The determination of two emerging perfluoroalkyl substances and related halogenated sulfonic acids and their significance for the drinking water supply chain†
Abstract
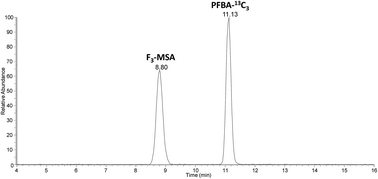
In the present study analytical methodologies were developed for two newly emerging polar perfluorinated alkyl substances (PFAS), namely F3-MSA, and HFPO-DA, in order to assess the occurrence and levels of these PFAS in Dutch and Belgian waters. Two separate methods were needed for analysing F3-MSA and HFPO-DA. A mixed-mode and a reversed phase C18 method were developed for F3-MSA and HFPO-DA, respectively, using a high resolution Orbitrap Fusion mass spectrometer for detection, yielding satisfactory LOD and LOQ results for both analytes. A sample campaign was performed collecting single grab samples from various locations and different stages of the drinking water production chain. Whereas both PFAS were absent in groundwaters, they were found to be present in surface waters, river bank and dune infiltrates, process water, and drinking water, demonstrating the persistence and mobility of both compounds. Based on provisional health-based guideline values (0.15 μg L−1 for HFPO-DA, 11.9 mg L−1 for F3-MSA), the current levels in drinking water from the suppliers involved in this study do not pose a health risk for the human population. Common removal processes used in drinking water production appeared to remove these polar compounds at most partially. At locations close to potential sources of these chemicals (e.g. fluoropolymer production sites), the quality of surface water or river bank filtrate abstracted for production of drinking water must therefore be monitored.
- This article is part of the themed collection: PFAS


 Please wait while we load your content...
Please wait while we load your content...