Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Towards organic neuromorphic devices for adaptive sensing and novel computing paradigms in bioelectronics
Eveline R. W.
van Doremaele
a,
Paschalis
Gkoupidenis
*b and
Yoeri
van de Burgt
 *a
*a
aMicrosystems, Institute for Complex Molecular Systems, Eindhoven University of Technology, 5612AZ Eindhoven, The Netherlands. E-mail: y.b.v.d.burgt@tue.nl
bDepartment of Molecular Electronics, Max Planck Institute for Polymer Research, 55128 Mainz, Germany. E-mail: gkoupidenis@mpip-mainz.mpg.de
First published on 30th July 2019
Abstract
Bioelectronics has made enormous progress towards the development of concepts, materials and devices that are capable of sensing, monitoring and controlling a biological environment, by incorporating concepts such as local drug delivery and electrical, chemical or mechanical stimulation. Nevertheless, fully autonomous bioelectronic applications demand not only the acquisition of biological signals, but also local low power data processing, storage and the extraction of specific features of merit. Here, we present an overview of the latest studies on organic neuromorphic and sensing devices. We also speculate on the need for smart and adaptive sensing and highlight the potential of these concepts to enhance the interaction efficiency between electronics and biological substances.
Traditional computing systems are unable to capture the capability of the brain in real world information processing as evidenced by the anticipated end to Moore's law. Organic (electrochemical) materials have recently emerged as building blocks of neural processing1 and possess basic forms of neuroplasticity that are able to emulate brain-like functionality at the device level2 (see Fig. 1). Compared to inorganic materials, the excellent characteristics of organic electronic materials, such as low energy operation and tunability, allows these materials to be used as a first step towards efficient neuromorphic computing systems.3 In fact, devices based on organic-mixed conductors demonstrate good electrical stability (105 sec retention time)4 as well as excellent endurance (>109 switching cycles)5 and allow for direct control of material characteristics. A recently demonstrated ionic floating gate concept based on organic devices shows efficient parallel updating of a small array.5 Small-scale perceptron-based artificial neural networks have also been demonstrated.6 Another concept was recently presented based on an evolvable organic electrochemical transistor, where long-term changes rely on the electro-polymerization of a conductive polymer between the device terminals.7 Apart from organic mixed conductors, ferroelectric organic neuromorphic transistors have also been demonstrated recently.8 These examples showcase the initial efforts towards building blocks for the realization of small organic-based neural networks and applications in local computation and feature extraction.
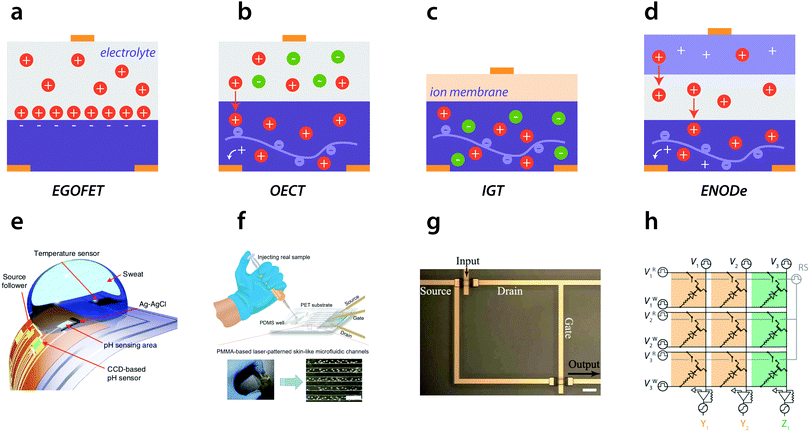 | ||
| Fig. 1 Recent examples of organic transistors in neuromorphic and sensing applications. (a) Electrolyte-gate organic field effect transistor. A gate pulse forms a double layer between the electrolyte and organic semiconductor, which turns the channel on. (b) Organic electrochemical transistor (OECT). The electrolyte and ions penetrate the complete volume of the (semi)-conducting polymer, thereby switching the channel on or off. (c) Internal ion-gated organic electrochemical transistor (IGT). Here, contained mobile ions within the conducting polymer channel are responsible for the conductance switching, which enables both volumetric capacitance and short ionic transit time. (d) Electrochemical non-volatile organic device (ENODe). This device combines an OECT with a polymeric gate material, to allow for non-volatile switching of the channel.1 (e) pH sensor based on a charge-coupled device.9 (f) Wearable cortisol sensor based on an OECT channel.10 (g) Example of a simple circuit based on IGT devices.11 (h) Simple neuromorphic array based on ENODe devices, that allows for parallel updating by employing memristors as access devices.5 | ||
