Rhodium nanoparticles supported on covalent triazine-based frameworks as re-usable catalyst for benzene hydrogenation and hydrogen evolution reaction†
Abstract
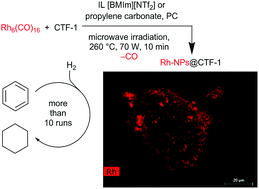
Metal nanoparticles (M-NPs) of ruthenium, rhodium, iridium and platinum were synthesized and supported on covalent triazine-based framework from 1,4-dicyanobenzene (CTF-1) by rapid microwave induced decomposition of their binary metal(0) carbonyls for Ru, Rh and Ir or Pt(acac)2 in the presence of CTF-1 in the ionic liquid (IL) 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ([BMIm][NTf2]) or in propylene carbonate (PC). (High-resolution) transmission electron microscopy, (HR-)TEM showed the formation of M-NPs on CTF-1 with, e.g., size distributions of 3.0 (±0.5) nm for Ru@CTF-1 synthesized in [BMIm][NTf2] and 2 (±1) nm for Rh@CTF-1 synthesized in PC. The crystalline phases of the M-NPs and the absence of significant impurities were verified by powder X-ray diffraction (PXRD) and selected area electron diffraction (SAED). The metal content of the M@CTF-1 composites was determined by flame atomic absorption spectroscopy (AAS) to be between 3 and 12 wt%. The Rh@CTF-1 composite nanomaterial proved to be a highly active (∼31 000 mol cyclohexane per (mol Rh) per h) heterogeneous catalyst for the hydrogenation of benzene to cyclohexane under mild (10 bar H2, 70 °C) and solvent-free conditions with over 99% conversion. The catalyst could be re-used for at least ten consecutive hydrogenation reactions. Additionally, Rh@CTF-1 is an active electrocatalyst for the hydrogen evolution reaction (HER) with an operating potential of −58 mV, while Pt@CTF-1 and commercial Pt/C shows a more negative operating potential of −111 and −77 mV. Also the onset potential of −31 mV for Rh@CTF-1 is much more positive than that of Pt@CTF-1 (−44 mV) and commercial Pt/C (−38 mV).


 Please wait while we load your content...
Please wait while we load your content...