 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correction: A biocatalytic cascade for the conversion of fatty acids to fatty amines
Joan
Citoler
a,
Sasha R.
Derrington
a,
James L.
Galman
a,
Han
Bevinakatti
b and
Nicholas J.
Turner
*a
aSchool of Chemistry, University of Manchester, Manchester Institute of Biotechnology, 131 Princess Street, Manchester, M1 7DN, UK. E-mail: Nicholas.Turner@manchester.ac.uk
bNouryon (formerly AkzoNobel Specialty Chemicals), 10 Finderne Ave, Bridgewater, NJ 08807, USA
First published on 23rd October 2019
Abstract
Correction for ‘A biocatalytic cascade for the conversion of fatty acids to fatty amines’ by Joan Citoler, et al., Green Chem., 2019, 21, 4932–4935.
Table 1 in the published manuscript is not displayed as was intended. Table 1 should be displayed as below.
| Substrate | Structure | n | Conversion to amine (%) |
|---|---|---|---|
| a Reaction conditions: 5 mM carboxylic acid, 31.25 mM IPA, 2 mg mL−1McCAR lysate, 2 mg mL−1Sp-TA lysate, 2 mg mL−1 PAP lysate, 2 mg mL−1 Adk lysate, 0.2 mg mL−1 CDX-901 GDH, 0.5 mM NADP+, 0.5 mM PLP, 4 mg mL−1 PolyP, 25 mM D-glucose, 5.5 mM MgCl2, 5% (v/v) DMSO, 100 mM pH 8 Tris buffer, 500 μL reaction volume, 500 μL n-heptane. 30 °C, 180 rpm, 20 h. | |||
| 1a |
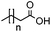
|
3 | 94 |
| 1b | 5 | 96 | |
| 1c | 7 | 93 | |
| 1d | 9 | 92 | |
| 1e | 11 | 44 | |
| 1f | 13 | 9 | |
| 1g | 15 | 1 | |
| 1h |
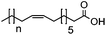
|
2 | 89 |
| 1i | 4 | 58 | |
| 1j | 6 | 6 | |
| 1k |
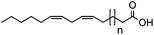
|
5 | 35 |
| 1l |

|
5 | 63 |
The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
| This journal is © The Royal Society of Chemistry 2019 |
