Open Access Article
Open Access ArticleSuperoleophobic surfaces
Jiale
Yong
a,
Feng
Chen
 *a,
Qing
Yang
*b,
Jinglan
Huo
a and
Xun
Hou
a
*a,
Qing
Yang
*b,
Jinglan
Huo
a and
Xun
Hou
a
aState Key Laboratory for Manufacturing System Engineering and Shaanxi Key Laboratory of Photonics Technology for Information, School of Electronics & Information Engineering, Xi’an Jiaotong University, Xi’an, 710049, P. R. China. E-mail: chenfeng@mail.xjtu.edu.cn
bSchool of Mechanical Engineering, Xi’an Jiaotong University, Xi’an, 710049, P. R. China. E-mail: yangqing@mail.xjtu.edu.cn
First published on 2nd May 2017
Abstract
Superoleophobicity is a phenomenon where the contact angles of various oil droplets with low surface tension on a solid surface are larger than 150°. In the past few years, there has been much growing interest in the design and application of superoleophobic surfaces. Such surfaces have great significance for both fundamental research and a variety of practical applications, including oil-repellent coatings, self-cleaning, oil/water separation, oil droplet manipulation, chemical shielding, anti-blocking, designing liquid microlens, oil capture, bioadhesion, guiding oil movement and floating on oil. Herein, we systematically summarize the recent developments of superoleophobic surfaces. This review focuses on the design, fabrication, characteristics, functions, and important applications of various superoleophobic surfaces. Although many significant advances have been achieved, superoleophobic surfaces are still in their “toddler stage” of development. The current challenges and future prospects of this fast-growing field of superoleophobicity are discussed.
1. Introduction
Solid, liquid and gas are the three basic states of matter. The wettability of solid surfaces, which depends on the interfacial interaction of the solid/liquid/gas phase has great significance for the survival of animals/plants and for our daily life, and the materials showing special extreme wettability are particularly captivating.1–9 Superhydrophobic surfaces and superoleophobic surfaces on which the water droplets and oil droplets present contact angles (CAs) above 150°, are the most widely studied interfaces in wettability, due to their excellent water- or oil-repellent ability.10–15 Inspired by the natural fascinating features of super-wetting surfaces, people have attempted to change the wettability of various materials either by creating a hierarchical rough microstructure, or by modifying the substrate with a layer of chemical coating for the reason that the wettability of the material surfaces is mainly governed by both the surface geometrical microstructure and the surface chemical composition.16–21 To date, a very large number of superhydrophobic surfaces have been developed, and such artificial materials have a wide variety of practical applications in self-cleaning coatings,20,22–25 antifouling,15 anti-fogging/icing/corrosion,26–29 drag reduction,30–35 microfluidics,36–41 lab-on-a-chip,42–45 cell engineering,24,46–50 microdroplet manipulation,51–55 oil/water separation,56–60 guiding liquid sliding,61–64 water collection,65–67 surface-enhanced Raman scattering (SERS)68–70 and super-buoyant micro-boats.71 However, superoleophobic surfaces that will not be wet by organic liquids are difficult to achieve, due to the fact that surface tensions of organic liquids are commonly very low, as shown in Table 1.72–74 Similar to superhydrophobic surfaces, superoleophobic surfaces have gained more and more attention recently because of their remarkable potential applications, including anti-oil coatings,75–78 self-cleaning,22,79,80 oil/water separation,56,81–88 oil droplet manipulation,78,80,89–93 chemical shielding,75 anti-blocking,79,94 designing liquid microlens,95 oil capture,96 bioadhesion,97 guiding oil movement,92,98 and floating on oil.99,100| Liquid | Surface tension (mN m−1) | Density (g cm−3) |
|---|---|---|
| Water | 72.8 | 1.0 |
| Glycerol | 64.0 | 1.26 |
| 1,2-Dichloroethane | 33.3 | 1.26 |
| Liquid paraffin | 33.1 | 0.83–0.86 |
| Toluene | 28.4 | 0.87 |
| Chloroform | 27.5 | 1.50 |
| n-Hexadecane | 27.5 | 0.77 |
| n-Tetradecane | 26.6 | 0.76 |
| Dichloromethane | 26.5 | 1.33 |
| Cyclohexane | 25.9 | 0.78 |
| n-Dodecane | 25.4 | 0.75 |
| Cyclohexane | 25.0 | 0.78 |
| n-Decane | 23.8 | 0.73 |
| Gasoline | ≈22.0 | 0.70–0.78 |
| n-Octane | 21.6 | 0.70 |
| n-Heptane | 20.1 | 0.68 |
| n-Hexane | 18.4 | 0.66 |
| n-Pentane | 18.3 | 0.63 |
