A rate equation model for the energy transfer mechanism of a novel multi-color-emissive phosphor, Ca1.624Sr0.376Si5O3N6:Eu2+†
Abstract
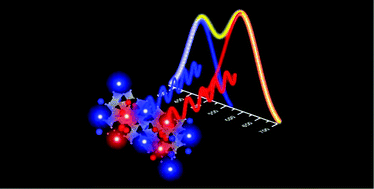
Multi-color emissions (or broadband emissions) from a single-phase phosphor with a single activator are an unfamiliar idea compared with those from multi-color-center materials. A single activator that is located in different crystallographic sites of a single-phase phosphor, however, could lead to multimodal emission peaks for multi-color (or broadband) emissions. The discovery of a single-phase-single-activator-broadband-phosphor is rare, and it is regarded as difficult to accomplish. The present investigation introduces a novel single-phase-single-activator-broadband-phosphor (Ca1.624Sr0.376Si5O3N6:Eu2+) and provides an in-depth examination of the energy transfer between different crystallographic sites which is the governing mechanism for the broadband emissions. Structural analysis is backed up by density functional theory (DFT) calculations, which validate the structural model of the discovered novel phosphor. Rate-equation modeling is introduced based on particle swarm optimization (PSO) to provide a complete quantitative analysis for the mechanism of the energy transfer.


 Please wait while we load your content...
Please wait while we load your content...