Open Access Article
Open Access ArticleChemical synthesis of culmorin metabolites and their biologic role in culmorin and acetyl-culmorin treated wheat cells†
Julia
Weber
 a,
Marta
Vaclavikova
b,
Gerlinde
Wiesenberger
a,
Marta
Vaclavikova
b,
Gerlinde
Wiesenberger
 c,
Maximilian
Haider
c,
Maximilian
Haider
 a,
Christian
Hametner
a,
Johannes
Fröhlich
a,
Christian
Hametner
a,
Johannes
Fröhlich
 a,
Franz
Berthiller
a,
Franz
Berthiller
 b,
Gerhard
Adam
b,
Gerhard
Adam
 c,
Hannes
Mikula
c,
Hannes
Mikula
 a and
Philipp
Fruhmann
a and
Philipp
Fruhmann
 *ad
*ad
aInstitute of Applied Synthetic Chemistry, Vienna University of Technology (TU Wien), Austria. E-mail: Philipp.fruhmann@tuwien.ac.at
bDepartment of Agrobiotechnology (IFA-Tulln), Center for Analytical Chemistry, University of Natural Resources and Life Sciences, Vienna (BOKU), Austria
cDepartment of Applied Genetics and Cell Biology, University of Natural Resources and Life Sciences, Vienna (BOKU), Tulln, Austria
dCenter for Electrochemical Surface Technology (CEST), Wiener Neustadt, Austria
First published on 21st February 2018
Abstract
The Fusarium metabolite culmorin (1) is receiving increased attention as an “emerging mycotoxin”. It co-occurs with trichothecene mycotoxins and potentially influences their toxicity. Its ecological role and fate in plants is unknown. We synthesized sulfated and glucosylated culmorin conjugates as potential metabolites, which are expected to be formed in planta, and used them as reference compounds. An efficient procedure for the synthesis of culmorin sulfates was developed. Diastereo- and regioselective glucosylation of culmorin (1) was achieved by exploiting or preventing unexpected acyl transfer when using different glucosyl donors. The treatment of a wheat suspension culture with culmorin (1) revealed an in planta conversion of culmorin into culmorin-8-glucoside (6) and culmorin acetate, but no sulfates or culmorin-11-glucoside (7) was found. The treatment of wheat cells with the fungal metabolite 11-acetylculmorin (2) revealed its rapid deacetylation, but also showed the formation of 11-acetylculmorin-8-glucoside (8). These results show that plants are capable of extensively metabolizing culmorin.
Introduction
Mycotoxins are widespread contaminants in food and feed. In-depth knowledge about their toxicity, metabolism and occurrence is of great importance.1 Besides a number of well-investigated mycotoxins such as deoxynivalenol, several compounds are receiving increased attention as so-called “emerging mycotoxins”. They frequently co-occur with other toxins and their toxicity and ecological role and the effect of combinations with known toxins have been insufficiently investigated so far.2Culmorin (CUL, 1, Fig. 1a) is considered an “emerging mycotoxin” although it was isolated in 1937, followed by its structural characterization in 1967.3,4 It possesses a sesquiterpene diol core structure and is produced by several different Fusarium species, such as F. culmorum (name giving), F. graminearum, F. crookwellense and F. venenatum. Moreover, it is produced by a recently identified basal species of the F. graminearum species complex, Fusarium praegraminearum, indicating that CUL production is an ancestral trait. In general, CUL is little investigated and might also be produced by other Fusarium species. Outside of the genus Fusarium, it has been identified in the marine ascomycete Leptosphaeria oreamaris.5–9 Besides CUL, various Fusarium species have been shown to produce related compounds including hydroxyculmorins, culmorone and hydroxyculmorone (Fig. 1b).7,10,11
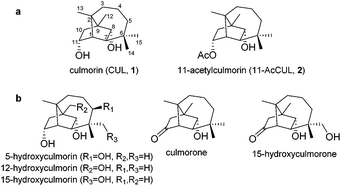 | ||
| Fig. 1 (a) Chemical structure of culmorin (CUL, 1) and 11-acetylculmorin (11-AcCUL, 2); (b) naturally occurring related compounds of CUL (1). | ||