There have been three breakthroughs that have made important contributions to the development of superoleophobicity. In 1997, Tsujii et al. first reported a superoleophobic surface, which was obtained by modifying an anodized rough aluminum plate with fluorinated monoalkylphosphate.101 The CA value of a rapeseed oil droplet on this surface was 150°; however, the surface tension of the rapeseed oil is not low (∼55 mN m−1). Subsequently, the research on superoleophobicity still developed very slowly, until in 2007, Tuteja et al. proposed and demonstrated the importance of re-entrant surface curvature to achieving superoleophobicity, except for ultralow surface free energy and rough microstructures.72 The surfaces with re-entrant textures generally have overhang, inverted trapezoid, mushroom-like microstructures, and so on.3,12,72,73,102–108 They further proposed two design parameters for forming stable superoleophobic surfaces.73,108–120 The concept of re-entrant texture has greatly accelerated the development of this field.3,12,109,110 In 2009, Jiang et al. revealed the internal mechanism of the anti-oil-pollution ability of fish skin in water.76 The ability stems from the unique underwater superoleophobicity of fish scales. Water can wet the fish skin and be trapped in the rough microstructures of the fish scale surface. The trapped water cushion repels oil and endows the fish scales with superoleophobicity in a water environment. This finding has opened up an important new way for achieving superoleophobicity underwater.76,79,111–116 By the combination of suitable surface roughness, ultralow or ultrahigh (in underwater case) surface-energy materials and even re-entrant surface texture, various superoleophobic surfaces are continuously being engineered either in air or in water.3,12–14,76,79,96,108–123 However, superoleophobicity by itself does not fully satisfy the needs of practical application. Endowing superoleophobic surfaces with multiple functions (e.g., controllable oil-adhesion,78,80,89,91,112,124 anisotropic oil-wettability,125–127 transparency,117,120,128,129 durability and self-healing)121,130–135 and smart properties (respond to external stimulus, e.g., light,136–138 pH,139–141 temperature,142,143 electrical potential,144,145 magnetic field,146 density of surroundings90) has become the main trend of this research field. The design and realization of more complicated and subtle superoleophobic surfaces have remained as immense challenges.
This review will systematically summarize the recent important developments of superoleophobic surfaces. First, creatures in nature with special superoleophobicity, and classical wetting models are introduced as the background (Section 2). Then, the constructing principle and various properties of superoleophobic surfaces both in air (Section 3) and underwater (Section 4) are reviewed. The next part (Section 5) focuses on the smart superoleophobic surfaces that can response to external stimulation to reversibly switch their static or dynamic wettability. After that, we present a variety of practically functional applications (Section 6). Finally, the challenges and prospects of superoleophobic surfaces are briefly discussed (Section 7).
2. Biological and theoretical backgrounds
2.1 Creatures in nature with special wettability
To survive and adapt to the harsh environment, natural species have evolved all sorts of perfect multifunctional surfaces after millions of years of evolution and natural selection, exhibiting the unification and coordination of structure and performance.147–150 Therefore, there are many animals and plants having surfaces with diversified wettabilities, such as the self-cleaning property of the lotus leaf (Fig. 1a),151,152 the high adhesive force of rose petals (Fig. 1b),153 the anisotropic wettability of the rice leaf (Fig. 1c),154,155 the directional adhesion of a butterfly wing (Fig. 1d),156 the anti-fogging ability of a mosquito eye (Fig. 1e),26 water-collection of a desert beetle (Fig. 1f),65 underwater long-term air retention of Salvinia plants (Fig. 1g),157 the ability of a water strider to walk and jump on the surface of water (Fig. 1h).158,159 It is found that both hierarchical microstructures and the chemical composition atop them lead to the specialities of the above creatures, which inspire humans to design and prepare bionic surfaces with special wettability.63,76,152,160–162 In recent years, many creatures were also found to show superoleophobicity and anti-oil properties. | ||
| Fig. 1 Photographs and surface microstructures (SEM images) of various natural animals and plants showing special wettability: (a) lotus leaf, (b) red rose petal, (c) rice leaf, (d) a butterfly wing, (e) mosquito eye, (f) back of a desert beetle, (g) Salvinia plant, and (h) leg of a water strider. (a) Reproduced from ref. 152 with permission from Wiley, copyright 2008. (b) Reproduced from ref. 153 with permission from ACS, copyright 2008. (c) Reproduced from ref. 155 with permission from Wiley, copyright 2011. (d) Reproduced from ref. 156 with permission from RSC, copyright 2007. (e) Reproduced from ref. 26 with permission from Wiley, copyright 2007. (f) Reproduced from ref. 65 with permission from NPG, copyright 2001. (g) Reproduced from ref. 157 with permission from Wiley, copyright 2010. (h) Reproduced from ref. 158 with permission from NPG, copyright 2004. | ||