Due to their ability to operate in global or shared electrolytes, organic neuromorphic devices have shown multi-terminal operations,12 global connectivity control13 as well as phase-dependent synchrony through global electrical oscillations.14 The introduction of global phenomena in organic neuromorphic devices demonstrates the effectiveness in mimicking brain-like functionality in a more bio-plausible way. Indeed, in the brain, apart from local neuronal development through synaptic potentiation and depression, the global electrochemical environment of biological neural networks is essential for the network functionality. Moreover, neuromorphic sensing implies a common electrochemical environment in which the neuromorphic circuitry might be immersed. Therefore, besides local device training, global electrochemical regulation of the circuitry can offer additional degrees of freedom that are necessary for feature extraction.
However, a challenge remains to develop organic devices and architectures capable of reproducing neuro-inspired information processing functions with the potential to actually operate at the interface with biology. Such architectures will have significant impact in applications that span from brain–computer-interfaces and neuroscience to bioinformatics and the definition of novel computational paradigms at the interface with biology. Initial reports in this direction include time-dependent classification, by leveraging the inherent global phenomena of organic electrochemical transistors in the framework of reservoir computing.15 However, the field is at an early stage and in need for novel materials, devices and circuit concepts. Nevertheless, the rapid rise of sensing platforms based on organic electrochemical transistors,16 as well as novel concepts as building blocks for bioelectronic circuits and devices,11 greatly enhance the validity of this field for smart or even neuromorphic bio-signal processing.
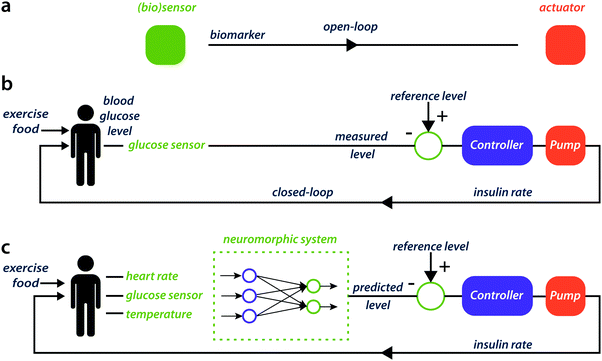
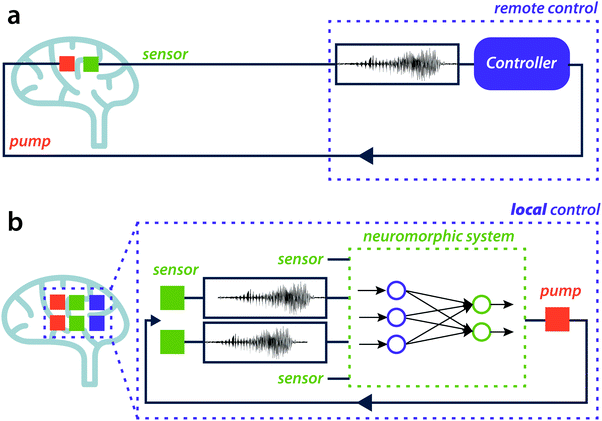
To effectively enhance the operational ability of bioelectronics beyond sensing or stimulation, a bi-directional coupling is essential. On the one hand, a biological process transfers a signal to an organic electronic device where for instance an enzymatic reaction, changes the current flow through the polymer transistor, see Fig. 2a. In the opposite direction, an organic electronic device can trigger a biochemical reaction or biological process, for instance by the application of electrical stimuli on a conducting polymer electrode, and the activity of biological neural networks can be modulated.17 This modulation can also be achieved chemically by local drug delivery.18 By merging both directions of coupling, a closed-loop system can be implemented. Closed-loop systems can be found in for instance glucose sensing for diabetes patients (see Fig. 2b), where temporal evolution of glucose release is a multi-variable function that also depends on the activity history of the patient. Closed-loop systems can also be found in less conventional but long-term applications, such as in control and training of neurorobotic systems using neuronal cultures on multi-electrode arrays.19 In the future, to further advance the sustainability of bioelectronic platforms in a biological environment, this bi-directional coupling should be smart, dynamic and adaptable, and rely on multiple input variables to improve the accuracy of the neuromorphic system, similar to the working principles of neural networks (see Fig. 2c).