The natural occurrence and contamination levels of CUL are usually strongly related to the occurrence and levels of deoxynivalenol (DON). Typically, CUL was found to occur in concentrations about 3-fold higher than those of DON in naturally contaminated samples.12,13 Although different parameters may influence the ratio of CUL and its metabolites, there are several examples of their occurrence in different regions of the world. In a recent study, CUL was detected in nearly all wheat, barley and oat samples from Norway with a median concentration of 100 (wheat), 292 (barley) and 2000 μg kg−1 (oats).13 A study with feed and feed raw material samples from Austria, Denmark and Hungary led to similar results, where 63% of all samples contained CUL with a median concentration of 195 μg kg−1.14 The frequent occurrence of CUL has been shown in a recent study reporting a median concentration of approximately 100 μg kg−1 in cereals, nuts and their processed products from Cameroon.15
There are only a limited number of studies describing the toxicological relevance of CUL. The compound has been shown to possess antifungal and phytotoxic properties.2,7,11 Previous studies on the toxicity of CUL include tests in baby hamster kidney cells (toxic at 20 μg mL−1), a chick embryo toxicity screening test (CHEST, LD50 around 70 μg CUL per egg) and the estimation of the LD50 (i.p.) for mice with a range of 250 to 1000 μg per kg BW.7,8,16 In all these trials, only high amounts of CUL were used and the obtained values therefore suggest a low toxicity to animal cells. The metabolism of CUL in humans or animals is unknown. To date, in vivo studies have been carried out only in swine and two insect species.17,18 Caterpillars of corn earworm (Heliothis zea) and armyworm (Spodoptera frugiperda) reared on a CUL-containing diet (25 mg kg−1, 7 days) showed an unaltered weight gain and mortality rate. A similar study with growing piglets (2 mg kg−1 diet, 21 days) also showed no negative impact. In these two studies, additionally, the combined effect of DON and CUL was investigated. Interestingly, the combination of DON (25 mg kg−1 diet) and CUL (10 mg kg−1 diet) significantly increased the mortality and decreased the weight gain of corn earworms.17 Although the evidence for a synergistic effect is still very limited, an enzyme involved in the biosynthesis of CUL has been proposed to be a target for mycotoxin reduction.19 The role of CUL in plant–pathogen interaction is unknown.
The biosynthesis of CUL has been elucidated in F. graminearum. A terpenoid synthase encoded by CLM1 produces longiborneol that is subsequently hydroxylated at C11 by a cytochrome P450 encoded by CLM2.20,21 CUL biosynthesis is induced in planta during the infection of several crop species and is co-regulated with DON biosynthesis,22,23 indicating a possible role of CUL as a Fusarium virulence factor. In general, plants may have the ability to at least partly inactivate and counteract fungal virulence factors using various mechanisms.24 Data regarding CUL metabolites formed by plants and their biological significance are lacking. Since the most prominent and important metabolites are usually the glycosylated forms of the parent toxin, it seems obvious to speculate that CUL glucosides (CUL-Glc) might also be formed.25 In addition, the occurrence of sulfated derivatives similar to other mycotoxins like DON, ZEN or AOH/AME seems possible (Fig. 2).26–29
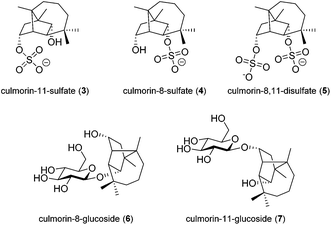 | ||
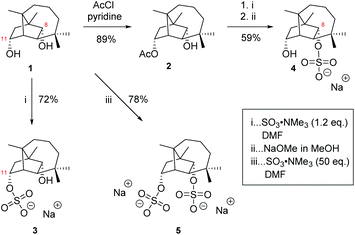
| Fig. 2 Target molecules for the in planta investigation: culmorin sulfates (3, 4), the corresponding disulfate (5) and both possible glucosides (6, 7). | ||
CUL is a longifolene sesquiterpene with a tricyclo-[6.3.0.0] undecane skeleton and the core structure appears in several other natural products such as longifolene or longiborneol. Several strategies for the synthesis of CUL and its core structure have been reported.30–33 The acetylation of CUL affording 8-acetyl- and 11-acetylculmorin (11-AcCUL 2, Fig. 1a) was described very recently by Bahadoor et al.20 Besides this, no further synthetic modifications or derivatizations have been described.