 | ||
| Fig. 2 Photographs and surface microstructures of natural creatures with superoleophobic surfaces: (a) springtail's skin, (b–e) fish scales, (f and g) clam's shell, (h and i) lower side of a lotus leaf, (j and k) seaweed, and (l and m) filefish skin. (a) Reproduced from ref. 167 with permission from Helbig et al., copyright 2011. (c–e) Reproduced from ref. 76 with permission from Wiley, copyright 2009. (f and g) Reproduced from ref. 77 with permission from Wiley, copyright 2012. (h and i) Reproduced from ref. 170 with permission from RSC, copyright 2011. (j and k) Reproduced from ref. 116 with permission from Wiley, copyright 2015. (l and m) Reproduced from ref. 127 with permission from Wiley, copyright 2014. | ||
Among the above-mentioned animals and plants, springtails skin shows superoleophobicity in air, while fish scales, clam shells, the lower side of the lotus leaf, seaweed and filefish skin show superoleophobicity in water. Inspired by these creatures, we find that there are two different routes for designing superoleophobic surfaces: in air and in water.
2.2 Theoretical basis
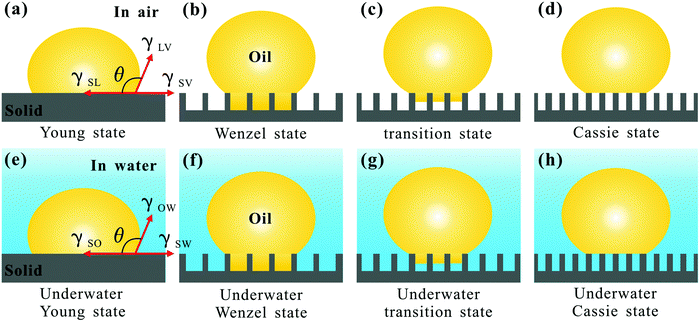
When a small liquid droplet is placed onto a solid surface, one phenomenon will occur: the droplet firstly touches the substrate and forms a three-phase contact line (TPCL); then the TPCL continues to advance and finally stops when it reaches a certain radius, resulting in an approximately spherical segment of this droplet. In 1805, the English physicist Thomas Young first described the forces acting on a droplet that is placed on an ideal flat surface, as shown in Fig. 3a.15 He considered that the contact angle (θ) of the liquid droplet was mainly dependent on the interfacial energy/tension between the solid–vapor (γSV), solid–liquid (γSL) and liquid–vapor (γLV) interfaces by the following: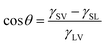 | (1) |
This equation is just suitable for an ideal smooth surface. As early as 1936, Wenzel realized the impact of roughness on the wettability of a material surface.171 According to the definition of surface tension, he pointed out that actual surface area of a rough substrate was larger than its apparent surface area, and further put forward a new model (Fig. 3b) to describe the contact angle θ* on a rough surface:
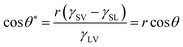 | (2) |
When the surface is porous and the voids are very difficult to be wetted by liquid but are filled with air, the wettability of such an interface is very different from the Wenzel state. In 1944, Cassie and Baxter proposed an advanced model to describe the apparent contact angle θ* of a small liquid droplet on a heterogeneous surface (Fig. 3d).169 For example, if a surface is made up of two parts of different materials, the θ* on such a composite surface can be expressed as follows:
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ* = f1 θ* = f1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ1 + f2 θ1 + f2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ2 θ2 | (3) |
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ* = f θ* = f![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ + (1 − f)cos θ + (1 − f)cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 180° = f 180° = f![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ + f − 1 θ + f − 1 | (4) |
Although both Wenzel and Cassie equations were originally put forward as semi-empirical formulas, their rigorous derivations were carried out afterwards from the point of thermodynamics.172–175 The wetting states mentioned above were verified in the research of superhydrophobicity, which has been widely studied. The surfaces belonging to the Wenzel model usually show very high adhesion to a liquid droplet, while the surfaces belonging to the Cassie model usually show extremely low adhesion to a liquid droplet.16,21,55,176–179 In addition, it is found that there also exists a transition state (Fig. 3c) between the Wenzel state and the Cassie state.16,55,174,178–184 The liquid droplet may partly penetrate into the valley of the rough surface microstructures. The surfaces in this state often have a liquid adhesion between the Wenzel state and the Cassie state. Such adhesion can usually be changed from low to high, depending on the extent of the droplet penetrating into the rough microstructures.
