Open Access Article
Open Access ArticleFlow-driven crystal growth of lithium phosphate in microchannels†
Michael
Emmanuel
a,
Dezső
Horváth
 b and
Ágota
Tóth
b and
Ágota
Tóth
 *a
*a
aDepartment of Physical Chemistry and Materials Science, University of Szeged, Rerrich Béla tér 1, Szeged, Hungary. E-mail: atoth@chem.u-szeged.hu; Fax: +36 62 546482; Tel: +36 62 544614
bDepartment of Applied and Environmental Chemistry, University of Szeged, Rerrich Béla tér 1, Szeged, H-6720, Hungary
First published on 19th June 2020
Abstract
Fine tuning of the precipitation process is possible through self-organization in flow-driven systems. The reacting precursors, lithium chloride and sodium phosphate, are injected simultaneously into the microchannel where the precipitation reaction takes place in the presence of concentration gradients. The growth kinetics of individual precipitate particles is studied by monitoring the particle growth along and transverse to the flow. In the former direction, growth is constant in the stationary concentration field, while in the latter a slowing down is observed. The temporal scaling reveals a significant advective contribution in the transverse orientation as well. Polycrystalline aggregates of orthorhombic lithium phosphate are synthesized with size controlled by the flow.
1 Introduction
The development of rechargeable batteries1,2 and the high demand for electric cars in recent years have led to increased popularity of the lithium ion. Lithium is not only useful in the battery technology but also has gained widespread industrial interest. It is an effective desiccant3 for ceramics and glass due to the high hygroscopy of its bromide and chloride salts. Hollow shaped lithium phosphate catalyzes propylene oxide isomerization,4 lithiated esters are high temperature resistant lubricants,5 and various lithium salts are used as medicine for the treatment of mental illnesses.6With all the applications of lithium, the common source depends on its mining from minerals,7 certain soils,8 underground9 or geothermal waters.10,11 These activities are costly and detrimental to the environment; hence recovery from spent lithium ion batteries is a very important step in using a completely environment-friendly source of energy.8,12
Lithium phosphate is the product of lithium ion battery recycling;12,13 hence, understanding its precipitation kinetics is of great importance if recycling is to be made efficient. Flow-driven precipitation where reactants are pumped at a specific flow rate into a chamber filled with the other reacting species is a convenient technique to provide information on how nucleation and crystal growth processes take place.14–17 Furthermore, it also enables the control of crystal size and morphology,18–20 making it feasible to study and analyze crystal formation. For example, the change from the spherulite form of insulin to a packed fiber structure18 or the enhancement of the calcite form of calcium carbonate21 is a result of confinement and a high flow rate. Ultrasound assistance in mixing during tubular crystallization, besides eliminating pipe clogging, helps to control crystal size as well.22 The production of active pharmaceutical ingredients via flow chemistry has led to the syntheses of many promising compounds.23 Metal ions in the form of sparingly soluble salts can also be spatially separated from their homogeneous solution mixture by the appropriate choice of precipitation.24 Even the type of fluid flow significantly alters the product distribution as polymorphic crystallization of L-histidine can be promoted by periodic Taylor vortex flow.25
Microfluidics serves as a special tool to study crystallization.26,27 Small to single crystals of proteins have been produced efficiently in droplet based devices28–30 where nucleation and growth can be decoupled by varying the flow rate and the reactant concentrations.31,32 When reactants are flowed parallel in microchannels, one-dimensional inorganic membranes may develop,33–35 offering the ease of monitoring the tubular growth of a confined chemical garden. The co-flow design also allows the effective screening of crystal polymorphs,36 essential for creating the desired crystalline material. In order to understand pathological microcalcification, microchannels with similar dimensions to the collecting duct in human kidneys have been utilized as biomimetic models for calcium oxalate formation.37 The confinement due to the microfluidic conditions slows down the conversion of the unstable amorphous calcium carbonate into the crystalline form as opposed to the well-stirred reaction where the stable crystalline form appears within seconds to minutes of mixing.38–40
Building upon the versatile applicability of microfluidics, we first study experimentally the precipitation of lithium phosphate in a microchannel and study the effect of flow on the growth of the precipitate particles. The results are then corroborated by simple calculations.