Wheat is an important crop plant infected by CUL-producing Fusarium species. We chose wheat suspension culture cells for the CUL metabolism tests because the application and extraction of the test compound are not hampered by anatomical structures and the variable macromolecular matrix (wax, lignin, starchy endosperm) as in developing wheat kernels, and the response of cells to the test compound in vitro is more uniform. The conjugation reactions catalyzed by glucosyltransferases and sulfatases are the expected metabolization pathways in plants (and potentially other organisms). We therefore set out to synthesize all possible CUL glucosides and sulfates, which could be used to develop analytical methods to allow the investigation of the natural occurrence of these conjugates.
Results and discussion
Synthesis of culmorin metabolites
To obtain sufficient amounts of CUL for synthetic studies, it was necessary to produce it on our own. We selected tri1 knockout strains derived from the Fusarium graminearum strains PH-1 and WG-934 which allowed the simultaneous production of calonectrin (for other studies) and as a by-product also high levels of CUL. A purification protocol was developed to obtain CUL (885 + 120 mg) and 11-AcCUL (42 mg), which represents a yield of 333 mg kg−1 (rice culture) and 177 mg L−1 (liquid culture). The identity and purity of both compounds were confirmed by NMR spectroscopy.10,20For the synthesis of the desired CUL sulfates, we used a procedure originally developed for the sulfation of carbohydrates.34 In a first attempt, CUL was reacted with a SO3-trimethylamine (NMe3) complex in N,N-dimethylformamide (DMF) at 55 °C showing the unexpected exclusive formation of CUL-11-sulfate as a trimethylammonium salt. To obtain sodium salt 3, which is required for biological investigations, the purified product was subjected to a cation exchange resin. Using the same procedure with a 50-fold excess of SO3-NMe3, CUL was sulfated at positions O-8 and O-11 affording CUL disulfate (5). The reactivity difference of the two hydroxyl groups is noteworthy and made it apparent that the hydroxyl group of CUL at position 11 needs to be protected to be able to produce CUL-8-sulfate (4). Hence, 11-AcCUL (2) that was previously isolated from the Fusarium culture material was used as a starting material and sulfated in a similar approach. The deprotection of the acetyl group was finally accomplished under basic conditions35 yielding directly the sodium salt of the sulfate 4. To make 11-AcCUL (2) easily accessible via a synthetic route, we screened for a regioselective acetylation method and were successful by treating CUL with acetyl chloride in pyridine (Scheme 1).
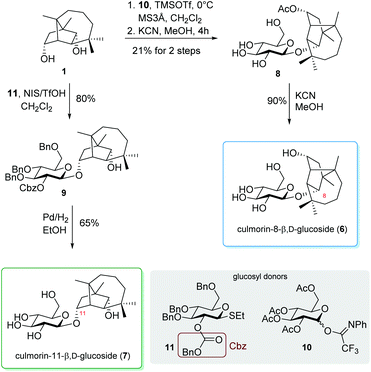
The glucosylation of CUL was performed by applying Lewis acid mediated glycosylation using an N-phenyltrifluoroacetimidoyl (NPTFA) glucosyl donor 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35,36 and a catalytic amount of TMSOTf (0.1 eq.). Unexpectedly, the glucosylation of CUL didn't take place like the sulfation of CUL on the hydroxyl group at pos. 11 but at pos. 8 and formed 11-AcCUL-8-β,D-tetra-O-acetylglucoside as an intermediate. We assume that this is caused by glucosylation after an initial acyl transfer, a known side reaction of glycosylation.37,38 Selective deprotection of the sugar moiety was achieved using sodium cyanide39 yielding 11-AcCUL-8-glucoside (11-AcCUL-8-Glc, 8). Further deprotection by using a fourfold excess of sodium cyanide afforded CUL-8-glucoside (CUL-8-Glc, 6).
35,36 and a catalytic amount of TMSOTf (0.1 eq.). Unexpectedly, the glucosylation of CUL didn't take place like the sulfation of CUL on the hydroxyl group at pos. 11 but at pos. 8 and formed 11-AcCUL-8-β,D-tetra-O-acetylglucoside as an intermediate. We assume that this is caused by glucosylation after an initial acyl transfer, a known side reaction of glycosylation.37,38 Selective deprotection of the sugar moiety was achieved using sodium cyanide39 yielding 11-AcCUL-8-glucoside (11-AcCUL-8-Glc, 8). Further deprotection by using a fourfold excess of sodium cyanide afforded CUL-8-glucoside (CUL-8-Glc, 6).
