Facile synthesis of Pd(ii) and Ni(ii) pincer carbene complexes by the double C–H bond activation of a new hexahydropyrimidine-based bis(phosphine): catalysis of C–N couplings†
Abstract
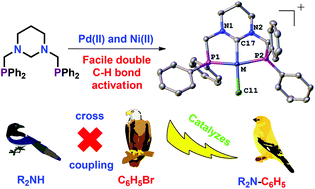
Hexahydropyrimidine-based bis(phosphine), a pro-NHC ligand, was synthesized in one step and excellent yield. It underwent spontaneous double C–H bond activation to give cationic pincer NHC complexes of the type [(PCP)MCl]X (M = Pd, Ni and X = Cl, BF4) in the absence of any external reagents. Their structures were determined by X-ray diffraction methods and the mechanism of formation of palladium carbene complexes as analyzed by DFT calculations showed two transition states. The Pd(II) carbene complex effectively catalyzes a few C–N cross coupling reactions.
- This article is part of the themed collection: New Talent: Asia-Pacific


 Please wait while we load your content...
Please wait while we load your content...