The above-mentioned four typical wetting states (Young, Wenzel, transition, and Cassie states) are also valid for an oil droplet on flat or rough solid substrates, as shown in Fig. 3a–d. In this situation, the liquid in those models and equations refers to corresponding oils. Recently, oil-wettability of solid materials in water medium has been receiving increasing attention in research, due to its broad, promising applications.76,79,111–116 The typical wetting states in air can be generalized to their underwater versions, i.e., underwater Young state (Fig. 3e), underwater Wenzel state (Fig. 3f), underwater transition state (Fig. 3g), and underwater Cassie state (Fig. 3h).
The oil droplet on a flat solid surface in water is in the underwater Young state, resulting in a solid/water/oil three-phase interface (Fig. 3e).76,94 The contact angle (θOW) of an oil droplet in the underwater Young state (on a flat substrate) satisfies the eqn (5).
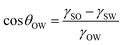 | (5) |
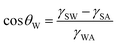 | (6) |
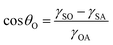 | (7) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θW + γSA and γSO = γOA
θW + γSA and γSO = γOA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θO + γSA. Then, eqn (5) can be changed as follows:
θO + γSA. Then, eqn (5) can be changed as follows: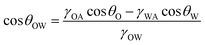 | (8) |
For a hydrophilic surface, it also shows oleophilicity in air because oils have lower surface tension than water (θO < θW < 90°), so the values of cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θO and cos
θO and cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θW are all positive. Since the surface tension of oil/organic liquids are much lower than that of water (γOA ≪ γWA), the value of γOA
θW are all positive. Since the surface tension of oil/organic liquids are much lower than that of water (γOA ≪ γWA), the value of γOA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θO − γWA
θO − γWA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θW is commonly negative. From eqn (8), it can be predicted that most hydrophilic surfaces in air show oleophobicity in water.13,76,81 After combining with rough hierarchical microstructures, the underwater Wenzel state, underwater transition state, or underwater Cassie state may form in the solid/water/oil system. In the underwater Wenzel state (Fig. 3f), the underwater oil droplet wets the substrate and fills the valleys of the rough surface microstructures.171 The Wenzel equation can be expressed as follows:
θW is commonly negative. From eqn (8), it can be predicted that most hydrophilic surfaces in air show oleophobicity in water.13,76,81 After combining with rough hierarchical microstructures, the underwater Wenzel state, underwater transition state, or underwater Cassie state may form in the solid/water/oil system. In the underwater Wenzel state (Fig. 3f), the underwater oil droplet wets the substrate and fills the valleys of the rough surface microstructures.171 The Wenzel equation can be expressed as follows:
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θOW* = r θOW* = r![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θOW θOW | (9) |
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θOW* = f θOW* = f![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θOW + f − 1 θOW + f − 1 | (10) |
2.3 Terminology about super-oil-wettability
With the rapid development of the research field related to wettability, thousands of surfaces showing special wetting properties have been developed. Meanwhile, many terms are created and used to describe those special wetting phenomena. In addition to the “classical” terms, “hydrophobic” and “hydrophilic”, other terms such as “oleophobic(philic)”, “hygrophobic(philic)”, “lyophobic(philic)”, “amphiphobic”, “omniphobic” have also appeared in the many recent publications. Sometimes they are preceded by a prefix “super” to express their extreme cases. Currently, the biggest problem is that the terms of wettability are not well-defined.3,185 A same term used in different reports (by different authors) may have different meanings. For example, some people think the superhydrophobic surface must have a CA larger than 150° and a SA (or CA hysteresis) smaller than 10° with respect to a small water droplet, while the others think it is a superhydrophobic surface only when a water droplet shows a CA larger than 150° on this surface. “Superomniphobicity” is used by some people to describe the surfaces that are repellent to both high and low surface tension liquids (oils); we find that the surface wettabilities in their articles are actually the same as the definition of “superamphiphobicity”, i.e., superomniphobicity is confused with superamphiphobicity. Some people used “superlyophobic” to express the probe liquid showing CA ≥ 150° on a solid substrate, but the prefix “lyo-” comes from the word “dispersion” or “dissolution”, so the “superlyophobic” is qualified as inappropriate for wettability classification. Creating uniform, clear terminology is very important to enable precise scientific and technical communication of measured and theoretical wettability results, and to establish guidelines for understanding and designing future surfaces. Recently, Marmur discussed the problem of the current terminology and attempted to classify the terminology