2 Experimental section
2.1 Experimental setup
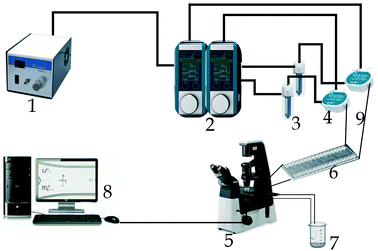
A pressure generator (FLPG plus, Fluigent) with manual pressure regulators and pressure sensors produced high quality dry air up to a maximum pressure of 2.3 bar. The pressure generator was coupled to a flow controller (Flow EZTM) in a dual lineup series enabling the control of the pressure to minimize the fluctuation in the flow. A flow sensor (Fluigent) was connected to each of the controller units to allow flow rate programming and increased reproducibility.Two separate reservoirs (Fluiwell 1C-50) – one for lithium chloride and one for sodium phosphate – were used with PTFE tubes (ID = 0.5 mm) linked to the pressure controller as shown in Fig. 1. The tubes passing through the flow sensors were also connected to an H-shaped and irreversibly sealed channel (Microfluidic Chipshop) serving as a microreactor made from poly(methyl methacrylate) with channel dimensions of w = 300 μm middle width, 150 μm inlet/outlet width, 75 μm depth, and 13.5 mm length. The microreactor was placed on an inverted research microscope (Nikon Eclipse Ts2R) coupled with a high-speed camera (Nikon LV-TV) to monitor the progressive growth of the particles in the microreactor and images were saved at time intervals of 30 s to a computer system.
2.2 Formation of lithium phosphate
Equal volumes from analytical grade chemicals of LiCl (VWR) and Na3PO4 (Sigma-Aldrich), in a stoichiometric ratio of R = [Li+]/[PO43−] = 3 with [Na3PO4] = 0.1 M, were prepared in deionized water, filtered through a 25 mm syringe filter cap (cellulose acetate membrane with 0.2 μm pore size, VWR) and poured into the reservoirs. The flow rate was increased from 0.5 μL min−1 per channel up to 2 μL min−1 per channel in steps of 0.25 μL min−1 per channel, for each separate experiment yielding the total flow rate (qV) in the range of 1 μL min−1 ≤ qV ≤ 4 μL min−1. The experiments were run for a maximum of 45 min to avoid clogging of the reactor. The precipitate product was collected through the two outlets and kept for analyses. After stopping the flow, the entire system underwent a cleaning procedure: first, by pumping filtered deionized water into the channel at higher pressure, the precipitate formed in the microreactor was collected. Then, 1.0 M hydrochloric acid was injected to dissolve any precipitate remaining in the microreactor, followed by purging the channels with deionized water again and blowing with dry air for about 2–4 minutes to dry the channels. The entire procedure was repeated with a concentration ratio of R = 6 corresponding to a non-stoichiometric composition of reactants with LiCl in excess.To characterize the particles, separate reference experiments were also performed in a well-stirred reactor of a 100 mL-volume beaker where equal volumes (25 mL) of the chemicals were mixed under vigorous stirring for 25 min. The precipitate was filtered and washed several times with deionized water then dried and used for characterization.
2.3 Characterization
The density of the reactants was measured with a precision of 10−4 g cm−3 using a density meter (Anton Paar, DMA 500).Particle position and size were determined from the stored images by a specially designed macro of ImageJ where the edge of the precipitate was identified as points of inflection in the gray scale intensity collected along and transverse to the flow.
The microstructure of the particles was determined from the images using a scanning electron microscope (Hitachi S4700). The product was dried and then placed on adhesive carbon paper, and sputtered with gold in a vacuum for 1 minute to increase the conductivity of the particle surface. The vacuum accelerating voltage in the scanning electron microscope was 20 kV, at a working distance of 12 mm. The X-ray diffractograms of the powdered Li3PO4 samples were recorded on a Philips X-ray diffractometer with copper Kα = 0.1542 nm as the radiation source at ambient temperature in the 2–60° (2θ) range applying a 0.02° step size. The percentage crystallinity was calculated as the ratio of the area of crystalline peaks to the total area of all peaks. The crystallite sizes were determined using the Scherrer equation
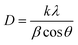 | (1) |
3 Results and discussion
By the use of a fully automated microfluidic setup, lithium chloride and sodium phosphate solutions are pumped simultaneously with the same flow rate into the microreactor. The two reactants come into contact at the middle of the channel where nucleation starts. The precipitation takes place in the middle along the flow giving rise to a so-called contact line. The nucleation itself is stochastic in time, implying that the time of appearance of the first precipitate particle in the channel is not a reproducible entity.Injecting the reactants at a stoichiometric ratio of R = 3, we have observed that the first particles appear at 8–10 min after starting the flow along the contact line. There are many nucleation sites yielding small-sized precipitate particles and hence hindering the growth of the individual particles (see Fig. 2(a)).