Traditional (organic) field effect transistors have been extensively implemented as bio-sensors,20 and can be operated in an liquid electrolyte, see Fig. 1a. Yet, in recent years, organic electrochemical transistors (OECTs), have been increasingly reported to transduce biological (ion- or chemical-based) signals into electronic signals.16 To a large extent, this trend results from the additional degrees of freedom that these electrochemical devices offer, compared with passive (capacitive) sensors. An OECT consists of a semiconductor polymer channel, a common example is poly(ethylenedioxythiophene):poly(styrene-sulfonate) (PEDOT:PSS), that can be gated through a solid or aqueous electrolyte, see Fig. 1b. By applying a gate potential, anions or cations (depending on the polarity of the gate) in the electrolyte are introduced into the organic semiconductor channel and dope/de-dope the entire volume, changing its conductivity.21 Typically, the electrolyte of OECT-based biosensors is aqueous as they target biomarkers/ionic species in a biological solution (e.g. sweat or in-vivo). Compared to standard potentiometric sensors, OECTs have the advantage of a significant signal amplification with the potential to yield biosensors with a low detection limit and high sensitivity, even in an interfering environment.22 The high sensitivity is based on the high transconductance, which results from the fact that the whole volume of the organic materials partakes in the channel switching (see Fig. 1b).16 Smart designs of the electronic circuit, such as the Wheatstone bridge layout, can even further improve the sensor sensitivity. This layout also yields an inherent background subtraction, eliminating interference arising from other factors.23,24 Using these concepts, a wide variety of sensors have been developed, ranging from in vitro to in vivo or wearables. The latter is useful for non-invasive monitoring of biomarkers such as long-term and continuous glucose monitoring25 or pH sensing.9 Towards this direction, cheap and disposable devices were recently shown, such as a fully inkjet-printed glucose sensor.26 Apart from sensing in humans, glucose sensing with organic devices is also used for real-time monitoring of biological processes in plants.27
Although originally OECTs transduce (biological) ion-based signals into electrical signals, targeting of specific molecules can also be done using antibodies or enzymes as specific recognition elements.28,29 These biosensors are label-free and require the immobilization of the bioreceptor at one of the relevant device interfaces. Since these biosensors are often vulnerable to interference from other ions present in the sample solutions, the use of a molecularly imprinted polymer can be beneficial. By positioning this molecular selective membrane in between the gate electrode and the channel, the ion flow is manipulated (blocked in case of high target concentration) resulting in a change in the difference of the source–drain current (ΔISD) which depends on the concentration of the target.10,30 This method also allows for targets without electronic charge to be detected. An extended gate FET concept can be used which has the advantage of separating the wet and dry environment. The sensing part extends off chip and can be immersed into the solution without the transducer part.20 A similar concept was also reported for an organic electrochemical transistor-based sensor system employing a floating gate31 and was used to detect gluten.32 Another concept that has no direct contact between the active material and the electrolyte or analyte, is the internal ion-gated transistor (IGT), see Fig. 1c. Compared to a regular OECT, where the speed is limited by the travel time of ions between the electrolyte and the conducting polymer channel, this device has a significantly faster time response.11 The ionic transit time was shortened by using contained mobile ions within the conducting polymer channel. In this way, the time that these ions have to travel to participate in the (de)doping process is reduced. The authors further demonstrate that these devices are of great potential for logic circuits, as the preloaded ions do not require an external source of ions, see Fig. 1g. The above mentioned building blocks and other simple circuits (for example differential measurements with Wheatstone bridges23), are used as an essential first step towards smart processing.
