Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Salan group 13 complexes – structural study and lactide polymerisation†
James
Beament
,
Mary F.
Mahon
,
Antoine
Buchard
 * and
Matthew D.
Jones
* and
Matthew D.
Jones
 *
*
Department of Chemistry, University of Bath, Claverton Down, Bath BA2 7AY, UK. E-mail: mj205@bath.ac.uk; a.buchard@bath.ac.uk; Fax: +44 (0)1225 386231; Tel: +44 (0)1225 384908
First published on 8th February 2017
Abstract
Herein the preparation and characterisation of a series of group 13 salan complexes, with a bipyrrolidine or N,N′-ethylenediamine backbone are disclosed. For the bipyrrolidine derived-salan ligand 1H2, the Al(III) and Ga(III) complexes are pseudo trigonal bipyramidal in the solid-state, whereas the In(III) complexes are best described as square based pyramidal structures. However, for the ethylenediamine derived-salan ligand 2H2, all complexes are effectively square based pyramidal in their structure. The complexes' solution behaviour is also investigated by NMR spectroscopic methods and it is observed that the solid-state structure is maintained in solution. The complexes have all been trialled for the ring opening polymerisation of rac-lactide. With In(1)Cl controlled polymerisation and narrow molecular weight distributions (1.01–1.08) are observed with heterotactic polylactide being prepared. Under the conditions tested the Ga(III) and Al(III) complexes were shown to be inactive.
Introduction
In recent years there has been an explosion of research concerning new initiators for the controlled ring opening polymerisation (ROP) of lactide (LA) affording polylactide (PLA).1 The resultant PLA is compostable and annually renewable from starch-rich plant based materials. This material has the potential to replace traditional petrochemical derived plastics for numerous applications, from high value biomedical areas to commodity uses.2 While organocatalysts and initiators commonly yield atactic PLA from rac-LA,3 there are many metal centres that are active for the stereoselective polymerisation of rac-LA to afford either heterotactic or isotactic stereoblock PLA. Examples of such metal centres include groups 1–4,4 lanthanides, Zn(II)4f,5 and pertinent to this study group 13 metals.6 There are many ligands that have found utility in this area such as salan,4a,b,6b,c salen,6e–h,7 salalen8 and variations thereof, including phosphasalen.9 In the case of indium the area is dominated by salen ligands.10 However, other ligand systems are known.11 InCl3 alone was first shown to be an active catalyst by Tolman and Hillmyer in 2009, with high heterotacticity being favoured in THF solution, when used in combination with triethylamine and benzylalcohol.12 Recently, extensive studies have been conducted by Mehrkhodavandi and co-workers who have elegantly illustrated the advantages of In(III) complexes for the controlled ROP of rac-LA.7,13 These have typically utilised salen ligands with chirality in the backbone. For example, when indium is complexed with Jacobsen's ligand the resultant complex was seen to be faster than the corresponding Al(III) system, with isotactic (Pm = 0.77) PLA being realised.13a In these studies aggregation of the indium complexes, due to the large ionic radii of In(III),13i has be shown to be troublesome, and was controlled by the use of bulky ligands. Ga(III) initiated ROP of lactide is rare with only a handful of examples reported in the literature.10,14 Further, complexes of In-“salan-like” ligands characterised in the solid-state are extremely rare.15 Thus, it is pertinent to study the coordination chemistry of simple salan related ligands with In(III) (to compare with Al/Ga) and screen for the ROP of rac-LA.New catalyst research in the area is driven by the need to develop structure–activity-relationships and understand the complex interplay between the ligand and the metal centre, such relationships are lacking in the area. It has been shown that very subtle changes to the ligand or switching the metal centre can have dramatic consequences to the polymerisation. For example, we have shown with a bipyrrolidine derived salan ligand there is a switch in selectivity from highly isotactic PLA Zr(IV)/Hf(IV) to highly heterotactic PLA for Al(III).4a Williams and co-workers have observed similar switches with phosphasalen complexes of different lanthanides.16 Further switching in control are noted by comparing Al-(SalBinap) (isotactic) to Y(SalBinap) (heterotactic) complexes of Coates and Pappalardo respectively.17 Kol first introduced the bipyrrolidine ligand in 2009 to pre-determine the chirality at a group 4 metal centre.18 We have previously illustrated that ligands based on the bipyrrolidine backbone are highly effective for the ROP of lactide.4a,b More recently, Kol has also utilised this backbone with Mg(II) and to some extent Al(III), highlighting the importance of these ligands in the literature.19 We have shown the importance of the meso chirality, in the controlled and selective polymerisation of lactide.4a,b Thus, in this paper we expand upon the chemistry of this system and report the full characterisation of a series of group 13 complexes with our bulky salan derived meso-bipyrrolidine ligand and an N,N′-ethylenediamine backbone for comparison. To the best of our knowledge this is also the first reported application of a monomeric In–salan complex for the ROP of rac-LA.
