Full solid-phase total synthesis of macrocyclic natural peptides using four-dimensionally orthogonal protective groups
Abstract
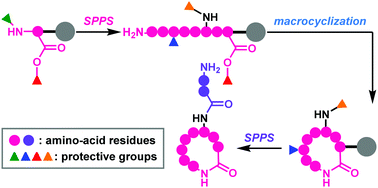
Fmoc-based solid-phase synthesis provides efficient access to both linear and macrocyclic peptides. To synthesize complex macrocyclic polyamides using Fmoc chemistry, multiple protective groups with orthogonal reactivities are generally employed because the free amines and carboxylic acids of specific residues must be selectively exposed prior to amide formation. This review focuses on four-dimensionally orthogonal protective group strategies for the full solid-phase synthesis of macrocyclic peptides with branched chains (polymyxin E2 and daptomycin) and a tricyclic natural peptide (lacticin 481).
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...