturation. The cell free extract (CFE) was collected as the supernatant after centrifugation at 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g, 1 h, 4 °C. GtHNL-WT was purified from the CFE by anion exchange chromatography with Q Sepharose Fast Flow columns (HiTrap Q FF, 70 mL; GE Healthcare, Uppsala, Sweden) applying an isocratic step of 10% buffer B and then a gradient from 10% to 100% buffer B (50 mM bis-Tris buffer + 1 M NaCl). GtHNL-WT eluted at 10% buffer B. All the fractions were tested with an activity assay, see below.
000g, 1 h, 4 °C. GtHNL-WT was purified from the CFE by anion exchange chromatography with Q Sepharose Fast Flow columns (HiTrap Q FF, 70 mL; GE Healthcare, Uppsala, Sweden) applying an isocratic step of 10% buffer B and then a gradient from 10% to 100% buffer B (50 mM bis-Tris buffer + 1 M NaCl). GtHNL-WT eluted at 10% buffer B. All the fractions were tested with an activity assay, see below.
GtHNL-WT was further purified using ultrafiltration with 100 kDa MWCO Amicon filter (Millipore) in order to remove any large proteins (>100 kDa).
GtHNL-TV was purified following the same method with slight modifications. Loading and elution buffers were at pH 7.4 and the ultrafiltration step was omitted because it had a negative effect on the enzyme stability. GtHNL-TV eluted at 10% buffer B.
GtHNL activity assay
GtHNL activity (wild type and variant) was measured spectrophotometrically (Agilent Technologies Cary 60 UV-VIS) using a method previously reported.10,36 The cleavage of rac-mandelonitrile into benzaldehyde and hydrogen cyanide was followed at 280 nm and 25 °C in quartz glass cuvettes. To 1300 μL of reaction buffer (100 mM sodium oxalate buffer, pH 5), 200 μL of enzyme solution (diluted in reaction buffer) and 500 μL of 60 mM rac-mandelonitrile solution (dissolved in 3 mM oxalic acid, pH 3) were added. The background reaction was evaluated without enzyme and its slope was subtracted in the final calculation. The activity was calculated based on the following equation:36| Activity = 2.0/(ε280 × 1 × 0.2 ) [U ml−1 diluted sample] |
| ΔA/min = ΔA/minsample − ΔA/minblank |
| ε280 = 1.376 [mM−1 × cm−1] |
Preparation of the hydrogen cyanide (HCN) solution in MTBE
An HCN solution in MTBE was prepared as described previously5,15 with slight modifications. 25 mL MTBE and 10 mL MilliQ water were mixed in a 100 mL Erlenmeyer and kept at 0 °C. 0.1 mol potassium cyanide (6.51 g) was dissolved in the mixture and magnetically stirred for 15 minutes. 10 mL of 30% (v/v) HCl solution was added slowly and stirring was continued for 2 minutes. The HCN solution was allowed to reach room temperature (circa 20 °C). The organic and aqueous phases were separated using a separation funnel and the organic layer containing HCN was collected. The separation was performed twice more after adding 7 mL of MTBE each time. Finally, 5 mL of 100 mM sodium acetate buffer pH 4 was added to the organic fraction collected and it was stored in a dark bottle at 4 °C.The HCN concentration in solution in MTBE was determined by titration. 1 mL of the HCN solution was added to 5 mL of 2 M NaOH and magnetically stirred for 2 minutes. A small amount of potassium chromate was added as indicator, then the solution was titrated using 0.1 M silver nitrate. The cyanide reacts 1:1 with the silver and precipitates. If there are no cyanide ions left in the mixture it will change colour from light yellow to brown.5,37 To determine a concentration between 1.5–2 M is necessary to add 15–20 mL of silver nitrate. The HCN solution was found to be between 1.5 and 2 M.
Caution: Potassium cyanide (KCN) and hydrogen cyanide (HCN) are highly poisonous chemicals. All experiments involving KCN and HCN were performed in a ventilated fume hood with 2 calibrated HCN detectors (inside and outside the fume hood). HCN wastes were neutralized over a large excess of commercial bleach (15% sodium hypochlorite solution) for disposal.