Experimental
General considerations
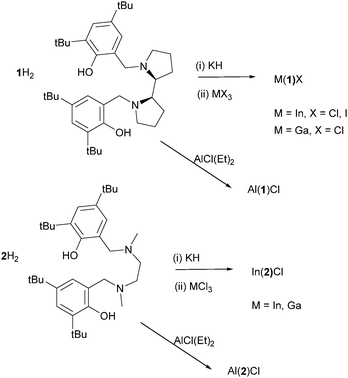
The preparation and characterisation of all metal complexes was carried out under an inert argon atmosphere using standard Schlenk or glovebox techniques. All chemicals used were purchased from Sigma-Aldrich and used as received except for rac-LA which was recrystallized twice from dry toluene before use. Dry solvents used in handling metal complexes were obtained via SPS (solvent purification system). 1H and 13C{1H}, NMR spectra were recorded on a Bruker 400 or 500 MHz instrument and referenced to residual solvent peaks. CDCl3 was dried over CaH2 prior to use. Coupling constants are given in Hertz. CHN microanalysis was performed by Mr Stephen Boyer of London Metropolitan University. The ligands were prepared as detailed in the literature.4a,20Synthesis of complexes
In a typical experiment: ligand (1H2) (2.6 mmol) dissolved in THF (10 mL) was added dropwise to a stirred suspension of potassium hydride (5.2 mmol) in THF (10 mL) and stirred for 16 hours. The solution was then cooled to −78 °C and a solution of InCl3 (2.6 mmol) in anhydrous THF (10 mL) was added dropwise and after complete addition, the solution was left to stir for a further 2 hours. After this time, the solvent was removed and the white powder was dissolved in dry chloroform and filtered through a pad of celite under Ar. The supernatant was collected and removal of solvent yielded the crude product, which was then recrystallised in dry hexane/toluene. In(1)Cl yield: (1.61 g, 2.22 mmol, 85%). 1H-NMR (400 MHz, CDCl3, δH, ppm); 7.30 (s, 2H; ArH), 6.76 (s, 2H; ArH), 4.73 (d, 12.7 Hz, 2H, NCHHC), 3.41 (d, 2H, 2.5 Hz; NCH), 3.34 (d, 2H, 12.7 Hz; NCHHC), 3.10 (m, 2H; CH2N), 3.00 (m, 2H; CH2N), 2.31 (m, 2H; CHH), 2.03 (m, 4H; CH2), 1.90 (m, 2H; CHH), 1.50 (s, 18H; C(CH3)3) 1.28 (s, 18H; C(CH3)3); 13C{1H} NMR (100 MHz, CDCl3, δC, ppm); 159.5 (C–O), 138.5 (Ar), 136.2 (Ar), 123.9 (C–H, Ar), 123.8 (C–H, Ar), 118.8 (Ar), 66.1 (NCH), 61.6 (NCH2), 52.4 (NCH2), 34.2 (C(CH3)3), 32.9 (C(CH3)3), 30.8 (C(CH3)3), 28.7 (C(CH3)3), 25.3 (CH2), 20.1 (CH2). Elemental analysis (C38H58N2O2In1Cl1), (calculated) C: 62.94%, H: 8.06%, N: 3.86% (experimental) C: 63.01%, H: 8.08%, N: 3.86%. Al(1)Cl (0.6 g, 0.94 mmol, 55%). 1H-NMR (400 MHz, CDCl3, δH, ppm); 7.23 (s, 2H; ArH), 6.73 (s, 2H; ArH), 4.45 (m, 2H; NCHHC), 3.32 (m, 6H; NCH), 2.84 (m, 2H; NCHHC), 2.17 (m, 2H; CH2N), 1.98 (m, 2H; CH2N), 1.86 (m, 2H; CHH), 1.42 (s, 18H; C(CH3)3), 1.24 (s, 18H; C(CH3)3); 13C{1H} NMR (100 MHz, CDCl3, δC, ppm); 156.2 (C–O), 138.4 (Ar), 138.1 (Ar), 124.4 (C–H, Ar), 123.2 (C–H, Ar), 120.4 (Ar), 67.8 (NCH), 62.3 (NCH2), 54.5 (NCH2), 35.3 (C(CH3)3), 34.3 (C(CH3)3), 32.1 (C(CH3)3), 30.1 (C(CH3)3), 27.0 (CH2), 21.8 (CH2). Elemental analysis (CHN), (calculated) C: 71.61%, H: 9.17%, N: 4.39% (experimental) C: 69.17%, H: 9.12%, N: 4.31%.Ga(1)Cl (0.93 g, 1.37 mmol, 53%). 1H-NMR (400 MHz, CDCl3, δH, ppm); 7.28 (s, 2H, Ar), 6.77 (s, 2H, Ar), 4.66 (d, 2H, 12.2 Hz, NCHHC), 3.75 (d, 2H, 2.5 Hz, NCH), 3.36 (d, 2H, 12.2 Hz, NCHHC), 2.92 (4H, m, CH2N), 2.27 (m, 2H, CHH), 2.03 (m, 4H, CH2), 1.90 (m, 2H, CHH), 1.48 (s, 18H, tBu), 1.28 (s, 18H, tBu); 13C{1H} (100 MHz, CDCl3, δC, ppm); 158.5 (C–O), 139.2 (Ar), 137.9 (Ar), 124.7 (C–H, Ar), 123.8 (C–H, Ar), 119.9 (Ar), 68.3 (NCH), 67.2 (NCH), 62.6 (NCH2), 54.2 (NCH2), 35.5 (C(CH3)3), 34.4 (C(CH3)3), 32.2 (C(CH3)3), 30.2 (C(CH3)3), 26.9 (CH2), 26.0 (CH2), 21.5 (CH2). Elemental analysis (CHN), (calculated) C: 67.11%, H: 8.60%, N: 4.12% (experimental) C: 66.82%, H: 8.56%, N: 4.64%. In(1)I (345 mg, 0.42 mmol, 42%). 1H-NMR (400 MHz, CDCl3, 253 K, δH, ppm); 7.29 (s, 2H; ArH), 6.76 (s, 2H; ArH), 4.76 (d, 12.7 Hz, 2H; NCHHC), 3.53 (d, 2.5 Hz, 2H; NCH), 3.34 (d, 12.7 Hz, 2H; NCHHC), 3.10 (m, 2H; CH2N), 2.97 (m, 2H; CH2N), 2.31 (m, 2H; CHH), 1.99 (m, 4H; CH2), 1.90 (m, 2H; CHH), 1.50 (s, 18H, (C(CH3)3)), 1.28 (s, 18H, (C(CH3)3)). 13C-NMR (100 MHz, CDCl3, 298 K, δC, ppm); 139.49 (Ar), 125.2 (Ar), 67.5 (NCH), 54.16 (NCH2), 52.4 (NCH2), 35.3 (C(CH3)3), 34.3 (C(CH3)3), 32.1 (C(CH3)3), 30.4 (C(CH3)3), 27.0 (CH2), 20.9 (CH2). Elemental analysis (CHN), (calculated) C: 55.89%, H: 7.16%, N: 3.43% (experimental) C: 56.05%, H: 7.19%, N: 3.42%. Al(2)Cl (680 mg, 1.16 mmol, 61%). 1H-NMR (400 MHz, CDCl3, δH, ppm): (major series) 7.30 (s, 2H, Ar), 6.81 (s, 2H, Ar), 4.52–4.49 (d, 12.4 Hz, 2H, CHHN), 3.20 (d, 12.4 Hz, 2H, CHHN), 2.98 (m, 4H, NCH2), 2.44 (s, 6H, NCH3), 1.47 (s, 18H, tBu), 1.29 (s, 18H, tBu), due to significant overlap in tBu, Ar region full assignment of minor series is not possible. But it is clear from the NMR below. 