For the preparation of CUL-11-glucoside (CUL-11-Glc, 7), we aimed to use glucosyl donors as very recently developed by our group.40 These 2-O-benzyloxycarbonyl (Cbz) protected donors can be applied for diastereoselective glucosylation without the need for acetyl or any acyl groups in general. Hence, side reactions such as orthoester formation and acyl transfer are prevented. The activation of 2-Cbz glycosyl donor 11 with N-iodosuccinimide and trifluoromethanesulfonic acid selectively formed benzyl protected CUL-11-glucoside 9. Subsequent deprotection by palladium-catalyzed hydrogenation41 afforded CUL-11-glucoside (7) in a yield of 90% (Scheme 2).
Metabolism of culmorin (1) and 11-acetyl-culmorin (9) in a wheat suspension culture
To investigate the metabolic fate of CUL in planta, a wheat suspension culture was treated with either CUL or 11-AcCUL (2, each 100 mg L−1) and the respective metabolites were analyzed after one day and one week, respectively. At the end point, the supernatant was removed and mixed in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with methanol to stop further reactions. The cell pellet was washed twice with 50% methanol to remove the adsorbed compounds, and the volume of the combined wash solution was brought up to twofold the volume of the initial culture. The cell pellet was disrupted by sonication and the volume was also brought up to twofold the culture volume with methanol of 50% final concentration (see the ESI†).
1 with methanol to stop further reactions. The cell pellet was washed twice with 50% methanol to remove the adsorbed compounds, and the volume of the combined wash solution was brought up to twofold the volume of the initial culture. The cell pellet was disrupted by sonication and the volume was also brought up to twofold the culture volume with methanol of 50% final concentration (see the ESI†).
In the case of 11-AcCUL (2), only 1-day incubation was investigated, because of the expected rapid deacetylation by the cells. The medium without cells was used as a control showing no background signals for CUL or any of its metabolites. The values obtained for CUL dissolved in cell free medium after 1 week were about 30% higher than the theoretical value of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μg L−1 (due to 1
000 μg L−1 (due to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dilution with methanol). This can only be partially explained by the evaporation of the 1.5 mL culture medium, but most likely by considerable matrix effects, which may be different and even more significant in the case of the cell culture and highly dependent on the age of the cells. Therefore our results are only semiquantitative. Yet, in the medium without cells CUL seemed to be stable for a week, while only about 37% of the 11-AcCUL (2) added to the medium without cells were measured in the spiked medium after 1 day. Nevertheless, no CUL was found, excluding non-enzymatic hydrolysis in the medium, and also pointing to matrix effects.
1 dilution with methanol). This can only be partially explained by the evaporation of the 1.5 mL culture medium, but most likely by considerable matrix effects, which may be different and even more significant in the case of the cell culture and highly dependent on the age of the cells. Therefore our results are only semiquantitative. Yet, in the medium without cells CUL seemed to be stable for a week, while only about 37% of the 11-AcCUL (2) added to the medium without cells were measured in the spiked medium after 1 day. Nevertheless, no CUL was found, excluding non-enzymatic hydrolysis in the medium, and also pointing to matrix effects.
The treatment of wheat cells with CUL revealed a clear reduction of the remaining CUL concentration in the medium (supernatant after spinning down the cells). While about 94% of CUL were detected after day 1, only 7% were observed after 1 week in one replicate, and in the other two replicates, CUL was already below the detection limit. Also the wash solution revealed interesting results. Assuming that in the worst case 50 μl medium may be retained in the 250 mg cell pellet, about 3% remaining CUL could be explained in the absence of any metabolization. Interestingly, in the wash solution on both the 1-day and the 7-day time points, about 11% of the CUL input was recovered, indicating considerable reversible binding of CUL to cells, presumably due to adsorption to cell wall structures preventing further metabolism.
