Synthesis, characterization and determination of absolute structures of palladium complexes of novel chiral acyclic tellurated Schiff base ligands†
Abstract
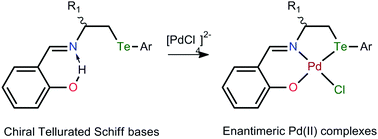
Enantiomerically pure chiral hybrid organotellurium ligands (R)-L1H, (S)-L2H and (R)-L3H have been synthesized via condensation of the chiral β-organotellurated amines 2a, 2b and 2c, respectively, with salicylaldehyde. When these chiral acyclic tellurated Schiff base ligands reacted with Na2PdCl4 in a 1 : 1 molar ratio, they afforded the corresponding orange-red asymmetric complexes [Pd(L1–3)Cl] (1–3). All these compounds were characterized by elemental analysis, polarimetry, and 1H, 13C{1H} NMR and FT-IR spectroscopies. The absolute structures of the complexes 1 and 2 were determined by single crystal X-ray diffraction, and the complexes 1 and 2 crystallized in the hexagonal and orthorhombic crystal systems with the space groups P65 and P212121, respectively. The characterisation data showed that the ligands coordinated in the deprotonated tridentate (Te, N, O−) mode and acquired a square planar geometry around the central palladium(II) ion along with a monodentate chloro (Cl−) in 1–3. The Pd–Te, Pd–Cl, Pd–O and Pd–N bond lengths found were 2.5008(9), 2.3286(19), 2.037(6) and 2.009(6) Å, respectively, in 1 and 2.497(2)/2.506(3), 2.293(6)/2.326(6), 2.035(15)/2.030(2) and 1.949(17)/2.010(2) Å, respectively, in 2. There was intermolecular CH⋯Cl [2.81–2.96 Å] and CH⋯O [2.40–2.64 Å] hydrogen bonding in 1 and 2, which resulted in supramolecular structures. The Te⋯Cl [3.309–3.710 Å] secondary interactions resulted in the helical structure of 1 and dimeric structure of 2 with a short Pd⋯Pd [3.500(1) Å] distance. The Flack parameters (x) of 1 [0.02(3)] and 2 [0.024(12)] confirmed their absolute configurations.


 Please wait while we load your content...
Please wait while we load your content...