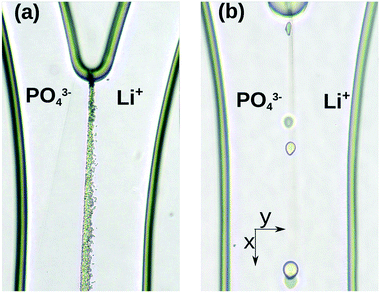 | ||
| Fig. 2 Precipitate particle growth in the microchannel with w = 300 μm, qV = 3.0 μL min−1 and chemical compositions, (a) R = 3 and (b) R = 6, at t = 45 min. | ||
Upon increasing R to 6, even after 15–20 min, fewer nuclei appear initially as orthorhombic crystals then grow into spherical aggregates which evolve separately as shown in Fig. 2(b). The well-spaced particles along the contact line are surrounded with more cations in the system than anions because of the lithium ion excess.
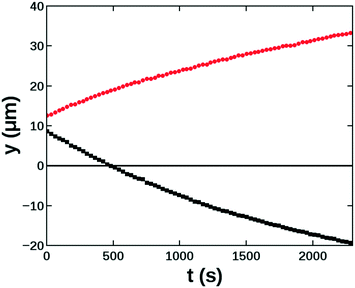
To characterize the growth of individual particles, a minimum of three parallel experiments have been performed with a stoichiometric ratio of R = 6 for every flow rate. For the selected particles, their position is monitored as a function of time. A typical growth profile is illustrated in Fig. 3, where the temporal evolution of the edge of a precipitate particle is presented in the transverse direction with y = 0 position corresponding to the center vertical plane of the channel and y > 0 to the side of the lithium source. The distance between the two positions yields the diameter of the particle transverse to the flow, i.e., along the y-axis, which is also measured in order to construct the growth profiles. The crystals form mainly in the vicinity of the contact line but on the side of the lithium chloride inlet which is due to its higher concentration gradient.
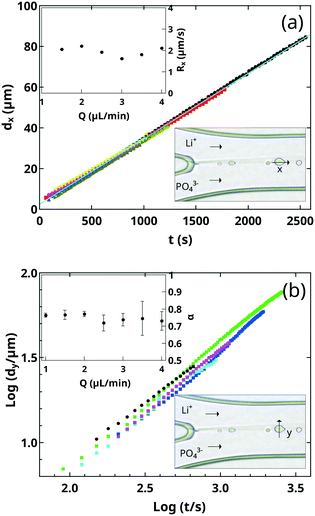
A similar technique is applied to determine the particle growth along the flow allowing us to observe the effect of flow in both directions. First at a given flow rate, several particles are chosen and their growth characteristics are determined. One example case is summarized in Fig. 4, where the growth profiles along the two directions are depicted for several particles (see the ESI† for more data). For a given injection rate, the particle diameters along the flow increase linearly with time (cf.Fig. 4(a)) for the individual particles independent of the time of appearance and their position as long as the particles are sufficiently separated from each other. From the slope, therefore, we can obtain a constant growth rate of 2.0 ± 0.2 μm min−1 along the flow independent of the particle position and for injection rates in the range of 1.5 μL min−1 ≤ qV ≤ 4.0 μL min−1. This observed behavior implies that the supersaturation cannot change significantly along the flow (x-axis). This can be rationalized by considering the mean linear flow rate, which falls in the range of 1.1–3.0 mm s−1 leading to a residence time of less than one second in the view area for the injected fluid. The precipitation reaction is the rate-determining factor yielding a reaction-controlled growth41 along the flow as the reactants are continuously brought to the contact line by the flow.