CUL was metabolized in wheat cells to CUL-8-glucoside (6), and unexpectedly to 11-AcCUL (2), but no CUL sulfates were detected (Tables 1 & 2). Within 7 days the level of CUL-8-glucoside (6) in the supernatant increased from an average value of 95 μg L−1 to 355 μg L−1. CUL-8-glucoside (6) could also be detected with an increasing concentration in the wash solution. CUL has two hydroxyl groups, so theoretically two different glucosides are possible. Yet, only one was found in the plant extract and this had the same retention time and fragmentation pattern as the synthesized CUL-8-Glc (6). The importance of CUL-11-Glc (7) therefore seems to be negligible from a plant physiology point of view.
| Day | CUL-8-Glc [μg L−1] | |
|---|---|---|
| Culture supernatant | 1 | 95.0 |
| Culture supernatant | 7 | 355.0 |
| Cell wash solution | 1 | 17.1 |
| Cell wash solution | 7 | 98.1 |
| Cell extract | 1 | 108.0 |
| Cell extract | 7 | 86.1 |
| Day | 11-AcCUL [μg L−1] | Average [μg L−1] | |||
|---|---|---|---|---|---|
| # 1 | # 2 | # 3 | |||
| Supernatant | 1 | 11.8 | 11.9 | 10.2 | 11.3 |
| Supernatant | 7 | 7.4 | <LOD | <LOD | — |
Interestingly, we also observed the formation of 11-AcCUL (2) when treating wheat cells with CUL suggesting the presence of wheat enzymes capable of acetylating CUL. Yet, the concentrations were lower than observed for the glucosides (day 1) and surprisingly 11-AcCUL (2) was not detected in the cell extract.
As the acetylation is performed by intracellular transferases depending on acetyl-CoA, the product AcCUL is seemingly rapidly pumped out of the cell.
We also performed a feeding assay with 11-AcCUL (2) at 100 mg L−1. By normalization to the 11-AcCUL (2) concentration found after 1 day in the medium without cells (which should in first approximation correct for matrix effects), only one of the three replicates showed 0.2% remaining 11-AcCUL (2) in the supernatant, whereas the concentration was below the detection limit in the case of the two other replicates. 11-AcCUL (2) was obviously rapidly deacetylated, indicated by a maximum level of 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 μg L−1 CUL in the supernatant. In addition, CUL-8-Glc (6, 32.5 μg L−1 found in the supernatant), formed by the glucosylation of CUL, and 11-AcCUL-8-Glc (8, 483 μg L−1) were detected indicating that 11-AcCUL (2) can enter the cells followed by the glucosylation and excretion of 11-AcCUL-8-Glc (8).
300 μg L−1 CUL in the supernatant. In addition, CUL-8-Glc (6, 32.5 μg L−1 found in the supernatant), formed by the glucosylation of CUL, and 11-AcCUL-8-Glc (8, 483 μg L−1) were detected indicating that 11-AcCUL (2) can enter the cells followed by the glucosylation and excretion of 11-AcCUL-8-Glc (8).
Although we observed the acetylation of CUL, no acetylated CUL glucoside was found in any CUL treated sample. Hence, we assume that intracellular levels of AcCUL sufficient for glycosylation are only reached in 11-AcCUL (2) treated cells, while in the case of the intracellular formation of 11-AcCUL (2), it seems to be excreted more rapidly than glycosylated.
Conclusions
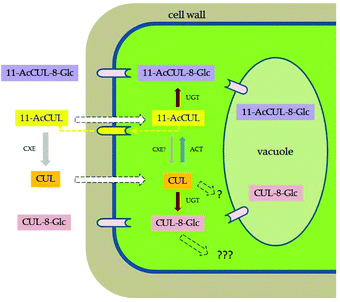
We were able to develop selective methods for the synthesis of CUL sulfates and glucosides (ESI† figures, NMR and MS spectra). Different glucosyl donors could be applied to exploit acetyl transfer to obtain CUL-8-Glc (6) and CUL-11-Glc (7) without the need for the laborious separation of two regioisomers. Considering the potential of the combination of the already described NPTFA donor (10) and our newly developed donor (11), we aim to use this system on other mycotoxins in order to selectively access masked mycotoxins. The in-depth evaluation of this donor and its use within the synthesis of other masked mycotoxins and metabolites is already in progress.Using the synthesized standards as calibrants we analyzed the culture media (supernatant), wash solution, and the extracts of the cells. Calibrants in neat solvents were used, rendering the results semiquantitative by not considering matrix effects. A validation of the analytical method would be necessary to evaluate the natural occurrence of CUL metabolites in cereals. A schematic representation of our results is shown in Fig. 3.
In contrast to a similar study treating wheat suspension culture cells with DON, no sulfate conjugates were identified in planta.28 While sulfation seems to be of minor relevance in plants, it is a prominent detoxification process for xenobiotics in poultry and other animal species.42 Recently the presence of DON sulfate in human urine has been reported for the first time.43 Data on the metabolic fate of CUL in humans and livestock are lacking. In an upcoming study, we intend to use the prepared CUL sulfates as analytical standards to assess the metabolization of CUL in poultry.