Recent examples for (biological) stimulation or delivery of biological components, were achieved by using an organic electronic ion pump (OEIP). An OEIP is an electrophoretic delivery device that transduces electronic signals into ionic fluxes. The polymer delivery channel is a material with high ionic but low electronic conductivity that allows for the selective transport of anions or cations. An advantage of this mechanism is that the electrophoretic transport does not require liquid flow and only the charged particles are delivered to the target region. Not only atomic ions can be transported but also larger molecules as demonstrated by the delivery of an aromatic compound to a living plant model by using a dendrolyte-based OEIP.36 Charged compounds can also be released independently from several delivery points within tens of milliseconds by adding control electrodes under each delivery point, resulting in vertical potential gradients through the device's thin films. A bipolar membrane allows for the control of the delivery as the resistance can be increased or decreased under reversed or forward bias, respectively.37 The main limitation of the OEIP for in vivo use is that often high voltages are required for its operation. To overcome this limitation, a microfluidic ion pump was designed to bring the drug molecules close to the delivery point, where they can be pumped outward to the target (by electrophoresis), offering low-voltage operation.18 Combining this microfluidic ion pump with a conformable electrocorticography device with recording cites embedded next to the drug delivery outlets, makes it possible to deliver drugs and record local neural activity simultaneously on the surface of the brain.33,38
The next step includes not only simultaneous sensing and actuation or stimulation, but also local processing of data. This concept has been demonstrated in recent biomimetic application examples, where artificial organs that sense relevant signals such as light, pressure or sound, are used as the input.39 An organic artificial afferent nerve was developed that can detect the movement of objects34 (see Fig. 3b). Another sensory device was reported to demonstrate tactile learning.40 Using light as the input to a photonic synapse can be used for artificial retinas41 as well as an input to organic optoelectronic sensorimotor synapses.42 This latter uses an organic optoelectronic synapse and a neuromuscular system based on a stretchable organic nanowire synaptic transistor (s-ONWST). The voltage pulses of a self-powered photodetector triggered by optical signals drive the s-ONWST. The resulting informative synaptic outputs can be used not only for optical wireless communication of human-machine interfaces but also for light-interactive actuation of an artificial muscle actuator mimicking the contraction of a biological muscle fiber.42
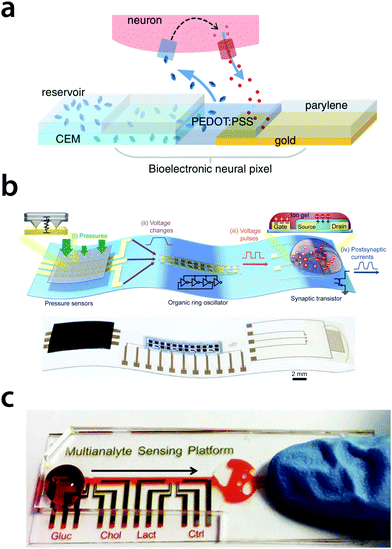 | ||
| Fig. 3 Examples of transducing, sensing and actuation microsystems. (a) Bioelectronic neural pixel with chemical stimulation and electrical sensing at the same site.33 (b) Bioinspired flexible organic artificial afferent nerve.34 (c) Organic-transistor array-based multi-analyte sensing platform for saliva testing.35 | ||
Ideally, complete systems and devices will include all three components locally: sensing, processing and actuation, and operate in a closed-loop fashion. For that, the devices need to be stable in an aqueous solution and operate with low-power consumption. Even with current technologies, control parameters can already be tuned for optimization, but novel systems can also be envisioned where a neuromorphic core is responsible for the prediction and classification of relevant behavior and signals. These autonomous applications demand operation in a real biological environment, where organic materials have an advantage, but also require long term operation. In fact, many applications for the classification of signals and relevant behaviour will require analysis in real time. These systems should be able to not only classify anomalies in the time domain but also predict long-living (i.e. low frequency) patterns.
