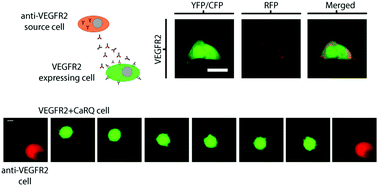
As monoclonal antibodies have two epitopes for their target ligand, they should theoretically dimerize target receptors upon binding. In particular, the dimerization of the vascular endothelial growth factor (VEGF) receptor 2 (VEGFR2) stimulates early events occurring within minutes (e.g. Ca2+ signal generation) and late events occurring over hours and days (e.g. cell migration in angiogenesis). Although studies have noted that antibodies targeting VEGFR2 (anti-VEGFR2) inhibited cell migration in angiogenesis, we show in this paper that an anti-VEGFR2 stimulus nevertheless triggered a Ca2+ signal in VEGFR2 expressing cells. This Ca2+ signal was then re-wired to promote cell migration by co-expressing an engineered Ca2+ activated RhoA (called CaRQ), thereby engineering the opposite anticipated effect of an anti-VEGFR2 antibody. In these cells, the anti-VEGFR2 antibody stimulus induced cellular blebbing, migration across a membrane, and in vitro scratch wound healing. This work expands the utility of monoclonal antibodies to induce tailored responses in engineered cells such as changes in cell fluorescence via Ca2+ reporters or migration patterns via CaRQ.