Metamorphosis of cycloalkenes for the divergent total synthesis of polycyclic indole alkaloids
Abstract
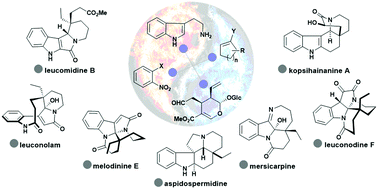
From tryptamine and secologanine, nature generates monoterpene indole alkaloids with an unprecedented level of skeletal diversity through a ‘couple–divert’ sequence. Intrigued by this biosynthetic machinery, new strategies and tactics to access skeletally distinct natural products are continuously emerging. In this Tutorial Review, we’ll present a simplified view of nature's logic for biosynthesis and the representative strategies and tactics developed for the divergent total synthesis of monoterpene indole alkaloids. Our group has been developing a ‘couple–divert’ approach with the strategic use of cycloalkene as a pluripotent motif in the synthetic design and has developed an integrated oxidation/reduction/cyclization (iORC) sequence for transforming functionalized cycloalkenes, easily available by a variety of cross-coupling reactions, to skeletally diverse natural products. The integration of controlled regio-, chemo- and stereo-selective cyclization and heteroannulation reactions into these domino sequences will be highlighted.
- This article is part of the themed collection: New Directions in Natural Product Synthesis


 Please wait while we load your content...
Please wait while we load your content...