13C-NMR (100 MHz, CDCl3, δC, ppm): 155.7 (C–O), 138.6 (Ar), 138.4 (Ar), 124.5 (Ar), 123.3 (Ar), 119.7 (Ar), 63.0 (CH2N), 55.2 (CH2N), 45.6 (NCH3), 35.5 (C(CH3)3), 34.2 (C(CH3)3), 32.0 (C(CH3)3), 30.1 (C(CH3)3). Elemental analysis (CHN): (calculated) C: 69.77%, H: 9.30%, N: 4.79% (experimental) C: 67.57%, H: 9.50%, N: 4.75%. Ga(2)Cl (565 mg, 0.9 mmol, 48%). 1H-NMR (400 MHz, CDCl3, δH, ppm): 7.31 (s, 2H, Ar), 6.80 (s, 2H, Ar), 4.71–4.68 (d, 12.1 Hz, 2H, CHHN), 3.25–3.22 (d, 12.1 Hz, 2H, CHHN), 3.16 (m, 2H, CH2N), 2.95 (m, 2H, CH2N), 2.42 (s, 6H, CH3), 1.51 (s, 18H, tBu), 1.28 (s, 18H, tBu); 13C-NMR (100 MHz, CDCl3, δC, ppm): 158.0 (C–O), 139.3 (Ar), 138.6 (Ar), 124.8 (Ar), 124.1 (Ar), 199.8 (Ar), 63.4 (CH2N), 55.1 (CH2N), 45.0 (NCH3), 35.5 (C(CH3)3), 34.2 (C(CH3)3), 32.0 (C(CH3)3), 30.2 (C(CH3)3). Elemental analysis (CHN): (calculated) C: 65.03%, H: 8.67%, N: 4.46% (experimental) C: 64.92%, H: 8.80%, N: 4.57%. In(2)Cl (790 mg, 1.18 mmol, 42%). 1H-NMR (400 MHz, CDCl3, δH, ppm): 7.31 (d, 2.5 Hz, 2H, Ar), 6.78 (d, 2.5 Hz, 2H, Ar), 4.81 (d, 11.8 Hz, 2H, CHHN), 3.20 (d, 11.83 Hz, 4H, CHHN), 2.93 (m, 2H, NCHHC), 2.40 (s, 6H, NCH3), 1.53 (s, 18H, tBu), 1.28 (s, 18H, tBu); 13C-NMR (100 MHz, CDCl3, δC, ppm): 160.5 (C–O), 139.8 (Ar), 138.3 (Ar), 129.3 (Ar), 128.4 (Ar), 64.3 (CH2N), 55.4 (CH2N), 43.8 (NCH3), 35.2 (C(CH3)3), 33.9 (C(CH3)3), 31.7 (C(CH3)3), 29.9 (C(CH3)3). Elemental analysis (CHN), (calculated) C: 60.67%, H: 8.09%, N: 4.16%, (experimental) C: 60.71%, H: 8.14%, N: 4.16%.
Crystallography
All data were collected, at 150 K, on a Rigaku SuperNova diffractometer using radiation CuKα (λ = 1.54184 Å). Structures were solved by direct methods throughout and refined on F2 data using the SHELXL-2014 suite of programs. All hydrogen atoms were included in idealised positions and refined using the riding model. Refinements was straightforward with only the following points that merit note: in Al(1)Cl one methyl group was disordered over two positions in a 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 ratio; In2(1)2(OH)(OEt) contains 1.5 molecules of solvent in the asymmetric unit and one tBu group is disordered over two positions in a 50
45 ratio; In2(1)2(OH)(OEt) contains 1.5 molecules of solvent in the asymmetric unit and one tBu group is disordered over two positions in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio; In(2)Cl contains two molecules of toluene in the asymmetric unit. One tBu group in Al(2)Cl was disordered over two sites in a 75
50 ratio; In(2)Cl contains two molecules of toluene in the asymmetric unit. One tBu group in Al(2)Cl was disordered over two sites in a 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 ratio and a partial occupancy hexane molecule (75%) was observed in the asymmetric unit. Two of the tBu moieties in Ga(2)Cl were disordered over two sites in ratios of 80
25 ratio and a partial occupancy hexane molecule (75%) was observed in the asymmetric unit. Two of the tBu moieties in Ga(2)Cl were disordered over two sites in ratios of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 and 60
20 and 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 ratio, respectively. Residual solvent in this structure {Ga(2)Cl} bore a strong resemblance to a molecule of hexane (the recrystallisation solvent) but the electron density was smeared in a manner that suggested that disorder, above and beyond this smearing, was also prevalent. Thus, an approach involving a very restrained solvent model was abandoned in favour of employing PLATON SQUEEZE to address the guest solvent in Ga(2)Cl and an allowance for one molecule of hexane in the motif has been made in the formula presented herein. Further for Ga(2)Cl residual electron density in this structure is in the region of the ligand based on O1 and N1. Attempts were made to model this, but at a site occupancy of approximately 10% for a potential minor component (which seemed to be the cis isomer), the merit of disorder modelling was questionable, and hence abandoned. Interestingly, the raw frames of data suggested some crystal twinning. However, the data from integration of the data as a twin were not ultimately used, as the scale factor for the second twin component refined to approximately 5%, with no tangible advantage to the overall convergence of the model.
