Crystal structure of Aquifex aeolicus gene product Aq1627: a putative phosphoglucosamine mutase reveals a unique C-terminal end-to-end disulfide linkage
Abstract
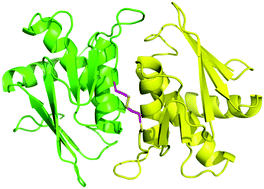
The Aq1627 gene from Aquifex aeolicus, a hyperthermophilic bacterium has been cloned and overexpressed in Escherichia coli. The protein was purified to homogeneity and its X-ray crystal structure was determined to 1.3 Å resolution using multiple wavelength anomalous dispersion phasing. The structural and sequence analysis of Aq1627 is suggestive of a putative phosphoglucosamine mutase. The structural features of Aq1627 further indicate that it could belong to a new subclass of the phosphoglucosamine mutase family. Aq1627 structure contains a unique C-terminal end-to-end disulfide bond, which links two monomers and this structural information can be used in protein engineering to make proteins more stable in different applications.


 Please wait while we load your content...
Please wait while we load your content...