A multi-state fluorescent switch with multifunction of AIE, methanol-responsiveness, photochromism and mechanochromism†
Abstract
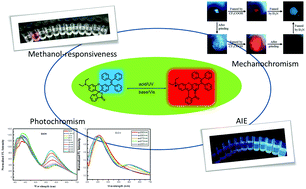
In this article, we report a newly designed molecule, RhTPE, synthesized by fusing rhodamine with TPE, which realizes both emission color change and on/off switching. Compared to most of the reported fluorescent switches, such a unique structure endowed RhTPE with the multifunction of AIE, methanol-responsiveness, photochromism and mechanochromism, owing to the dynamic ring opening–closing isomerization. RhTPE is acid-sensitive and can recover to the ring-closed form (OF) by base treatment. Both RhTPE and trifluoroacetic acid (TFA)-treated RhTPE display AIE properties. It is noteworthy that during the aggregation process of TFA-treated RhTPE, the cyan emission strengthens while the red emission disappears, due to the aggregation-induced ring-closing reaction. RhTPE shows selective responsiveness to methanol out of all the twelve screened solvents, probably owing to the low pKa value and high polarity of methanol. More interestingly, RhTPE demonstrates excitation wavelength-dependent photochromism in several different organic solvents, which is very rarely reported in rhodamine systems. In the solid state, RhTPE has three well-distinguished high contrast states of cyan, red and dark. The reversible switch between any two states can be achieved with acid/base or mechanical force/chemical vapor.
- This article is part of the themed collection: 2018 Journal of Materials Chemistry C HOT Papers


 Please wait while we load your content...
Please wait while we load your content...