11-AcCUL (2) is a fungal metabolite and potentially a biosynthetic precursor of CUL similar to DON, which is formed by the deacetylation of the initial metabolites 3- and 15-acetyl-DON. Acetylated CUL may have higher membrane permeability, and thus reach targets in plants more easily. The wheat cells can rapidly deacetylate 11-AcCUL (2), seemingly by extracellular esterase. If high concentrations of 11-AcCUL (2) are applied externally, the compound enters the cell and is converted into 11-AcCUL-8-Glc (8) by cytosolic glucosyltransferases. Likewise when high concentrations of CUL are applied, CUL-8-Glc (6) is formed intracellularly.
Assuming that in the original culture only about 250 μl (250 mg wet weight of cells on average) of the total 1500 μl culture volume corresponds to intracellular space, the actual intracellular glucoside concentration is about 6× higher than what was measured in the extract. Consequently, the intracellular concentration of CUL-Glc (6 × 108 μg L−1) is about 6.8-fold higher than the glucoside concentration found in the medium (average 95 μg L−1). Although this result is only semiquantitative, it clearly indicates that at least a part of the CUL-Glc formed inside the plant is concentrated (presumably in vacuoles) but partly also translocated to the apoplast and released into the medium in the case of the suspension culture.
Interestingly, a small portion of the added CUL was converted into 11-AcCUL (2) and found in the medium, but not in the cells. The observed ability of the plant to acetylate mycotoxins is in line with recent findings in studies on the fate of T-2 and HT-2 in various cereals, wherein the formation of acetylated derivatives has been reported.44,45 In the case of trichothecenes, acetylating the C3-OH is clearly a detoxification reaction.46 It is not known which plant enzymes are responsible for this reaction, but CUL might be a competing substrate, modulating the toxicity of trichothecenes. In-depth studies are required to investigate whether a synergism exists between CUL and trichothecenes in plants.
On a molecular basis, only a minor part of CUL can be accounted for the formation of the identified metabolites. Besides other possible metabolization routes, CUL might be rapidly metabolized mainly into CUL-Glc and then further into still uncharacterized metabolites and conjugates. The low recovery is comparable to previously reported results for the mycotoxin zearalenone. The formation of several di-glucosides and malonylglucosides was reported as well as a large portion that eventually ends up as an “insoluble residue”.47,48 Further studies with a stable isotope or radiolabeled CUL will help to better understand its fate in planta and its ecological role. Since the toxicity of CUL itself is low, the newly described masked mycotoxins are probably of minor toxicological relevance, but provide insights on how plants cope with this suspected DON synergist.
Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
The authors would like to thank the Austrian Science Fund (SFB Fusarium F3701, F3702 and F3715) and the Austrian Research Promotion Agency (FFG, BRIDGE-Project 843488) for financial support including the open access publication costs. The authors thank Dr. Gerald Siegwart for maintaining the wheat suspension culture and Dr. Markus Schwarz for valuable support with HRMS measurements.References
- J. P. F. D'Mello and A. M. C. Macdonald, Anim. Feed Sci. Technol., 1997, 69, 155–166 CrossRef.
- C. Gruber-Dorninger, B. Novak, V. Nagl and F. Berthiller, J. Agric. Food Chem., 2017, 65(33), 7052–7070 CrossRef CAS PubMed.
- J. N. Ashley, B. C. Hobbs and H. Raistrick, Biochem. J., 1937, 31, 385–397 CrossRef CAS PubMed.
- D. H. R. Barton and N. H. Werstiuk, Chem. Commun., 1967, 30–31, 10.1039/C19670000030.
- R. Greenhalgh, R. M. Meier, B. A. Blackwell, J. D. Miller, A. Taylor and J. W. ApSimon, J. Agric. Food Chem., 1984, 32, 1261–1264 CrossRef CAS.
- D. R. Lauren, A. Ashley, B. A. Blackwell, R. Greenhalgh, J. D. Miller and G. A. Neish, J. Agric. Food Chem., 1987, 35, 884–889 CrossRef CAS.
- J. D. Miller and S. MacKenzie, Mycologia, 2000, 92, 764–771 CrossRef CAS.