The precipitation process in the transverse direction, however, slows down. The size of the precipitate particles along the y-axis scales with time according to the power law dy = ktα as shown in Fig. 4(b), where the exponent is invariant to the injection rate under our experimental conditions. This is the result of the decrease in supersaturation as we move away from the contact line, which lowers the driving force of precipitation. The fast supply of reactants by the flow affects the entire width of the microchannel; therefore the determined exponent is greater than 0.5, which is a characteristic of reactions between initially segregated reactants with only a diffusional support.42
3.1 Modeling of the concentration field in the microchannel
The flow in the reactor is laminar as indicated by the Reynolds number in the range of 0.09–0.36 as calculated according to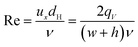 | (2) |
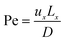 | (3) |
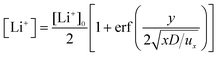 | (4) |
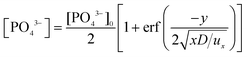 | (5) |
| 3Li+(aq) + PO43−(aq) ⇌ Li3PO4(s) | (6) |
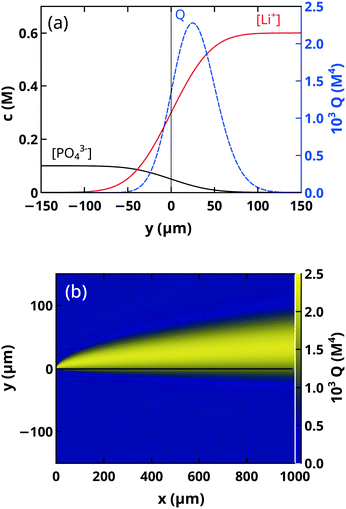
By considering the spatial distribution of the supersaturation, we can rationalize the growth rate of precipitate particles. In the vicinity of the contact line, the vertical xz-plane is characterized by an almost constant supersaturation; hence the growth rate of isolated particles (Rx) along the flow remains constant. Transverse growth on the other hand encounters a significant lowering of supersaturation; therefore the growth rate decreases with time. The scaling exponent is greater than 0.5, which indicates that the fresh reactants are transported by advective transport besides diffusion to the surface of the precipitate particles.
For more accurate simulation to describe the spatial distribution of the precipitate particles at the time of nucleation, the density difference between the injected liquids has to be considered as well. Although under the experimental conditions the densities of the reactant solutions, 1.0153 g cm−3 for 0.1 M Na3PO4 and 1.0111 g cm−3 for 0.6 M LiCl, are comparable, weak buoyant forces can arise in the yz-plane transverse to the main flow. This additional fluid motion will further increase the asymmetry in the spatial distribution of precipitate particles sedimented in the microfluidic reactor.
3.2 Microstructure
The morphology visualized by the SEM images of the particles from the well-stirred system and the microchannels appears similar (see Fig. 6); spherical particles with a spherulite-like structure are formed which consist of small rod-like crystallites of about 1–2 μm in size (see the ESI† for further images.) Besides the crystallites, amorphous particles are also observable. | ||
| Fig. 6 SEM images of precipitate particles (a) from a well-stirred system with (b) an enlargement and (c) from the microchannel with (d) an enlargement for R = 6. | ||
From the diffractograms in Fig. 7, we have identified that the crystals are the orthorhombic, low form of lithium phosphate.43 The X-ray diffraction data also indicate that the lithium phosphates obtained from both the microchannel and the well-stirred reference system belong to the same space group of Pmn21 (orthorhombic)43 which corresponds to β-Li3PO4 prepared mostly from a wet chemical reaction,44 with corner-sharing tetrahedra pointing towards the same direction along the main c axis.45,46
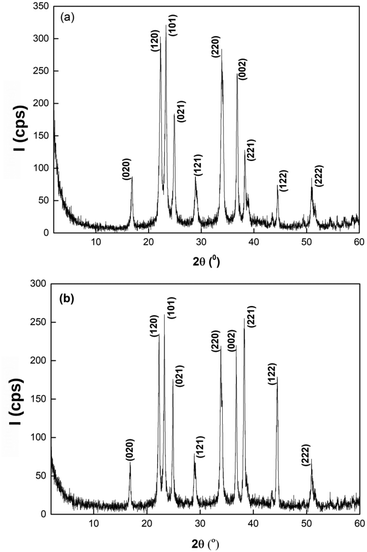 | ||
| Fig. 7 X-ray diffractograms of samples (a) from the well-stirred experiment and (b) from the microchannel. | ||
The crystallinity of the samples is ∼67% from both the reference and the microfluidic system within the experimental error. The average size of the crystallites comprising the polycrystalline aggregates calculated from the Scherrer equation is 18.25 nm and 23.33 nm in the well-stirred and in the microfluidic system, respectively, indicating that microfluidics offers a simple possibility to increase the crystallite size in a controlled manner.