40 ratio, respectively. Residual solvent in this structure {Ga(2)Cl} bore a strong resemblance to a molecule of hexane (the recrystallisation solvent) but the electron density was smeared in a manner that suggested that disorder, above and beyond this smearing, was also prevalent. Thus, an approach involving a very restrained solvent model was abandoned in favour of employing PLATON SQUEEZE to address the guest solvent in Ga(2)Cl and an allowance for one molecule of hexane in the motif has been made in the formula presented herein. Further for Ga(2)Cl residual electron density in this structure is in the region of the ligand based on O1 and N1. Attempts were made to model this, but at a site occupancy of approximately 10% for a potential minor component (which seemed to be the cis isomer), the merit of disorder modelling was questionable, and hence abandoned. Interestingly, the raw frames of data suggested some crystal twinning. However, the data from integration of the data as a twin were not ultimately used, as the scale factor for the second twin component refined to approximately 5%, with no tangible advantage to the overall convergence of the model.
Ring-opening polymerisation (ROP) studies
For polymerisations, under an argon atmosphere, the required monomer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalyst
catalyst![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3
NEt3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BnOH ratio was suspended in toluene (10 mL) then heated up to 80 °C (upon which the mixture solubilised) and stirred. In all cases 1.0 g of rac-LA was used. After the reaction time the vessel was opened to air and methanol (1–2 drops) was added to quench the reaction and the resulting solid was dissolved in dichloromethane. The solvents were removed in vacuo, an NMR spectrum was recorded to establish conversion. The crude mixture was washed with copious amounts of methanol to remove unreacted monomer to afford the pure polymer. 1H NMR spectroscopy (CDCl3) and GPC (THF) were used to determine tacticity and molecular weights (Mn and Mw) of the polymers produced; Pr (the probability of racemic linkages) values were determined by analysis of the methine region of the homonuclear decoupled 1H NMR spectra.4f GPC were recorded on an Agilent instrument using triple detection and the multi analysis software. Polymer end-group analysis was carried out using MALDI-ToF spectrometry on a Bruker Autoflex speed instrument using DCTB (trans-2-[3-(4-tert-butylphenyl)-2-methyl-2-propenylidene]malononitrile) as the matrix and ionised using NaOAc.
BnOH ratio was suspended in toluene (10 mL) then heated up to 80 °C (upon which the mixture solubilised) and stirred. In all cases 1.0 g of rac-LA was used. After the reaction time the vessel was opened to air and methanol (1–2 drops) was added to quench the reaction and the resulting solid was dissolved in dichloromethane. The solvents were removed in vacuo, an NMR spectrum was recorded to establish conversion. The crude mixture was washed with copious amounts of methanol to remove unreacted monomer to afford the pure polymer. 1H NMR spectroscopy (CDCl3) and GPC (THF) were used to determine tacticity and molecular weights (Mn and Mw) of the polymers produced; Pr (the probability of racemic linkages) values were determined by analysis of the methine region of the homonuclear decoupled 1H NMR spectra.4f GPC were recorded on an Agilent instrument using triple detection and the multi analysis software. Polymer end-group analysis was carried out using MALDI-ToF spectrometry on a Bruker Autoflex speed instrument using DCTB (trans-2-[3-(4-tert-butylphenyl)-2-methyl-2-propenylidene]malononitrile) as the matrix and ionised using NaOAc.
Results and discussion
Complexes
The complexes were prepared as shown in Scheme 1. They were isolated as colourless crystals in high purity, as confirmed by elemental analysis.The Ga(III) and In(III) complexes (Scheme 1 and Fig. 1) were prepared via first synthesising the potassium salt of the ligand and subsequent reaction with the metal–halide. Attempts to prepare the potassium salt of the ligand with Me groups instead of tBu on the aromatic ring were unsuccessful and an insoluble material was isolated, thus synthetic efforts were concentrated on 1/2H2. The Al(III) complex could more readily be synthesised utilising AlCl(Et)2 as the precursor. All complexes were recrystallised in hexane/toluene mixture, see Tables 1 and 2 for selected metric data. To the best of our knowledge these are the first examples of either a salan–In–Cl, salan–In–I or salan–Ga–Cl complex characterised in the solid-state. For In(1)Cl/I the metal centres are seen to be in a pseudo square based pyramidal (sqp) geometry, which is exemplified by τ = 0.32 {for In(1)I this is 0.28}. The τ value is used as an aid regarding the preference of the complex to form either a sqp structure or trigonal bipyramidal (tbp) one.21 This is analogous to the In–Cl complexes of Jacobsen's ligand previously discussed.13a For this literature complex the In–N bond lengths are 2.171(7) and 2.207(7) which are within 3 esds and can therefore be considered equivalent. However, for both In(1)Cl/I and there is a significant difference between the In–N bond lengths e.g. In(1)Cl 2.2684(11) vs. 2.3539(11) Å. For Ga(1)Cl τ = 0.76 indicative of a distorted trigonal bipyramidal geometry at the metal centre, this is also the case for Al(1)Cl with τ = 0.68. Previously, we reported the complex Al(1)(OiPr), which also afforded τ = 0.68.4a This tbp geometry is further exemplified by analysis of the angles between the two planes formed by the C6-aromatic rings, Al(1)Cl = 133°, Ga(1)Cl = 134° whereas In(1)Cl/I = 161/158°. As a structural comparison Al/Ga/In(2)Cl were prepared. The solid state structure of In(2)Cl is analogous to In(1)Cl, with τ = 0.38 and similar metric data. However, there is a difference for Al/Ga(2)Cl with τ = 0.26/0.29 respectively indicating a preference for a square based pyramidal geometry in those cases. Interestingly, the N–Me groups are effectively cis to each other in In(2)Cl, whereas in the solid-state structures for Al/Ga(2)Cl these groups are trans.
