Acellular biomaterial strategies for endodontic regeneration
Abstract
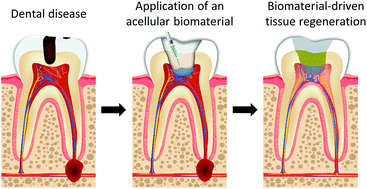
Dental decay is treated by removing infected dental tissues such as dentine and restoring the tooth with a material. However, the vast majority of these materials have been designed to be mechanically robust and bioinert, whereas the potential regenerative properties of a biomaterial have not been considered. In endodontics for example, materials are used to seal the pulp cavity to avoid bacterial colonisation of the tooth and prevent further infection. While these treatments are effective in the short term, many of these materials have not been designed to interface with the pulp tissue in a biocompatible manner and are often cytotoxic. This can lead to less favourable long-term outcomes such as devitalisation of the tooth via root-canal therapy or extraction of the tooth. Clinical outcomes could be improved if regenerative approaches were followed whereby the biology of the tooth is engineered for repair and regeneration often with the support of a biomaterial. Within these, acellular or cell homing approaches are particularly interesting, as some regulatory hurdles associated with cellular therapies could be circumvented which may aid their clinical translation. In this review, we highlight progress in regenerative dentistry and focus on exciting developments using acellular biomaterials for regenerating dental tissues.
- This article is part of the themed collection: Biomaterials Science Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...