Immobilisation on Celite R-633 for batch and continuous flow reactions
Enzyme immobilisation on Celite was performed according to literature.5 Celite R-633 was washed with 100 mM sodium acetate buffer pH 4 using a Büchner funnel and dried 24 h under vacuum in a desiccator over silica gel. Given volumes of wild type GtHNL or triple variant GtHNL were concentrated with Amicon ultrafiltration filters with a 10 kDa MW cut-off, and subsequently added dropwise to Celite R-633 and dried 24 h under vacuum in a desiccator over silica gel. The ratio of enzyme solution to carrier (μL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mg) was 2
mg) was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The enzyme concentration in solutions was adjusted in the concentration step to the required amount of enzyme for the immobilisation. By using this ratio of enzyme solution to Celite, the enzyme solution was completely absorbed by the carrier, ensuring that all the enzyme was immobilised into the porous material. The immobilised enzyme was stored in the fridge at 4 °C.
1. The enzyme concentration in solutions was adjusted in the concentration step to the required amount of enzyme for the immobilisation. By using this ratio of enzyme solution to Celite, the enzyme solution was completely absorbed by the carrier, ensuring that all the enzyme was immobilised into the porous material. The immobilised enzyme was stored in the fridge at 4 °C.
Synthesis reactions of (R)-mandelonitrile in batch systems
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.
4.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.
4.
Synthesis reactions of (R)-mandelonitrile on continuous flow
Immobilised GtHNL-TV on Celite R-633 (1 U mg−1) was placed into a 1 mL stainless steel flow reactor. It was filled with 150 mg of non-porous glass beads and 150 mg of Celite R-633 containing immobilised enzyme. The packed bed reactor had a reaction volume of 0.394 mL (ESI†-C). 20 cm of polytetrafluoroethylene (PTFE) tubing with 1.5 mm inner diameter connect a high-pressure pump (Knauer, Germany) with the starting materials. Initial conditions were as follow: 0.5 M benzaldehyde, 1.5–2 M HCN in 100 mM acetate buffered MTBE pH 4 and 50 mM 1,3,5 tri-isopropylbenzene as internal standard. The synthesis of (R)-mandelonitrile was evaluated at different flow rates (from 0.1 to 1 mL min−1) by chiral HPLC. The flow rate was checked at each sampling time by the difference of weight. Reactions were performed at room temperature.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol 95
2-propanol 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 in 1.5 mL Eppendorf tubes. A small amount of anhydrous magnesium sulphate (MgSO4) was used to remove the water from the solution. The Eppendorf tubes were centrifuged at 13000 rpm for 1 min. 850 μL of the supernatant was transferred to a 4 mL HPLC vial and 10 μL was injected into the HPLC (Chiralpak AD-H column, column size: 0.46 cm I.D × 25 cm). Heptane and 2-propanol were used as mobile phase with a flow rate of 1 mL min−1 and the UV detector was set at 216 nm. The column temperature was set at 40 °C. The samples in the autosampler were maintained at 4 °C.
5 in 1.5 mL Eppendorf tubes. A small amount of anhydrous magnesium sulphate (MgSO4) was used to remove the water from the solution. The Eppendorf tubes were centrifuged at 13000 rpm for 1 min. 850 μL of the supernatant was transferred to a 4 mL HPLC vial and 10 μL was injected into the HPLC (Chiralpak AD-H column, column size: 0.46 cm I.D × 25 cm). Heptane and 2-propanol were used as mobile phase with a flow rate of 1 mL min−1 and the UV detector was set at 216 nm. The column temperature was set at 40 °C. The samples in the autosampler were maintained at 4 °C.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
This project is financed by the Secretary of Higher Education, Science, Technology and Innovation of Ecuador and Universidad Laica Eloy Alfaro de Manabí (ULEAM).References
- J. Holt and U. Hanefeld, Curr. Org. Synth., 2009, 6, 15–37 CrossRef CAS.