- P. B. Pedersen and J. D. Miller, Nat. Toxins, 1999, 7, 305–309 CrossRef CAS PubMed.
- D. B. Strongman, J. D. Miller, L. Calhoun, J. A. Findlay and N. J. Whitney, Journal, 1987, 30, 21 Search PubMed.
- G. C. Kasitu, J. W. ApSimon, B. A. Blackwell, D. A. Fielder, R. Greenhalgh and J. D. Miller, Can. J. Chem., 1992, 70, 1308–1316 CrossRef CAS.
- W. Langseth, M. Ghebremeskel, B. Kosiak, P. Kolsaker and D. Miller, Mycopathologia, 2001, 152, 23–34 CrossRef CAS PubMed.
- M. Ghebremeskel and W. Langseth, Mycopathologia, 2001, 152, 103–108 CrossRef CAS PubMed.
- S. Uhlig, G. Eriksen, I. Hofgaard, R. Krska, E. Beltrán and M. Sulyok, Toxins, 2013, 5, 1682 CrossRef PubMed.
- E. Streit, C. Schwab, M. Sulyok, K. Naehrer, R. Krska and G. Schatzmayr, Toxins, 2013, 5, 504 CrossRef CAS PubMed.
- W. A. Abia, B. Warth, M. Sulyok, R. Krska, A. N. Tchana, P. B. Njobeh, M. F. Dutton and P. F. Moundipa, Food Control, 2013, 31, 438–453 CrossRef CAS.
- D. B. Prelusky, R. M. G. Hamilton and H. L. Trenholm, Microbiol., Aliments, Nutr., 1989, 7, 57–65 CAS.
- P. F. Dowd, J. D. Miller and R. Greenhalgh, Mycologia, 1989, 81, 646–650 CrossRef CAS.
- R. G. Rotter, H. L. Trenholm, D. B. Prelusky, K. E. Hartin, B. K. Thompson and J. D. Miller, Can. J. Anim. Sci., 1992, 72, 107–116 CrossRef CAS.
- E. Bresso, V. Leroux, M. Urban, K. E. Hammond-Kosack, B. Maigret and N. F. Martins, J. Mol. Model., 2016, 22, 163 CrossRef CAS PubMed.
- A. Bahadoor, D. Schneiderman, L. Gemmill, W. Bosnich, B. Blackwell, J. E. Melanson, G. McRae and L. J. Harris, J. Nat. Prod., 2016, 79, 81–88 CrossRef CAS PubMed.
- S. P. McCormick, N. J. Alexander and L. J. Harris, Appl. Environ. Microbiol., 2010, 76, 136–141 CrossRef CAS PubMed.
- L. J. Harris, M. Balcerzak, A. Johnston, D. Schneiderman and T. Ouellet, Fungal Biol., 2016, 120, 111–123 CrossRef CAS PubMed.
- C. M. Sieber, W. Lee, P. Wong, M. Munsterkotter, H. W. Mewes, C. Schmeitzl, E. Varga, F. Berthiller, G. Adam and U. Guldener, PLoS One, 2014, 9, e110311 Search PubMed.
- G. Adam, G. Wiesenberger and U. Gueldener, Biosynthesis and Molecular Genetics of Fungal Secondary Metabolites, Springer-Verlag, New York, 2015, vol. 2 Search PubMed.
- F. Berthiller, C. Crews, C. Dall'Asta, S. D. Saeger, G. Haesaert, P. Karlovsky, I. P. Oswald, W. Seefelder, G. Speijers and J. Stroka, Mol. Nutr. Food Res., 2013, 57, 165–186 CAS.
- A. Brodehl, A. Moller, H. J. Kunte, M. Koch and R. Maul, FEMS Microbiol. Lett., 2014, 359, 124–130 CrossRef CAS PubMed.
- M. J. Kelman, J. B. Renaud, K. A. Seifert, J. Mack, K. Sivagnanam, K. K. Yeung and M. W. Sumarah, Rapid Commun. Mass Spectrom., 2015, 29, 1805–1810 CrossRef CAS PubMed.
- C. Schmeitzl, B. Warth, P. Fruhmann, H. Michlmayr, A. Malachová, F. Berthiller, R. Schuhmacher, R. Krska and G. Adam, Toxins, 2015, 7, 3112 CrossRef CAS PubMed.
