Astemizole-based turn-on fluorescent probes for imaging hERG potassium channel†
Abstract
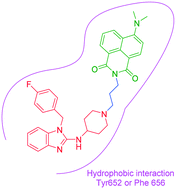
Based on the scaffold of astemizole, three novel turn-on fluorescent probes (N1–N3) for human ether-a-go-go-related gene (hERG) potassium channel were developed herein. These probes have reasonable fluorescence properties, acceptable cell toxicity, and potent inhibitory activity, all of which contribute to cell imaging at the nanomolar level. Overall, these probes have the potential for setting up a screening system for hERG channels.


 Please wait while we load your content...
Please wait while we load your content...