 | ||
| Fig. 1 Solid-state structure for In(1)Cl, Ga(1)Cl and In(2)Cl. Ellipsoids are shown at the 30% probability level and all hydrogen atoms have been removed for clarity. | ||
| Al(1)Cl | Ga(1)Cl | In(1)Cl | In(1)I | |
|---|---|---|---|---|
| M–X | 2.1802(6) | 2.2043(4) | 2.3683(4) | 2.69445(19) |
| M–O(1) | 1.7820(11) | 1.8436(10) | 2.0768(9) | 2.0536(14) |
| M–O(2) | 1.7540(11) | 1.8828(10) | 2.0574(10) | 2.0907(14) |
| M–N(1) | 2.0285(13) | 2.2527(11) | 2.2684(11) | 2.2723(17) |
| M–N(2) | 2.2147(13) | 2.0513(12) | 2.3539(11) | 2.3719(16) |
| N(1)–M–O(2) | 126.79(6) | 122.74(5) | 134.92(4) | 134.49(6) |
| O(1)–M–N(2) | 167.65(5) | 168.58(4) | 154.02(4) | 151.10(6) |
| N(1)–M–X | 114.99(4) | 94.37(3) | 111.62(3) | 102.49(4) |
| Al(2)Cl | Ga(2)Cl | In(2)Cl | |
|---|---|---|---|
| M–X | 2.1720(9) | 2.2073(6) | 2.3700(5) |
| M–O(1) | 1.7789(18) | 1.8603(16) | 2.0310(13) |
| M–O(2) | 1.7551(17) | 1.8411(13) | 2.0737(13) |
| M–N(1) | 2.052(2) | 2.0873(18) | 2.3544(17) |
| M–N(2) | 2.211(2) | 2.2344(18) | 2.2572(16) |
| N(1)–M–O(2) | 143.66(9) | 142.42(7) | 155.56(6) |
| O(1)–M–N(2) | 159.19(9) | 159.98(9) | 132.71(6) |
| N(1)–M–X | 103.78(6) | 104.74(5) | 97.55(4) |
In all cases {bar Al/Ga(2)Cl} the solid-state structures appear to be maintained in solution and the ligand remains “locked” once coordinated as evidenced by the formation of clear diastereotopic doublets in the 1H NMR spectrum (CDCl3, see ESI†). Furthermore, there are two singlets in the aromatic region of 1H NMR spectrum and six aromatic resonances in the 13C{1H} NMR spectrum indicating that both phenoxides are equivalent in solution. For the bipyrrolidine complexes there are four carbon –CH2– resonances and one –CH– resonance present in the NMR spectrum. For Al/Ga(2)Cl there are clearly two species present in solution in approximately a 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 ratio for Al(2)Cl and 95
25 ratio for Al(2)Cl and 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 for Ga(2)Cl. The major species has an NMR spectrum analogous to In(2)Cl with one resonance for the N–CH3 group, implying in solution the major species is most likely the cis isomer. The minor series has two N–CH3 resonances and a more complex series of doublets for the methylene protons which is expected for the trans isomer. DOSY indicated that the two species in solution have analogous diffusion constants, ca. 6.3 × 10−10 m2 s−1, which would be expected for cis/trans isomers, thus ruling out possible monomer–dimer equilibria. In(1)I showed fluxionality on the NMR time scale and it was necessary to cool the sample to 253 K to obtain sharp resonances. This may be related to tbp–sbp exchanges in solution or cis–trans exchange processes.
5 for Ga(2)Cl. The major species has an NMR spectrum analogous to In(2)Cl with one resonance for the N–CH3 group, implying in solution the major species is most likely the cis isomer. The minor series has two N–CH3 resonances and a more complex series of doublets for the methylene protons which is expected for the trans isomer. DOSY indicated that the two species in solution have analogous diffusion constants, ca. 6.3 × 10−10 m2 s−1, which would be expected for cis/trans isomers, thus ruling out possible monomer–dimer equilibria. In(1)I showed fluxionality on the NMR time scale and it was necessary to cool the sample to 253 K to obtain sharp resonances. This may be related to tbp–sbp exchanges in solution or cis–trans exchange processes.
Reaction of In(1)Cl with NaOEt, in an attempt to generate an alkoxide, led to an intractable mixture of products, as previously observed for a SalBinap–InCl complex of Mehrkhodavandi.13b However, we were able to isolate a bridged In–OH/OiPr complex (see ESI†) although this was in low yield. The coordination motif of the ligand in this octahedral complex is analogous to that observed for similar meso-ligated group 4 complexes.4a Presumably, during the polymerisation the indium centre is 6 coordinate (ligand, lactide and polymer chain) thus this illustrates the likely coordination of ligand in the catalytically active species.
Polymerisation studies
The complexes were initially trialled for the polymerisation of rac-LA in toluene at conditions of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (LA
1 (LA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) metal
metal![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BnOH
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3) at 80 °C, Table 3. These conditions are typical for In–Cl initiated polymerisation, the exact role of NEt3/BnOH is open to debate and the reader is directed to the following for pertinent mechanistic studies with InX3.12b Regardless of the exact mechanism, the polymerisation is initiated by benzyl alcohol and catalysed by the Indium complex (as confirmed by MALDI-ToF and the lack of activity observed when no In complex is present). Under these conditions and time (up to 5 days at 80 °C) only the indium complexes displayed any polymerisation activity. With the Ga/Al complexes only monomer was isolated after work up. This may well be related to the fact that the “smaller” Lewis acidic metals are screened from coordinating a molecule of lactide or the strength of the M–X bond prevents the formation of the alkoxide.