4 Conclusion
By injecting LiCl and Na3PO4 solutions with equal flow rates into a microchannel, we have studied the crystal growth of lithium phosphate in the presence of concentration gradients. Nucleation occurs along the thin zone with maximum supersaturation near the contact line, formed in the middle of the microfluidic channel. For individual particles, the growth rate parallel to the flow is constant and independent of the fluid velocity, which is a result of the time independent concentration profiles maintained by the fast injection. It leads to a nearly constant supersaturation along the main flow. In the transverse direction on the other hand, the growth of particles slows down because of the sudden drop in the supersaturation between the initially segregated reactants. As the scaling exponent indicates, advection drives the precipitation process besides diffusion. Particle morphology analysis confirms the production of orthorhombic lithium phosphate with a size controlled by the flow.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We acknowledge the help of Dr. Gábor Schuszter with SEM and Ágota Imre-Deák with XRD measurements. This work has also been supported by the National Research, Development and Innovation Office (NN 125746), M-ERA.Net MaSNEC and GINOP-2.3.2-15-2016-00013 projects and the University of Szeged Open Access Fund (4816).References
- J. Goodenough and Y. Kim, Chem. Mater., 2010, 22, 587–603 CrossRef CAS.
- M. S. Whittingham, Chem. Rev., 2004, 104, 4271–4302 CrossRef CAS.
- Z. Wang, D. Orejon, K. Sefiane and Y. Takata, Phys. Chem. Chem. Phys., 2019, 21, 1046–1058 RSC.
- H. Li, W. Jiang, W. Ma, H. Qu and Q. Zhong, Chem. Eng. Commun., 2016, 203, 339–344 CrossRef CAS.
- P. K. Alaboina, J.-S. Cho, M.-J. Uddin and S.-J. Cho, Electrochim. Acta, 2017, 258, 623–630 CrossRef CAS.
- R. Oruch, M. A. Elderbi, H. A. Khattab, I. F. Pryme and A. Lund, Eur. J. Pharmacol., 2014, 740, 464–473 CrossRef CAS.
- H. Aral and A. Vecchio-Sadus, Ecotoxicol. Environ. Saf., 2008, 70, 349–356 CrossRef CAS PubMed.
- L. Kavanagh, J. Keohane, G. Garcia-Cabellos, A. Lloyd and J. Cleary, Resources, 2018, 7, 57 CrossRef.
- B. Shahzad, M. Tanveer, W. Hassan, A. N. Shah, S. A. Anjum, S. A. Cheema and I. Ali, Plant Physiol. Biochem., 2016, 107, 104–115 CrossRef CAS PubMed.
- J. A. Epstein, E. M. Feist and J. Zmora, Hydrometallurgy, 1981, 6, 269–275 CrossRef CAS.
- C. Fouillac and G. Michard, Geothermics, 1981, 10, 55–70 CrossRef.
- H. Li, S. Xing, Y. Liu, F. Li, H. Guo and G. Kuang, ACS Sustainable Chem. Eng., 2017, 5, 8017–8024 CrossRef CAS.
- T. Ryu, J. Shin, J. Ryu, I. Park, H. Hong, B.-G. Kim and K.-S. Chung, Mater. Trans., 2013, 54, 1029–1030 CrossRef CAS.
- J. H. E. Cartwright, J. M. García-Ruiz, M. L. Novella and F. Otálora, J. Colloid Interface Sci., 2002, 256, 351–359 CrossRef CAS.
- A. Yashina, F. Meldrum and A. deMello, Biomicrofluidics, 2012, 6, 022001 CrossRef PubMed.
- P. Pusztai, E. Tóth-Szeles, D. Horváth, A. Tóth, A. Kukovecz and Z. Kónya, CrystEngComm, 2015, 17, 8477–8485 RSC.
- R.-B. Zhou, H.-L. Cao, C.-Y. Zhang and D.-C. Yin, CrystEngComm, 2017, 19, 1143–1155 RSC.
- V. Foderá, S. Pagliara, O. Otto, U. F. Keyser and A. M. Donald, J. Phys. Chem. Lett., 2012, 3, 2803–2807 CrossRef.
- B. Bohner, G. Schuszter, O. Berkesi, D. Horváth and Á. Tóth, Chem. Commun., 2014, 50, 4289–4291 RSC.
- E. Tóth-Szeles, G. Schuszter, A. Tóth, Z. Kónya and D. Horváth, CrystEngComm, 2016, 18, 2057–2064 RSC.
- B. Bohner, G. Schuszter, D. Horváth and Á. Tóth, Chem. Phys. Lett., 2015, 631–632, 114–117 CrossRef CAS.
- N. Ezeanowi, H. Pajari, A. Laitinen and T. Koiranen, Cryst. Growth Des., 2020, 20, 1458–1466 CrossRef CAS.