of wettability.185 He proposed some new terms (e.g., parahydrophobic, superhygrophobic, etc.) and re-defined some terms with new condition (e.g., “superhydrophilic”, referring to only the complete wetting state with a CA of zero, etc.). Although his suggestions are very reasonable, those terms were rarely adopted by researchers in the next few years, probably because the step from the widely accepted terms to those suggested terminologies seems too big. The aforementioned terminology problem is still far from being solved. We believe that preserving the traditional meaning of those terms as much as possible and making appropriate modifications are the best ways to enable the wettability classification is clear and acceptable.As is well know, “phobic” and “philic” come from the Greek words for “fear” and “love”, respectively; “hydro” (from Greek), “oleo” (from Latin), and “hygro” (from Greek) refer to “water”, “oils or fats”, and “liquid”, respectively. The first four roots have gone deep in the brains of researchers, and the fifth root was suggested by Marmur.185 We should build a clear terminology system for wettability on the basis of these roots. It is also worth noting that “oleo” is from Latin while “lipo” is from Greek for oils or fats. Although “lipo” should replace “oleo” in the publications, we suggest still using “oleo” rather than “lipo” because researchers have become accustomed to using “oleo”. According to the classical partition, the CA of 90° is the boundary between “phobic” (>90°) and “philic” (<90°). The prefix of “hydro”, “oleo”, and “hygro” can be combined with “phobic” and “philic”, respectively, to generate six terms: hydrophobic, hydrophilic, oleophobic, oleophilic, hygrophobic, hygrophilic. “Hydrophobic” and “hydrophilic” are used for the probe liquid of water; “oleophobic” and “oleophilic” are used for the probe liquids of low-surface-tension oils and other organic liquids; when the probe liquid is not certain, we can consider temporarily using “hygrophobic” and “hygrophilic” (for example, when we introduce different wetting states in Section 2.2, the liquid droplets in those models can be water or it can be oils, and it can also be other kind of liquids, so “hygrophobic” and “hygrophilic” are temporarily adopted for liquid-repelling and liquid-loving). When the above six properties reach their extreme cases, the terms that describe such special wetting states can be derived by prefixing the word “super” before the fundamental terms, i.e., superhydrophobic, superhydrophilic, superoleophobic, superoleophilic, superhygrophobic, superhygrophilic. We suggest here that only the static wetting property is used to determine the above-mentioned terms without regard to dynamic wettability, to avoid the double standards of one term (for example, superhydrophobicity referring to both CA ≥ 150° and SA ≤ 10° or just CA ≥ 150°); i.e., those terms just depend on the CA values of the probe liquid droplet on the material surfaces. When CA ≥ 150°, the surfaces are superhydrophobic, superoleophobic, and superhygrophobic. In contrast, surfaces are superhydrophilic, superoleophilic, and superhygrophilic as long as CA ≤ 10°. The dynamic characteristics of a small liquid droplet on a solid surface, which are usually assessed by SA, CA hysteresis, and adhesive force measurement, reflect another aspect of the surface property, i.e., liquid adhesion. Some superhydrophobic surfaces show low adhesion (such as the lotus leaf), while some superhydrophobic surfaces show high adhesion (such as a rose petal) to a water droplet. These wettabilities can be expressed as “superhydrophobicity with low/high adhesion”, “low/high-adhesive superhydrophobicity”, “roll-down/sticky superhydrophobicity”, etc., to differentiate them. Such forms of expression apply to superoleophobicity as well. The surfaces simultaneously showing superhydrophobic and superoleophobic properties are generally known as the “superamphiphobic” interfaces. Sometimes “superomniphobic” is also used to describe a surface that repels various liquids from high-surface-tension water to low-surface-tension oils. However, in most current cases, “superomniphobic” is overlapped and confused with “superamphiphobic”. The re-definition of “superomniphobic” and making it different from “superamphiphobic” is badly needed. Since “amphi” comes from the Greek word for “both” and “omni” comes from the Latin word for “all”, there is a relation: “oleo” ⊂ “amphi” ⊂ “omni”. Constructing a “superomniphobic” surface should be more difficult than a “superamphiphobic” surface, therefore, we suggest that superomniphobic surfaces can super-repel almost all liquids, especially very low surface tension fluids (10–20 mN m−1), while superoleophobic/superamphiphobic surfaces tend to only work above this 20 mN m−1 limit. The superomniphobic surfaces have been fabricated by Tuteja and Kim et al.73,107 The above-mentioned fourteen terms (hydrophobic, hydrophilic, oleophobic, oleophilic, hygrophobic, hygrophilic, superhydrophobic, superhydrophilic, superoleophobic, superoleophilic, superhygrophobic, superhygrophilic, superamphiphobic, and superomniphobic) are enough to describe the various wetting states. They are all adjectives, and their corresponding nouns are generated by add the suffix of “-ity”. The word-building of these terms for super-wettability is summarized in Fig. 4.