- E. M. M. Abdelraheem, H. Busch, U. Hanefeld and F. Tonin, React. Chem. Eng., 2019, 4, 1878–1894 RSC.
- A. Nahrstedt, Plant Syst. Evol., 1985, 150, 35–47 CrossRef CAS.
- R. Lieberei, D. Selmar and B. Biehl, Plant Syst. Evol., 1985, 150, 49–63 CrossRef CAS.
- G. Torrelo, N. van Midden, R. Stloukal and U. Hanefeld, ChemCatChem, 2014, 6, 1096–1102 CrossRef CAS.
- M. North, Tetrahedron: Asymmetry, 2003, 14, 147–176 CrossRef CAS.
- M. Dadashipour and Y. Asano, ACS Catal., 2011, 1, 1121–1149 CrossRef CAS.
- P. Bracco, H. Busch, J. von Langermann and U. Hanefeld, Org. Biomol. Chem., 2016, 14, 6375–6389 RSC.
- I. Hajnal, A. Lyskowski, U. Hanefeld, K. Gruber, H. Schwab and K. Steiner, FEBS J., 2013, 280, 5815–5828 CrossRef CAS PubMed.
- R. Wiedner, B. Kothbauer, T. Pavkov-Keller, M. Gruber-Khadjawi, K. Gruber, H. Schwab and K. Steiner, ChemCatChem, 2015, 7, 325–332 CrossRef CAS.
- F. Vertregt, G. Torrelo, S. Trunk, H. Wiltsche, W. R. Hagen, U. Hanefeld and K. Steiner, ACS Catal., 2016, 6, 5081–5085 CrossRef CAS.
- U. Hanefeld, Chem. Soc. Rev., 2013, 42, 6308–6321 RSC.
- M. Hartmann and X. Kostrov, Chem. Soc. Rev., 2013, 42, 6277–6289 RSC.
- K. Zielinska, K. Szymanska, R. Mazurkiewicz and A. Jarzebski, Tetrahedron: Asymmetry, 2017, 28, 146–152 CrossRef CAS.
- P. Bracco, G. Torrelo, S. Noordam, G. de Jong and U. Hanefeld, Catalysts, 2018, 8, 287 CrossRef.
- D. Okrob, M. Paravidino, R. V. A. Orru, W. Wiechert, U. Hanefeld and M. Pohl, Adv. Synth. Catal., 2011, 353, 2399–2408 CrossRef CAS.
- E. Wehtje, P. Adlercreutz and B. Mattiasson, Biotechnol. Bioeng., 1990, 36, 39–46 CrossRef CAS PubMed.
- R. Lindeque and J. Woodley, Catalysts, 2019, 9, 262 CrossRef CAS.
- H. Larsson, P. A. Schjøtt, E. Byströ, K. V. Gernaey and U. Krühne, Ind. Eng. Chem. Res., 2017, 56, 3853–3865 CrossRef CAS.
- H. Mallin, J. Muschiol, E. Byström and U. Bornscheuer, ChemCatChem, 2013, 5, 3529–3532 CrossRef CAS.
- M. P. van der Helm, P. Bracco, H. Busch, K. Szymańska, A. Jarzębski and U. Hanefeld, Catal. Sci. Technol., 2019, 9, 1189–1200 RSC.
- R. Munirathinam, J. Huskens and W. Verboom, Adv. Synth. Catal., 2015, 357, 1093–1123 CrossRef CAS.
- N. N. Rao, S. Lütz, K. Würges and D. Minör, Org. Process Res. Dev., 2009, 13, 607–616 CrossRef CAS.
- K. G. Hugentobler, M. Rasparini, L. A. Thompson, K. E. Jolley, A. J. Blacker and N. J. Turner, Org. Process Res. Dev., 2017, 21, 195–199 CrossRef CAS.
- A. Brahma, B. Musio, U. Ismayilova, N. Nikbin, S. Kamptmann, P. Siegert, G. E. Jeromin, S. V. Ley and M. Pohl, Synlett, 2016, 27, 262–266 CAS.
