Effect of including the carbon nanotube and graphene oxide on the electrocatalytic behavior of the Ni–W alloy for the hydrogen evolution reaction†
Abstract
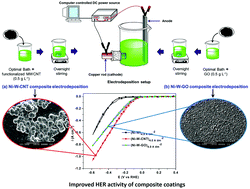
The present work reports the electrocatalytic activities of the composite coatings of Ni–W developed using the carbon nanotube (CNT) and graphene oxide (GO). Ni–W–CNT and Ni–W–GO coatings were developed by exploiting the advantages of the composite electrodeposition technique. The effect of CNT and GO on the induced codeposition behavior of the reluctant metal W and the relationship with their electrocatalytic efficiency was studied. The electrocatalytic alkaline water splitting efficiency for the hydrogen evolution reaction (HER) of each of the electrode materials was tested by using cyclic voltammetry (CV) and chronopotentiometry (CP) techniques. Furthermore, the practical utility of each of the electrode materials was evaluated by measuring the amount of H2 gas evolved. The variation in electrocatalytic activity with composition, structure, and morphology of the coatings was examined systematically using XRD, SEM, and EDS analyses. The Ni–W–CNT and Ni–W–GO composite coatings yielded much better electrocatalytic activities for the HER than did the as-coated Ni–W alloy. The obtained results showed Ni–W–CNT composite coating as the best electrode material for alkaline HER, attributed by both increased W content and number of electroactive centres. Moreover, the number of electroactive centres was found to be affected by the homogeneous distribution of CNT in the alloy matrix.


 Please wait while we load your content...
Please wait while we load your content...