NEt3) at 80 °C, Table 3. These conditions are typical for In–Cl initiated polymerisation, the exact role of NEt3/BnOH is open to debate and the reader is directed to the following for pertinent mechanistic studies with InX3.12b Regardless of the exact mechanism, the polymerisation is initiated by benzyl alcohol and catalysed by the Indium complex (as confirmed by MALDI-ToF and the lack of activity observed when no In complex is present). Under these conditions and time (up to 5 days at 80 °C) only the indium complexes displayed any polymerisation activity. With the Ga/Al complexes only monomer was isolated after work up. This may well be related to the fact that the “smaller” Lewis acidic metals are screened from coordinating a molecule of lactide or the strength of the M–X bond prevents the formation of the alkoxide.
| Catalyst | M![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I I |
Co-catalyst | Con.a/% | Theor. Mnb | M n | Đ | P r |
|---|---|---|---|---|---|---|---|
| a Determined from analysis of the 1H NMR spectrum. b Theoretical Mn = (144 × equiv. LA) × conv./100 + end-groups (rounded to nearest 50). c As determined by GPC (THF) using triple detection methods. d As determined from 1H{1H} NMR. | |||||||
| In(1)Cl | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3 NEt3 |
95 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 650 650 |
1.01 | 0.82 |
| In(1)Cl | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH | 60 | 8750 | 9600 | 1.08 | 0.76 |
| In(1)Cl | 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3 NEt3 |
95 | 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 150 150 |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 350 350 |
1.07 | 0.80 |
| In(1)Cl | 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH | 30 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050 050 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 950 950 |
1.02 | 0.84 |
| In(1)Cl | 900![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3 NEt3 |
86 | 111![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 550 550 |
77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 850 850 |
1.07 | 0.80 |
| In(1)Cl | 900![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH | 42 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 550 550 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.08 | 0.82 |
| In(1)I | 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3 NEt3 |
48 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 850 850 |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 650 650 |
1.08 | 0.67 |
| In(2)Cl | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3 NEt3 |
95 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 450 450 |
1.10 | 0.73 |
Previous work has shown that the Al–OiPr complex of 1H2 {Al(1)(OiPr)} was active under melt conditions (75% conversion after 48 h) producing atactic PLA.4a In this present study the Al/Ga–Cl complexes were tested under melt conditions but failed to produce any polymer. However, there is a dramatic switch in selectivity and activity with In(III), which produced heterotactic PLA in 24 h in solution. Using InCl3 (with NEt3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BnOH at 100
BnOH at 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 {LA
1 {LA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In
In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BnOH
BnOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NEt3}) we achieved a conversion of 96% in toluene at 80 °C under the same time frame.12 The molecular weight of the resulting PLA was 17
NEt3}) we achieved a conversion of 96% in toluene at 80 °C under the same time frame.12 The molecular weight of the resulting PLA was 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 g mol−1 with a dispersity of 1.45 and, Pr = 0.71. The PLA produced with our complexes is more monodispersed, with increased tacticity control. Further, given the high purity and good control of molecular weight any activity caused by residual or generated InCl3 is unlikely.
800 g mol−1 with a dispersity of 1.45 and, Pr = 0.71. The PLA produced with our complexes is more monodispersed, with increased tacticity control. Further, given the high purity and good control of molecular weight any activity caused by residual or generated InCl3 is unlikely.
In all indium cases PLA with a high heterotactic was observed, as indicated by an enhancement in the isi and sis tetrads in the 1H{1H} NMR spectrum (see ESI†). It was observed that In(2)Cl was significantly less selective than In(1)Cl, thus polymerisation optimisation was performed with In(1)Cl. Without the addition of BnOH and NEt3 no polymer could be observed, however, without the addition of NEt3 polymerisation still proceeded albeit at a significantly reduced conversion. With increasing monomer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalyst ratio the appropriate increase in polymer Mn was observed, indicating a very well controlled polymerisation. This was further supported by very low dispersities in all cases. The MALDI-ToF of the PLA 100
catalyst ratio the appropriate increase in polymer Mn was observed, indicating a very well controlled polymerisation. This was further supported by very low dispersities in all cases. The MALDI-ToF of the PLA 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at 80 °C indicated the BnO– and –H end groups and the main repeat unit was 144 g mol−1 with a minor series with a repeat unit of 72 g mol−1 indicative of a low degree of transesterification (see Fig. SI12, ESI†). Interestingly the iodide complexes afforded a significantly lower yield than the chloride. A similar trend was observed when using InX3 (InCl3vs. InI3).12b It was hypothesised that the halide remains in the coordination sphere of the indium centre and this accounts for the differences in reactivity.
1 at 80 °C indicated the BnO– and –H end groups and the main repeat unit was 144 g mol−1 with a minor series with a repeat unit of 72 g mol−1 indicative of a low degree of transesterification (see Fig. SI12, ESI†). Interestingly the iodide complexes afforded a significantly lower yield than the chloride. A similar trend was observed when using InX3 (InCl3vs. InI3).12b It was hypothesised that the halide remains in the coordination sphere of the indium centre and this accounts for the differences in reactivity.
Conclusions
In conclusion we have prepared the first examples of salan complexes of the form M–X (X = Cl, I and M = In, Ga). There is a switch from trigonal bipyramidal to square based pyramid upon increasing the size of the cation for 1H2, whilst for 2H2 no such change is observed, illustrating the rich and diverse chemistry of the group 13 elements. This change, in part, is presumably related to the rigidity of the bipyrrolidine derived ligand system. For the first time we have shown that indium–salan complexes can be effective initiators for the production of heterotactic PLA with narrow dispersities and controllable molecular weights, these results are complementary to the rich chemistry of In–salen complexes. This highlights the importance of the combination of metal/ligand in the rational design of initiators for the controlled ROP of cyclic esters.Acknowledgements
We thank the University of Bath and EPSRC for funding (MDJ, JB), Roger and Sue Whorrod for supporting a fellowship for AB.Notes and references
- (a) P. J. Dijkstra, H. Z. Du and J. Feijen, Polym. Chem., 2011, 2, 520–527 RSC; (b) B. J. O'Keefe, M. A. Hillmyer and W. B. Tolman, J. Chem. Soc., Dalton Trans., 2001, 2215–2224 RSC; (c) R. H. Platel, L. M. Hodgson and C. K. Williams, Polym. Rev., 2008, 48, 11–63 CrossRef CAS; (d) A. Sauer, A. Kapelski, C. Fliedel, S. Dagorne, M. Kol and J. Okuda, Dalton Trans., 2013, 42, 9007–9023 RSC.