3. Superoleophobicity in air
There are enormous difficulties in fabricating superoleophobic surfaces when the vast majority of organic liquids have ultralow surface tension.12,13,72,101,112,117,186 Both the biological and theoretical backgrounds reveal that enough roughness is very important to achieve superoleophobicity in air.167,169 In addition, materials having extremely low surface energy are preferable. Considering eqn (1), when θ = 90°, which is the boundary of hygrophilicity and hygrophobicity, γSV must be equal to γSL. The γSL can be approximated as follows:101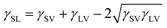 | (11) |
According to eqn (1) and (11), we get γSV = γLV/4 as the condition for θ = 90°. As shown in Table 1, typical surface tensions of common oils are 18–33 mN m−1. To further achieve superoleophobicity, the required surface tension of material surface must be as low as a few mN m−1. The –CF3 group, almost alone, has quite low surface energy.13,101 Therefore, to fabricate superoleophobic surfaces, most materials need be modified with a low-surface-energy layer after the formation of rough microstructures. Some of the commonly used superoleophobic chemical modifications are summarized in Table 2.
| Chemical compounds | CAS no. | Chemical structures | Ref. |
|---|---|---|---|
| Fluorinated polyhedral oligomeric silsesquioxane (POSS) | As shown in the inset of Fig. 5e | 72, 73, 75, 108, 121, 187, 202 and 256 | |
| 1H,1H,2H,2H-Perfluorodecyltrichlorosilane | 78560-44-8 |
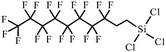
|
96, 120, 138, 200 and 238 |
| 1H,1H,2H,2H-Perfluorooctyltrichlorosilane | 78560-45-9 |
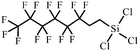
|
103, 105, 125, 193–195 and 203 |
| 1H,1H,2H,2H-Perfluorodecyltriethoxysilane | 101947-16-4 |
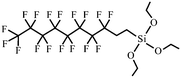
|
131, 204, 254 and 256 |
| 1H,1H,2H,2H-Perfluoroalkyltriethoxysilane | 51851-37-7 |
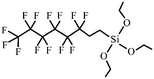
|
206 and 257 |
| 1H,1H,2H,2H-Perfluorodecanethiol | 34143-74-3 |

|
89 and 255 |
| Perfluorodecanoic acid | 335-76-2 |

|
192 |
| Perfluorooctanoic acid | 335-67-1 |
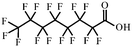
|
130 |
3.1 Re-entrant texture
The formation of the Cassie wetting state is crucial to achieving superoleophobicity. However, not all rough microstructures can result in a robust Cassie wetting state with the liquid droplets having extremely low surface tension (such as oils, alcohols and other organic solvents). The Young's CAs of these liquids are far less than 90° on any flat solid surface. In addition to the rough surface microstructure and low-surface-energy chemical composition, re-entrant surface curvature as the third parameter plays a very important role in designing superoleophobic surfaces.3,12,72,73,102–108Fig. 5a and b shows two schematic diagrams of hypothetical solid–air–liquid situations if a liquid contacts two types of rough substrates. ψ is the local geometric angle of the texture. We presume that such two substrates are made up of the same material and therefore have the same surface free energy, so the given liquid on the two substrates shows equal intrinsic or Young's contact angle, θ. Regarding the situation of θ < ψ, as shown in Fig. 5a, there is a downward net traction on the liquid–air interface. This traction can drive the liquid to penetrate into the valley of the rough microstructures and wet the solid texture, forming a fully-wetted Wenzel contact state. However, in the case of θ > ψ in Fig. 5b, the net traction is in the upward direction. The net traction tends to drive the liquid–air interface to recede to the top edge of the microstructures, resulting in a three-phase (solid/air/liquid) interface. Therefore, to form a robust Cassie state, the condition of θ ≥ ψ must be met.72,73,109 The microstructures of the material surface can be roughly divided into two types: trapezoid (ψ > 90°) and inverted-trapezoid (ψ < 90°). According to the design constraint (θ ≥ ψ), any liquid on these two textures in the Cassie state should be one of the situations depicted in Fig. 5c and d. Oils commonly have very low surface tension and almost all of their Young's CAs are smaller than 90°; i.e., θ < 90°. Consequently, oil liquids on trapezoid textures with ψ > 90° are unable to be at robust Cassie states due to θ < ψ, but the Cassie state can possibly be realized on inverted-trapezoid textures with ψ < 90° (Fig. 5d). Such inverted-trapezoid surface microstructures (ψ < 90°) are usually termed re-entrant textures.72,73,109
