Visible-light-driven activation of persulfate over cyano and hydroxyl group co-modified mesoporous g-C3N4 for boosting bisphenol A degradation†
Abstract
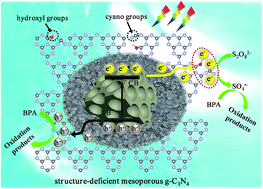
In this work, structure-deficient mesoporous g-C3N4 (DMCN) was fabricated via a facile hard template approach with water-bath aging pretreatment. The in situ introduced cyano groups (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) and hydroxyl groups (–OH) effectively modulated the energy levels, improved visible light absorption, and meanwhile served as strong electron-withdrawing groups, promoting the efficient separation and transfer of photogenerated charge carriers. With the addition of persulfate (PS) as an oxidant, DMCN exhibited superior catalytic activity and stability for bisphenol A (BPA) degradation. 100% BPA could be degraded for optimal DMCN-3.5 with 0.5 g L−1 catalyst and 1.0 g L−1 PS under visible light (420 nm ≤ λ ≤ 780 nm) within 15 min, whose reaction rate (0.317 min−1) was ∼39.6 times higher than that of bulk g-C3N4 (0.008 min−1). Based on EPR and quenching tests, h+ and SO4˙− radicals were determined as major oxidizing species in the DMCN/PS/Vis system. The enhanced catalytic performance was mainly attributed to PS serving as the electron acceptor and transfer of photogenerated e− to PS via –C
N) and hydroxyl groups (–OH) effectively modulated the energy levels, improved visible light absorption, and meanwhile served as strong electron-withdrawing groups, promoting the efficient separation and transfer of photogenerated charge carriers. With the addition of persulfate (PS) as an oxidant, DMCN exhibited superior catalytic activity and stability for bisphenol A (BPA) degradation. 100% BPA could be degraded for optimal DMCN-3.5 with 0.5 g L−1 catalyst and 1.0 g L−1 PS under visible light (420 nm ≤ λ ≤ 780 nm) within 15 min, whose reaction rate (0.317 min−1) was ∼39.6 times higher than that of bulk g-C3N4 (0.008 min−1). Based on EPR and quenching tests, h+ and SO4˙− radicals were determined as major oxidizing species in the DMCN/PS/Vis system. The enhanced catalytic performance was mainly attributed to PS serving as the electron acceptor and transfer of photogenerated e− to PS via –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N and C–OH groups, resulting in the efficient activation of PS for SO4˙− and a high light quantum efficiency. This work gives novel insights into in situ defect engineering for g-C3N4 and offers a new chance for visible-light-induced sulfate radical-based Fenton-like system to govern contaminated water.
N and C–OH groups, resulting in the efficient activation of PS for SO4˙− and a high light quantum efficiency. This work gives novel insights into in situ defect engineering for g-C3N4 and offers a new chance for visible-light-induced sulfate radical-based Fenton-like system to govern contaminated water.


 Please wait while we load your content...
Please wait while we load your content...