- R. Porta, M. Benaglia and A. Puglisi, Org. Process Res. Dev., 2016, 20, 2–25 CrossRef CAS.
- E. Tóth-Szeles, B. Bohner, A. Tóth and D. Horváth, Cryst. Growth Des., 2017, 17, 5000–5005 CrossRef.
- S. Park and W.-S. Kim, Cryst. Growth Des., 2018, 18, 710–722 CrossRef CAS.
- J. Leng and J.-B. Salmon, Lab Chip, 2009, 9, 24–34 RSC.
- M. Maeki, S. Yamazaki, A. S. Pawate, A. Ishida, H. Tani, K. Yamashita, M. Sugishima, K. Watanabe, M. Tokeshi, P. A. Kenis and M. Miyazaki, CrystEngComm, 2016, 18, 7722–7727 RSC.
- M. Ildefonso, E. Revalor, P. Punniam, J. B. Salmon, N. Candoni and S. Veesler, J. Cryst. Growth, 2012, 342, 9–12 CrossRef CAS.
- P. Laval, A. Crombez and J.-B. Salmon, Langmuir, 2009, 25, 1836–1841 CrossRef CAS PubMed.
- M. Ildefonso, N. Candoni and S. Veesler, Cryst. Growth Des., 2013, 13, 2107–2110 CrossRef CAS.
- J.-u. Shim, G. Cristobal, D. R. Link, T. Thorsen, Y. Jia, K. Piattelli and S. Fraden, J. Am. Chem. Soc., 2007, 129, 8825–8835 CrossRef CAS PubMed.
- J. Cavanaugh, M. Whittaker and D. Joester, Chem. Sci., 2019, 10, 5039–5043 RSC.
- B. C. Batista and O. Steinbock, J. Phys. Chem. C, 2015, 119, 27045–27052 CrossRef CAS.
- Q. Wang, M. R. Bentley and O. Steinbock, J. Phys. Chem. C, 2017, 121, 14120–14127 CrossRef CAS.
- Q. Wang and O. Steinbock, ChemCatChem, 2020, 12, 63–74 CrossRef CAS.
- P. Coliaie, M. S. Kelkar, N. K. Nere and M. R. Singh, Lab Chip, 2019, 19, 2373–2382 RSC.
- G. Laffite, C. Leroy, C. Bonhomme, L. Bonhomme-Coury, E. Letavernier, M. Daudon, V. Frochot, J. P. Haymann, S. Rouziére, I. T. Lucas, D. Bazin, F. Babonneau and A. Abou-Hassan, Lab Chip, 2016, 16, 1157–1160 RSC.
- Y. Zeng, J. Cao, Z. Wang, J. Guo and J. Lu, Cryst. Growth Des., 2018, 18, 1710–1721 CrossRef CAS.
- Y. Zeng, J. Cao, Z. Wang, J. Guo, Q. Zhou and J. Lu, Cryst. Growth Des., 2018, 18, 6538–6546 CrossRef CAS.
- A. Perazzo, L. Sicignano, G. Tomaiuolo, R. Marotta, R. Andreozzi and S. Guido, Chem. Eng. Sci., 2019, 207, 581–587 CrossRef CAS.
- R. D. Sosa, X. Geng, M. A. Reynolds, J. D. Rimer and J. C. Conrad, Lab Chip, 2019, 19, 1534–1544 RSC.
- L. Gálfi and Z. Rácz, Phys. Rev. A: At., Mol., Opt. Phys., 1988, 38, 3151–3154 CrossRef PubMed.
- E. S. Howard, C. M. Marlene, E. H. Evans and L. Ulmer, Standard X-ray Diffraction Powder Patterns, National Bureau of Standards, U.S. Department of Commerce, 25th edn, 1965 Search PubMed.
- N. I. P. Ayu, E. Kartini, L. D. Prayogi, M. Faisal and A. Supardi, Ionics, 2016, 22, 1051–1057 CrossRef CAS.
- N. Yabuuchi, Y. Yamakawa, K. Yoshii and S. Komaba, Dalton Trans., 2011, 40, 1846–1848 RSC.
- M. E. Arroyo-deDompablo, R. Dominko, J. M. Gallardo-Amores, L. Dupont, G. Mali, H. Ehrenberg, J. Jamnik and E. Morán, Chem. Mater., 2008, 20, 5574–5584 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/d0ce00540a |
| This journal is © The Royal Society of Chemistry 2020 |