 | ||
| Fig. 5 The crucial role of re-entrant texture in designing superoleophobic surfaces in air. (a and b) Schematic of hypothetical solid–air–liquid interfaces on two types of rough surfaces. (c and d) Schematic of two possible robust Cassie states: (c) trapezoid texture with θ > ψ ≥ 90°; (d) inverted-trapezoid (re-entrant) texture with 90° > θ > ψ. (e–l) Superoleophobic surfaces with various re-entrant microstructures: (e) electrospun fibers; (f) regular micro-hoodoo surface; (g) micro-pillars with wavy vertical side walls; (h) inverse-trapezoidal microstructure; (i) T-shaped overhang microstructure; (j) silicon nanonails composed of a wavy stem and a bigger head; (k) microscale posts with nanoscale vertical overhangs (doubly re-entrant structure); (l) rough stainless steel wire meshes. The inset in (e) is the structure of POSS molecule. The insets in (g, h and i) are the magnified images of the corresponding re-entrant structures. The inset in (l) shows a variety of oils (n-dodecane, n-heptane, repeseed oil and n-hexadecane) on the rough metal mesh. (e and f) Reproduced from ref. 73 with permission from the National Academy of Sciences of the USA, copyright 2008. (g) Reproduced from ref. 103 with permission from ACS, copyright 2011. (h) Reproduced from ref. 104 with permission from RSC, copyright 2010. (i) Reproduced from ref. 105 with permission from RSC, copyright 2014. (j) Reproduced from ref. 106 with permission from ACS, copyright 2008. (k) Reproduced from ref. 107 with permission from AAAS, copyright 2014. (l) Reproduced from ref. 108 with permission from Wiley, copyright 2012. | ||
The importance of re-entrant surface curvature to achieving superoleophobicity was first demonstrated by Tuteja et al. in 2007.72,73 A series of fluorodecyl polyhedral oligomeric silsesquioxane (POSS) molecules were synthesized by them (inset of Fig. 5e). Then, they used POSS to modify the fiber mats, which were prepared by electrospinning (Fig. 5e).73 Unexpectedly, the composition of fluorodecyl POSS had intrinsic OCAs smaller than 90°, while the final fiber nets showing re-entrant surface curvature displayed superoleophobicity. Similarly, a regular micro-hoodoo surface was also prepared, which presented superoleophobicity in air (Fig. 5f).73 Inspired by Tuteja's work, recently, many superoleophobic surfaces were constructed using the re-entrant surface curvature factor.12,109 For example, Zhao et al. created a micro-pillar array structure on a silicon substrate through the Bosch etching process (Fig. 5g).103 The pillars were 7 μm in height and 3 μm in diameter. The interval of the array was 6 μm. In particular, the vertical side wall of the pillars was not smooth, but consisted of a nanoscale wavy structure (∼300 nm) from top to bottom. After coating with a fluorosilane molecular layer, the resultant sample exhibited superoleophobicity. The measured OCA value was 158° and the OSA was as small as 10° to a hexadecane droplet. Both the coated chemical layer and the microstructure of the pillar array were indispensable elements for achieving such superoleophobicity. Im et al. fabricated an inverse-trapezoidal microstructure array by using the backside three dimensional (3D) diffuser lithography and template replication method (Fig. 5h).104 After coating a layer of Teflon, the surface became oleophobic with OCA of 135° for a methanol droplet (γ = 22.5 mN m−1). Yuan et al. transferred T-shaped overhang microstructures from Si substrate to various curable materials (such as polydimethylsiloxane (PDMS), polymethyl methacrylate (PMMA) and glass resin) by the multiple soft replication method (Fig. 5i).105 Both the CA values of a water droplet and a hexadecane droplet on those surfaces were larger than 150°, indicating that the fabricated surfaces had simultaneous superhydrophobicity and superoleophobicity. Ahuja et al. prepared a silicon nanonail array with re-entrant structure by reactive ion etching (Fig. 5j).106 Each nanonail was composed of a stem with diameter of about 280 nm and height of 7 μm and a head with diameter of 405 nm and thickness of 125 nm. The nanonails were arranged in a square array, and the interval of the nanonails was 2 μm. The as-prepared surface displayed superoleophobicity for various liquids with surface