Charge-transfer-energy-dependent oxygen evolution reaction mechanisms for perovskite oxides†
Abstract
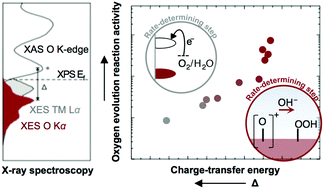
Numerous studies have reported electronic activity descriptors of oxygen evolution reaction (OER) for oxide catalysts under a single reaction mechanism. However, recent works have revealed that a single mechanism is not at play across oxide chemistries. These works underscore a need to deeply investigate the electronic structure details of active oxide catalysts and how they align with the OER potential, which is critical to understanding the interfacial charge-transfer kinetics that dictate catalytic mechanisms. In this work, we use soft X-ray emission and absorption spectroscopy of perovskites to analyze the partial density of states on an absolute energy scale, from which energetic barriers for electron transfer and surface deprotonation were estimated and correlated with OER activity. Through this lens, we identify that decreasing the solid-state charge-transfer energy of perovskites can change the mechanisms of the OER from electron-transfer-limited to proton–electron-coupled, to proton-transfer-limited reactions. This concept is supported by the analysis of potential energy surfaces for sequential and concerted proton–electron transfer pathways using a Marcus model. Our work highlights the importance of understanding the physical origin of experimental OER activity trends with electronic descriptors and the need to promote surface deprotonation from oxides to discover new catalysts with enhanced activity.
- This article is part of the themed collection: 2017 Energy and Environmental Science HOT articles


 Please wait while we load your content...
Please wait while we load your content...