Potentially the inter-correlation of multi-variables that influence the biological environment can be adopted to increase the effectiveness. An example of a multi-analyte sensing platform was demonstrated on saliva,35 as shown in Fig. 3c. By combining such multi-sensor information with local neuromorphic processing as well as stimulation, a dynamic smart and adaptable device can be envisioned. By measuring and processing information locally with an organic neuromorphic sensing platform relevant signals and information can be extracted. The platform can thus be trained for classification applications, and as such does not require extremely high-bandwidth signals (to be analyzed remotely), nor risks losing the essence of the information at the source (when high-bandwidth is not possible).
A hypothetical application is show in Fig. 4, based on the bioelectronic neural pixel,33 where a device can both sense neuronal activity as well as release a drug (to reduce epileptic seizures). However, in this previously demonstrated example the decision to release the drug is done manually and at a remote location. Ideally, this should be done locally and in situ. By combining multi-modal sensors at various locations, the neuromorphic core can be trained to improve the analysis and classification of the signals and behavior of the biological environment autonomously in a closed loop fashion. In this particular case, the platform could start the drug release to counteract the seizures.
In summary, we have highlighted the recent trends in organic neuromorphic devices and organic bioelectronic sensing platforms. We see opportunities in merging these two fields to further close the loop between sensing and stimulation/actuation for smart bioelectronics applications. Materials and devices that exhibit memory and have the potential to operate at the interface with biology can pave the way for novel data classification paradigms with bio-inspired features in information processing. These materials might thus offer promising solutions for the manipulation and the processing of biological signals, spanning from novel brain–computer-interfaces and adaptive prosthetics to bioinformatics and the definition of novel computational paradigms at the interface with biology. For such applications, we envision smart multi-modal sensor platforms and local neuromorphic processing. Sensing and processing will be tightly bound in a closed loop manner in order to reduce local data volume and thus allow fully autonomous applications in the future.
Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
The authors gratefully acknowledge funding from the European Union's Horizon 2020 Research and Innovation Programme, grant agreement no. 802615. Open Access funding provided by the Max Planck Society.References
- Y. van de Burgt, et al., A non-volatile organic electrochemical device as a low-voltage artificial synapse for neuromorphic computing, Nat. Mater., 2017, 16, 414–418 CrossRef CAS PubMed.
- P. Gkoupidenis, N. Schaefer, B. Garlan and G. G. Malliaras, Neuromorphic Functions in PEDOT:PSS Organic Electrochemical Transistors, Adv. Mater., 2015, 27, 7176–7180 CrossRef CAS PubMed.
- Y. Burgt, A. van de, Melianas, S. T. Keene, G. Malliaras and A. Salleo, Organic electronics for neuromorphic computing, Nat. Electron., 2018, 1, 386–397 CrossRef.
- S. T. Keene, A. Melianas, Y. van de Burgt and A. Salleo, Mechanisms for Enhanced State Retention and Stability in Redox-Gated Organic Neuromorphic Devices, Adv. Electron. Mater., 2018, 1800686 Search PubMed.
- E. J. Fuller, et al., Parallel programming of an ionic floating-gate memory array for scalable neuromorphic computing, Science, 2019, 364, 570–574 CrossRef CAS PubMed.