- L. van den Biggelaar, P. Soumillion and D. P. Debecker, Catalysts, 2017, 7, 54 CrossRef.
- M. Movsisyan, E. I. P. Delbeke, J. K. E. T. Berton, C. Battilocchio, S. V. Ley and C. V. Stevens, Chem. Soc. Rev., 2016, 45, 4892–4928 RSC.
- L. H. Andrade, W. Kroutil and T. F. Jamison, Org. Lett., 2014, 16, 6092–6095 CrossRef CAS PubMed.
- M. Bajić, I. Plazl, R. Stloukal and P. Žnidaršič-Plazl, Process Biochem., 2017, 52, 63–72 CrossRef.
- M. Planchestainer, M. L. Contente, J. Cassidy, F. Molinari, L. Tamborini and F. Paradisi, Green Chem., 2017, 19, 372–375 RSC.
- C. Zor, H. A. Reeve, J. Quinson, L. A. Thompson, T. H. Lonsdale, F. Dillon, N. Grobert and K. A. Vincent, Chem. Commun., 2017, 53, 9839–9841 RSC.
- F. Dall'Oglio, M. L. Contente, P. Conti, F. Molinari, D. Monfredi, A. Pinto, D. Romano, D. Ubiali, L. Tamborini and I. Serra, Catal. Commun., 2017, 93, 29–32 CrossRef.
- V. De Vitis, F. Dall'Oglio, A. Pinto, C. De Micheli, F. Molinari, P. Conti, D. Romano and L. Tamborini, ChemistryOpen, 2017, 6, 668–673 CrossRef CAS PubMed.
- H. Lamble, S. F. Royer, D. W. Hough, M. J. Danson, G. L. Taylor and S. D. Bull, Adv. Synth. Catal., 2007, 349, 817–821 CrossRef CAS.
- B. Grabner, Y. Pokhilchuk and H. Gruber-Woelfler, Catalysts, 2020, 10, 137 CrossRef CAS.
- U. Hanefeld, A. J. J. Straathof and J. J. Heijnen, Biochim. Biophys. Acta, 1999, 1432, 185–193 CrossRef CAS.
- L. van Langen, F. van Rantwijk and R. A. Sheldon, Org. Process Res. Dev., 2003, 7, 828–831 CrossRef CAS.
- U. Hanefeld, L. Gardossi and E. Magner, Chem. Soc. Rev., 2009, 38, 453–468 RSC.
- F. Effenberger, J. Eichhorn and J. Roos, Tetrahedron: Asymmetry, 1995, 6, 271–282 CrossRef CAS.
- D. Costes, E. Wehtje and P. Adlercreutz, Enzyme Microb. Technol., 1999, 25, 384–391 CrossRef CAS.
- A. Basso, L. De Martin, C. Ebert, L. Gardossi and P. Linda, J. Mol. Catal. B: Enzym., 2000, 8, 245–253 CrossRef CAS.
- K. Szymanska, K. Odrozek, A. Zniszczoł, W. Pudło and A. B. Jarzebski, Chem. Eng. J., 2017, 315, 18–24 CrossRef CAS.
- S. E. Charma and B. L. Wong, Enzyme Microb. Technol., 1981, 3, 111–118 CrossRef.
- A. H. M. M. El-Sayed, W. M. Mahmoud and R. W. Coughlin, Biotechnol. Bioeng., 1990, 36, 83–91 CrossRef CAS.
- N. J. Gleason and J. D. Carbeck, Langmuir, 2004, 20, 6374–6381 CrossRef CAS PubMed.
- T. R. Besanger, R. J. Hodgson, J. R. A. Green and J. D. Brennan, Anal. Chim. Acta, 2006, 564, 106–115 CrossRef CAS PubMed.
- Y. Zhu, Q. Chen, L. Shao, Y. Jiacef and X. Zhang, React. Chem. Eng., 2020, 5, 9–32 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/d0cy00604a |
| This journal is © The Royal Society of Chemistry 2020 |