NaHS·nH2O-induced umpolung: the synthesis of 2-acyl-3-aminoindoles from aryl methyl ketones and 2-aminobenzonitriles†
Abstract
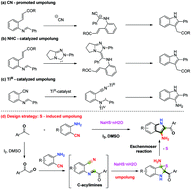
An efficient method for constructing 2-acyl-3-aminoindoles from methyl ketones and 2-aminobenzonitriles is described, in which NaHS·nH2O is used as a novel umpolung reagent for the first time in organic synthesis. Mechanistic studies revealed that the key step involved an Eschenmoser sulfide contraction reaction.


 Please wait while we load your content...
Please wait while we load your content...