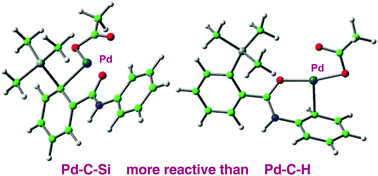
Silyl-substituted aromatic compounds can participate as the electrophilic component in palladium-catalysed cross-couplings, and reactivity is enhanced by a neighbouring silyl-group. Products analogous to those obtained from C–H activation chemistry are accessible by this means with the additional benefit of regiochemistry defined by the site of silyl substitution. DFT studies described here show that the mechanism of C–Si cleavage is distinct from previously recognised mechanisms for C–H cleavage, with a cascade of silyl intermediates en route to a stable product. The amide directing-groups are involved only in the stabilisation of palladacyclic intermediates, and are never disposed to activate silicon directly. 5-Membered and 6-membered palladacycles are known to behave differently in coupling reactions and the calculations reveal underlying reasons in the cationic pathways studied here.