- (a) A. P. Gupta and V. Kumar, Eur. Polym. J., 2007, 43, 4053–4074 CrossRef CAS; (b) L. T. Lim, R. Auras and M. Rubino, Prog. Polym. Sci., 2008, 33, 820–852 CrossRef CAS; (c) K. M. Nampoothiri, N. R. Nair and R. P. John, Bioresour. Technol., 2010, 101, 8493–8501 CrossRef PubMed.
- (a) D. Bourissou, S. Moebs-Sanchez and B. Martin-Vaca, C. R. Chim., 2007, 10, 775–794 CrossRef CAS; (b) J. B. Zhu and E. Y. X. Chen, J. Am. Chem. Soc., 2015, 137, 12506–12509 CrossRef CAS PubMed.
- (a) M. D. Jones, L. Brady, P. McKeown, A. Buchard, P. M. Schafer, L. H. Thomas, M. F. Mahon, T. J. Woodman and J. P. Lowe, Chem. Sci., 2015, 6, 5034–5039 RSC; (b) M. D. Jones, S. L. Hancock, P. McKeown, P. M. Schafer, A. Buchard, L. H. Thomas, M. F. Mahon and J. P. Lowe, Chem. Commun., 2014, 50, 15967–15970 RSC; (c) A. Amgoune, C. M. Thomas and J. F. Carpentier, Macromol. Rapid Commun., 2007, 28, 693–697 CrossRef CAS; (d) A. Amgoune, C. M. Thomas, S. Ilinca, T. Roisnel and J. F. Carpentier, Angew. Chem., Int. Ed. Engl., 2006, 45, 2782–2784 CrossRef CAS PubMed; (e) A. Amgoune, C. M. Thomas, T. Roisnel and J. F. Carpentier, Chem. – Eur. J., 2006, 12, 169–179 CrossRef CAS PubMed; (f) B. M. Chamberlain, M. Cheng, D. R. Moore, T. M. Ovitt, E. B. Lobkovsky and G. W. Coates, J. Am. Chem. Soc., 2001, 123, 3229–3238 CrossRef CAS PubMed; (g) M. H. Chisholm, J. Gallucci and K. Phomphrai, Chem. Commun., 2003, 48–49 RSC; (h) M. H. Chisholm, J. C. Gallucci and K. Phomphrai, Inorg. Chem., 2004, 43, 6717–6725 CrossRef CAS PubMed; (i) A. J. Chmura, M. G. Davidson, C. J. Frankis, M. D. Jones and M. D. Lunn, Chem. Commun., 2008, 1293–1295 RSC; (j) Z. R. Dai, Y. Y. Sun, J. Xiong, X. B. Pan, N. Tang and J. C. Wu, Catal. Sci. Technol., 2016, 6, 515–520 RSC; (k) A. Stopper, J. Okuda and M. Kol, Macromolecules, 2012, 45, 698–704 CrossRef CAS; (l) Y. Y. Sun, J. Xiong, Z. R. Dai, X. B. Pan, N. Tang and J. C. Wu, Inorg. Chem., 2016, 55, 136–143 CrossRef CAS PubMed; (m) A. L. Zelikoff, J. Kopilov, I. Goldberg, G. W. Coates and M. Kol, Chem. Commun., 2009, 6804–6806 RSC.
- C. K. Williams, L. E. Breyfogle, S. K. Choi, W. Nam, V. G. Young, M. A. Hillmyer and W. B. Tolman, J. Am. Chem. Soc., 2003, 125, 11350–11359 CrossRef CAS PubMed.
- (a) M. H. Chisholm, N. J. Patmore and Z. P. Zhou, Chem. Commun., 2005, 127–129 RSC; (b) H. Z. Du, A. H. Velders, P. J. Dijkstra, J. R. Sun, Z. Y. Zhong, X. S. Chen and J. Feijen, Chem. – Eur. J., 2009, 15, 9836–9845 CrossRef CAS PubMed; (c) P. Hormnirun, E. L. Marshall, V. C. Gibson, A. J. P. White and D. J. Williams, J. Am. Chem. Soc., 2004, 126, 2688–2689 CrossRef CAS PubMed; (d) N. Nomura, R. Ishii, M. Akakura and K. Aoi, J. Am. Chem. Soc., 2002, 124, 5938–5939 CrossRef CAS PubMed; (e) N. Spassky, M. Wisniewski, C. Pluta and A. LeBorgne, Macromol. Chem. Phys., 1996, 197, 2627–2637 CrossRef CAS; (f) M. Wisniewski, A. LeBorgne and N. Spassky, Macromol. Chem. Phys., 1997, 198, 1227–1238 CrossRef CAS; (g) Z. Y. Zhong, P. J. Dijkstra and J. Feijen, Angew. Chem., Int. Ed. Engl., 2002, 41, 4510–4513 CrossRef CAS; (h) Z. Y. Zhong, P. J. Dijkstra and J. Feijen, J. Am. Chem. Soc., 2003, 125, 11291–11298 CrossRef CAS PubMed.
- K. M. Osten, D. C. Aluthge and P. Mehrkhodavandi, Dalton Trans., 2015, 44, 6126–6139 RSC.
- (a) E. L. Whitelaw, G. Loraine, M. F. Mahon and M. D. Jones, Dalton Trans., 2011, 40, 11469–11473 RSC; (b) K. Nie, W. K. Gu, Y. M. Yao, Y. Zhang and Q. Shen, Organometallics, 2013, 32, 2608–2617 CrossRef CAS; (c) A. Pilone, N. De Maio, K. Press, V. Venditto, D. Pappalardo, M. Mazzeo, C. Pellecchia, M. Kol and M. Lamberti, Dalton Trans., 2015, 44, 2157–2165 RSC; (d) A. Pilone, K. Press, I. Goldberg, M. Kol, M. Mazzeo and M. Lamberti, J. Am. Chem. Soc., 2014, 136, 2940–2943 CrossRef CAS PubMed; (e) I. D. Vieira, E. L. Whitelaw, M. D. Jones and S. Herres-Pawlis, Chem. – Eur. J., 2013, 19, 4712–4716 CrossRef CAS PubMed.