- S. Battistoni, V. Erokhin and S. Iannotta, Organic memristive devices for perception applications, J. Phys. D: Appl. Phys., 2018, 51, 284002 CrossRef.
- J. Y. Gerasimov, et al., An Evolvable Organic Electrochemical Transistor for Neuromorphic Applications. Advanced, Science, 2019, 6, 1801339 Search PubMed.
- S. Jang, et al., Ultrathin Conformable Organic Artificial Synapse for Wearable Intelligent Device Applications, ACS Appl. Mater. Interfaces, 2019, 11, 1071–1080 CrossRef CAS PubMed.
- S. Nakata, et al., A wearable pH sensor with high sensitivity based on a flexible charge-coupled device, Nat. Electron., 2018, 1, 596 CrossRef.
- O. Parlak, S. T. Keene, A. Marais, V. F. Curto and A. Salleo, Molecularly selective nanoporous membrane-based wearable organic electrochemical device for noninvasive cortisol sensing, Sci. Adv., 2018, 4, eaar2904 CrossRef PubMed.
- G. D. Spyropoulos, J. N. Gelinas and D. Khodagholy, Internal ion-gated organic electrochemical transistor: A building block for integrated bioelectronics, Sci. Adv., 2019, 5, eaau7378 CrossRef PubMed.
- P. Gkoupidenis, D. A. Koutsouras, T. Lonjaret, J. A. Fairfield and G. G. Malliaras, Orientation selectivity in a multi-gated organic electrochemical transistor, Sci. Rep., 2016, 6, 27007 CrossRef CAS PubMed.
- P. Gkoupidenis, D. A. Koutsouras and G. G. Malliaras, Neuromorphic device architectures with global connectivity through electrolyte gating, Nat. Commun., 2017, 8, ncomms15448 CrossRef PubMed.
- D. A. Koutsouras, T. Prodromakis, G. G. Malliaras, P. W. M. Blom and P. Gkoupidenis, Functional Connectivity of Organic Neuromorphic Devices by Global Voltage Oscillations, Adv. Intell. Syst., 2019, 1, 1900013 CrossRef.
- S. Pecqueur, et al., Neuromorphic Time-Dependent Pattern Classification with Organic Electrochemical Transistor Arrays, Adv. Electron. Mater., 2018, 4, 1800166 CrossRef.
- J. Rivnay, et al., Organic electrochemical transistors. Nature Reviews, Materials, 2018, 3, 17086 CAS.
- R. M. Owens and G. G. Malliaras, Organic Electronics at the Interface with Biology, MRS Bull., 2010, 35, 449–456 CrossRef CAS.
- I. Uguz, et al., A Microfluidic Ion Pump for In Vivo Drug Delivery, Adv. Mater., 2017, 29, 1701217 CrossRef PubMed.
- M. Bisio et al. , Closed-Loop Systems and In Vitro Neuronal Cultures: Overview and Applications, in In Vitro Neuronal Networks: From Culturing Methods to Neuro-Technological Applications, ed. M. Chiappalone, V. Pasquale and M. Frega, Springer International Publishing, 2019, pp. 351–387, DOI:10.1007/978-3-030-11135-9_15.
- M. Kaisti, Detection principles of biological and chemical FET sensors, Biosens. Bioelectron., 2017, 98, 437–448 CrossRef CAS PubMed.
- M. Berggren and G. G. Malliaras, How conducting polymer electrodes operate, Science, 2019, 364, 233–234 CAS.
- M. Giordani, et al., Specific Dopamine Sensing Based on Short-Term Plasticity Behavior of a Whole Organic Artificial Synapse. ACS, Sensors, 2017, 2, 1756–1760 CAS.
- M. Braendlein, et al., Lactate Detection in Tumor Cell Cultures Using Organic Transistor Circuits, Adv. Mater., 2017, 29, 1605744 CrossRef PubMed.
