Fe3O4@l-Proline/Pd nanocomposite for one-pot tandem catalytic synthesis of (±)-warfarin from benzyl alcohol: synergistic action of organocatalyst and transition metal catalyst†‡
Abstract
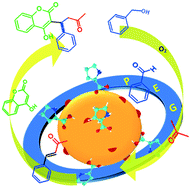
One-pot synthesis of (±)-warfarin, an anticoagulant, has been achieved from benzyl alcohol in a ‘green way’ by using a multicomponent catalyst. For the purpose, L-proline capped Fe3O4 nanoparticles (Fe3O4@L-proline NPs) were synthesized and metallic palladium was loaded on its surface (Fe3O4@L-proline/Pd NCs). The morphology, particle size and shape were studied by using FESEM and TEM analysis. The Pd present on the surface was responsible for oxidation of benzyl alcohol and its derivatives to the corresponding aldehyde in situ. This in turn, condensed with acetone to form the aldol condensation product, benzylideneacetone, at 70 °C due to the presence of the L-proline organocatalyst on the surface of Fe3O4 NPs. Later, 4-hydroxycoumarin was introduced to condense with in situ generated benzylideneacetone by a Michael addition to form the target product (±)-warfarin. It was established that benzyl alcohol can be converted into the final product, (±)-warfarin, with an overall 35% yield within 5 days in a single-pot process. This process requires a rise in temperature in stages to a maximum of 100 °C and 1 atm pressure of dioxygen gas. An important aspect of the developed process is the avoidance of loss of costly Pd by leaching and catalyst recovery by the use of a magnetic field. The use of a solvent like PEG-400 makes the process green in a true sense. The interaction of L-proline with Fe3O4 NPs and the presence of Pd on the surface were confirmed by the FTIR and XRD patterns, respectively. The present study hereby suggests a combined 3-step mechanism for the production of the target product warfarin. Pilot-scale one-pot production of (±)-warfarin was carried out and a flow diagram with various unit processes is presented.


 Please wait while we load your content...
Please wait while we load your content...