- (a) C. Bakewell, T. P. A. Cao, X. F. Le Goff, N. J. Long, A. Auffrant and C. K. Williams, Organometallics, 2013, 32, 1475–1483 CrossRef CAS; (b) T. P. A. Cao, A. Buchard, X. F. Le Goff, A. Auffrant and C. K. Williams, Inorg. Chem., 2012, 51, 2157–2169 CrossRef CAS PubMed.
- S. Dagorne, M. Normand, E. Kirillov and J. F. Carpentier, Coord. Chem. Rev., 2013, 257, 1869–1886 CrossRef CAS.
- (a) J. C. Buffet, J. Okuda and P. L. Arnold, Inorg. Chem., 2010, 49, 419–426 CrossRef CAS PubMed; (b) A. Kapelski and J. Okuda, J. Polym. Sci., Part A: Polym. Chem., 2013, 51, 4983–4991 CrossRef CAS; (c) I. Peckermann, A. Kapelski, T. P. Spaniol and J. Okuda, Inorg. Chem., 2009, 48, 5526–5534 CrossRef CAS PubMed.
- (a) A. Pietrangelo, M. A. Hillmyer and W. B. Tolman, Chem. Commun., 2009, 2736–2737 RSC; (b) A. Pietrangelo, S. C. Knight, A. K. Gupta, L. J. Yao, M. A. Hillmyer and W. B. Tolman, J. Am. Chem. Soc., 2010, 132, 11649–11657 CrossRef CAS PubMed.
- (a) D. C. Aluthge, B. O. Patrick and P. Mehrkhodavandi, Chem. Commun., 2013, 49, 4295–4297 RSC; (b) D. C. Aluthge, E. X. Yan, J. M. Ahn and P. Mehrkhodavandi, Inorg. Chem., 2014, 53, 6828–6836 CrossRef CAS PubMed; (c) A. F. Douglas, B. O. Patrick and P. Mehrkhodavandi, Angew. Chem., Int. Ed. Engl., 2008, 47, 2290–2293 CrossRef CAS PubMed; (d) A. B. Kremer, K. M. Osten, I. Yu, T. Ebrahimi, D. C. Aluthge and P. Mehrkhodavandi, Inorg. Chem., 2016, 55, 5365–5374 CrossRef CAS PubMed; (e) K. M. Osten, D. C. Aluthge, B. O. Patrick and P. Mehrkhodavandi, Inorg. Chem., 2014, 53, 9897–9906 CrossRef CAS PubMed; (f) K. M. Osten, I. S. Yu, I. R. Duffy, P. O. Lagaditis, J. C. C. Yu, C. J. Wallis and P. Mehrkhodavandi, Dalton Trans., 2012, 41, 8123–8134 RSC; (g) C. L. Xu, I. S. Yu and P. Mehrkhodavandi, Chem. Commun., 2012, 48, 6806–6808 RSC; (h) I. Yu, A. Acosta-Ramirez and P. Mehrkhodavandi, J. Am. Chem. Soc., 2012, 134, 12758–12773 CrossRef CAS PubMed; (i) D. C. Aluthge, J. M. Ahn and P. Mehrkhodavandi, Chem. Sci., 2015, 6, 5284–5292 RSC.
- (a) C. Bakewell, A. J. P. White, N. J. Long and C. K. Williams, Inorg. Chem., 2013, 52, 12561–12567 CrossRef CAS PubMed; (b) S. Ghosh, R. R. Gowda, R. Jagan and D. Chakraborty, Dalton Trans., 2015, 44, 10410–10422 RSC; (c) P. Horeglad, M. Cybularczyk, B. Trzaskowski, G. Z. Zukowska, M. Dranka and J. Zachara, Organometallics, 2015, 34, 3480–3496 CrossRef CAS; (d) P. Horeglad, A. Litwinska, G. Z. Zukowska, D. Kubicki, G. Szczepaniak, M. Dranka and J. Zachara, Appl. Organomet. Chem., 2013, 27, 328–336 CrossRef CAS.
- N. Maudoux, J. Fang, T. Roisnel, V. Dorcet, L. Maron, J. F. Carpentier and Y. Sarazin, Chem. – Eur. J., 2014, 20, 7706–7717 CrossRef CAS PubMed.
- C. Bakewell, A. J. P. White, N. J. Long and C. K. Williams, Angew. Chem., Int. Ed. Engl., 2014, 53, 9226–9230 CrossRef CAS PubMed.
- (a) T. M. Ovitt and G. W. Coates, J. Am. Chem. Soc., 2002, 124, 1316–1326 CrossRef CAS PubMed; (b) D. Pappalardo, M. Bruno, M. Lamberti, M. Mazzeo and C. Pellecchia, J. Mol. Catal. A: Chem., 2013, 379, 303–308 CrossRef CAS.
- E. Sergeeva, J. Kopilov, I. Goldberg and M. Kol, Chem. Commun., 2009, 3053–3055 RSC.
- (a) K. Press, I. Goldberg and M. Kol, Angew. Chem., Int. Ed. Engl., 2015, 54, 14858–14861 CrossRef CAS PubMed; (b) T. Rosen, I. Goldberg, V. Venditto and M. Kol, J. Am. Chem. Soc., 2016, 138, 12041–12044 CrossRef CAS PubMed.
- A. J. Chmura, M. G. Davidson, M. D. Jones, M. D. Lunn, M. F. Mahon, A. F. Johnson, P. Khunkamchoo, S. L. Roberts and S. S. F. Wong, Macromolecules, 2006, 39, 7250–7257 CrossRef CAS.
- D. A. Atwood, A. R. Hutchison and Y. Zhang, in Group 13 Chemistry III: Industrial Applications, ed. H. W. Roesky and D. A. Atwood, Springer Berlin Heidelberg, Berlin, Heidelberg, 2003, pp. 167–201 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Full experimental and the crystal data in the .cif format. CCDC 1510494–1510501. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6nj03844a |
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 |