- B. Warth, P. Fruhmann, G. Wiesenberger, B. Kluger, B. Sarkanj, M. Lemmens, C. Hametner, J. Fröhlich, G. Adam, R. Krska and R. Schuhmacher, Anal. Bioanal. Chem., 2015, 407, 1033–1039 CrossRef CAS PubMed.
- M. Ihara, Chem. Pharm. Bull., 2006, 54, 765–774 CrossRef CAS PubMed.
- B. W. Roberts, M. S. Poonian and S. C. Welch, J. Am. Chem. Soc., 1969, 91, 3400–3401 CrossRef CAS.
- K. Takasu, S. Mizutani, M. Noguchi, K. Makita and M. Ihara, Org. Lett., 1999, 1, 391–394 CrossRef CAS.
- K. Takasu, S. Mizutani, M. Noguchi, K. Makita and M. Ihara, J. Org. Chem., 2000, 65, 4112–4119 CrossRef CAS PubMed.
- E. Varga, G. Wiesenberger, C. Hametner, T. J. Ward, Y. Dong, D. Schofbeck, S. McCormick, K. Broz, R. Stuckler, R. Schuhmacher, R. Krska, H. C. Kistler, F. Berthiller and G. Adam, Environ. Microbiol., 2015, 17, 2588–2600 CrossRef CAS PubMed.
- M. Thomas, J.-P. Gesson and S. Papot, J. Org. Chem., 2007, 72, 4262–4264 CrossRef CAS PubMed.
- B. Yu and H. Tao, J. Org. Chem., 2002, 67, 9099–9102 CrossRef CAS PubMed.
- A. Bérces, D. M. Whitfield, T. Nukada, I. do Santos, Z. A. Obuchowska and J. J. Krepinsky, Can. J. Chem., 2004, 82, 1157–1171 CrossRef.
- T. Nukada, A. Berces, M. Z. Zgierski and D. M. Whitfield, J. Am. Chem. Soc., 1998, 120, 13291–13295 CrossRef CAS.
- J. Herzig, A. Nudelman, H. E. Gottlieb and B. Fischer, J. Org. Chem., 1986, 51, 727–730 CrossRef CAS.
- J. Weber, manuscript in preparation, 2017.
- T. Mahmud, I. Tornus, E. Egelkrout, E. Wolf, C. Uy, H. G. Floss and S. Lee, J. Am. Chem. Soc., 1999, 121, 6973–6983 CrossRef CAS.
- H. Schwartz-Zimmermann, P. Fruhmann, S. Dänicke, G. Wiesenberger, S. Caha, J. Weber and F. Berthiller, Toxins, 2015, 7, 4706 CrossRef CAS PubMed.
- B. Warth, G. Del Favero, G. Wiesenberger, H. Puntscher, L. Woelflingseder, P. Fruhmann, B. Sarkanj, R. Krska, R. Schuhmacher, G. Adam and D. Marko, Sci. Rep., 2016, 6, 33854 CrossRef CAS PubMed.
- J. Meng-Reiterer, C. Bueschl, J. Rechthaler, F. Berthiller, M. Lemmens and R. Schuhmacher, Toxins, 2016, 8(12), E364 CrossRef PubMed.
- A. V. Nathanail, E. Varga, J. Meng-Reiterer, C. Bueschl, H. Michlmayr, A. Malachova, P. Fruhmann, M. Jestoi, K. Peltonen, G. Adam, M. Lemmens, R. Schuhmacher and F. Berthiller, J. Agric. Food Chem., 2015, 63, 7862–7872 CrossRef CAS PubMed.
- M. Kimura, I. Kaneko, M. Komiyama, A. Takatsuki, H. Koshino, K. Yoneyama and I. Yamaguchi, J. Biol. Chem., 1998, 273, 1654–1661 CrossRef CAS PubMed.
- F. Berthiller, U. Werner, M. Sulyok, R. Krska, M. T. Hauser and R. Schuhmacher, Food Addit. Contam., 2006, 23, 1194–1200 CrossRef CAS PubMed.
- G. R. Engelhardt, M. Ruhland and P.-R. Wallnoefer, Metabolism of mycotoxins in plants, Parlar Scientific Publications, Freising, Allemagne, 1999 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Synthetic procedures, NMR data of all compounds and details about the plant experiments. See DOI: 10.1039/c7ob02460f |
| This journal is © The Royal Society of Chemistry 2018 |