- G. Scheiblin, R. Coppard, R. M. Owens, P. Mailley and G. G. Malliaras, Referenceless pH Sensor using Organic Electrochemical Transistors, Adv. Mater. Technol., 2017, 2, 1600141 CrossRef.
- T. Mano, et al., Printed Organic Transistor-Based Enzyme Sensor for Continuous Glucose Monitoring in Wearable Healthcare Applications, ChemElectroChem, 2018, 5, 3881–3886 CrossRef CAS.
- E. Bihar, et al., A fully inkjet-printed disposable glucose sensor on paper, npj Flexible Electron., 2018, 2, 30 CrossRef.
- C. Diacci, et al., Real-Time Monitoring of Glucose Export from Isolated Chloroplasts Using an Organic Electrochemical Transistor, Adv. Mater. Technol., 2019, 1, 1900262 CrossRef.
- C. Diacci, et al., Label-free detection of interleukin-6 using electrolyte gated organic field effect transistors, Biointerphases, 2017, 12, 05F401 CrossRef PubMed.
- M. Berto, et al., Label free detection of plant viruses with organic transistor biosensors, Sens. Actuators, B, 2019, 281, 150–156 CrossRef CAS.
- L. Zhang, et al., Selective recognition of Histidine enantiomers using novel molecularly imprinted organic transistor sensor, Org. Electron., 2018, 61, 254–260 CrossRef CAS.
- S. P. White, K. D. Dorfman and C. D. Frisbie, Operating and Sensing Mechanism of Electrolyte-Gated Transistors with Floating Gates: Building a Platform for Amplified Biodetection, J. Phys. Chem. C, 2016, 120, 108–117 CrossRef CAS.
- S. P. White, C. D. Frisbie and K. D. Dorfman, Detection and Sourcing of Gluten in Grain with Multiple Floating-Gate Transistor Biosensors, ACS Sens., 2018, 3, 395–402 CrossRef CAS PubMed.
- A. Jonsson, et al., Bioelectronic neural pixel: Chemical stimulation and electrical sensing at the same site, PNAS, 2016, 113, 9440–9445 CrossRef CAS PubMed.
- Y. Kim, et al., A bioinspired flexible organic artificial afferent nerve, Science, 2018, 360, 998–1003 CrossRef CAS PubMed.
- A.-M. Pappa, et al., Organic Transistor Arrays Integrated with Finger-Powered Microfluidics for Multianalyte Saliva Testing, Advanced Healthcare, Materials, 2016, 5, 2295–2302 CrossRef CAS PubMed.
- D. J. Poxson, et al., Regulating plant physiology with organic electronics, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 4597–4602 CrossRef CAS PubMed.
- A. Jonsson, T. A. Sjöström, K. Tybrandt, M. Berggren and D. T. Simon, Chemical delivery array with millisecond neurotransmitter release, Sci. Adv., 2016, 2, e1601340 CrossRef PubMed.
- C. M. Proctor, et al., An Electrocorticography Device with an Integrated Microfluidic Ion Pump for Simultaneous Neural Recording and Electrophoretic Drug Delivery In Vivo, Adv. Biosyst., 2019, 3, 1800270 CrossRef.
- Y. Lee and T.-W. Lee, Organic Synapses for Neuromorphic Electronics: From Brain-Inspired Computing to Sensorimotor Nervetronics, Acc. Chem. Res., 2019, 52, 964–974 CrossRef CAS PubMed.
- C. Wan, et al., An Artificial Sensory Neuron with Tactile Perceptual Learning, Adv. Mater., 2018, 30, 1801291 CrossRef PubMed.
- Y. Wang, et al., Photonic Synapses Based on Inorganic Perovskite Quantum Dots for Neuromorphic Computing, Adv. Mater., 2018, 30, 1802883 CrossRef PubMed.
- Y. Lee, et al., Stretchable organic optoelectronic sensorimotor synapse, Sci. Adv., 2018, 4, eaat7387 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2019 |