tension ranging from 21.8 mN m−1 (ethanol) to 72.0 mN m−1 (water). By designing an opportune rough topography with overhanging microstructures, it can even make the oleophilic materials repel low-surface-energy oils and organic solvents. However, these re-entrant microstructures cannot stop being wetted by the liquids with extremely low surface tension (i.e., γ < 15 mN m−1), such as fluorinated solvents, which even thoroughly wet most hydrophobic materials. Recently, Kim et al. pointed out that the surface with the doubly re-entrant structure is more powerful than the surface with the single re-entrant structure to repel liquids as well as achieve superomniphobicity.107 They further fabricated an array of doubly re-entrant structures, which were composed of microscale posts with nanoscale vertical overhangs, as shown in Fig. 5k. Without any chemical modification, the as-prepared surface super-repelled all available liquids, including various oils, organic solvents, and even fluorinated solvents (for instance, perfluorohexane, γ < 10 mN m−1). When a completely wetting liquid contacted the doubly re-entrant structure, the liquid first wetted the top of the micro-posts and then went downward along with the vertical overhangs. The liquid would stop advancing once it reached the vertical overhangs' bottom tip because the direction of surface tension switched to upward at this location, forming a stable liquid suspension on the rough microstructure. Therefore, the doubly re-entrant texture can provide a stronger resistance against wetting.
It is worth noting that the re-entrant texture is not limited to a microstructure with overhang. In fact, some materials such as rough metal meshes,75,108,187 electrospun fibers,120,188,189 fabrics,190 and porous structures96,118,119,124,191 can also be viewed as the analogues with re-entrant surface curvature because these microstructures have a certain degree of re-entrances. Tuteja et al. coated stainless steel meshes with electrospinning microbeads (PMMA + fluorodecyl POSS blend) and obtained re-entrant texture on both the macroscale and microscale (Fig. 5l).108 The combined effect of hierarchical texture and low solid surface energy endowed the rough mesh with superoleophobicity (inset of Fig. 5l). An n-heptane droplet on the sample surface showed an advanced OCA of 155° and an ultralow CA hysteresis of 4°. Choi et al. fabricated a poly(2,2,2-trifluoroethyl methacrylate) fiber web through the typical electrospinning method.189 The web repelled both water and oil (hexadecane) droplets whose CAs were 153° and 154.2°, respectively. Yang et al. obtained a suspension of copper perfluorooctanoate based on the chemical reaction between perfluorooctanoic acid and copper acetate in water, and then sprayed the suspension onto various substrates, such as glass, copper, steel, etc.191 In addition to the hierarchical textured microstructure, there were abundant protrusions and pores on the coating surface. Those protrusions and pores generated a large number of re-entrant geometries. A composite solid/air/oil interface (Cassie state) was formed when an oil droplet was put on the resultant samples, resulting in robust superoleophobicity of the uniform coatings.
Furthermore, it should be noted that although the re-entrant surface curvature is especially helpful to fabricate superoleophobic surfaces (in air), it is a necessary but insufficient condition.
3.2 Superamphiphobicity
Since Tuteja et al. in 2007 revealed the importance of re-entrant surface curvature for achieving superoleophobicity,72 it is well recognized that the synergy between the chemical composition with ultralow surface energy (mainly –CF3 compounds) and the appropriate surface roughness (including re-entrant microstructures) is the most promising route to fabricating in-air superoleophobic surfaces.3,12,72,73,102–108 Considering the fact that most oils have smaller surface tension than water, the superoleophobic surfaces in air usually repel water also. Until now, various kinds of surfaces showing both superoleophobicity and superhydrophobicity have been reported.12,109,117–119,192–206 The nature of such surfaces is defined by the terminology “superamphiphobicity”.12,14,109 All the superoleophobic surfaces with re-entrant microstructures, in Section 3.1, simultaneously exhibit superhydrophobicity in air. Therefore, those re-entrant texture-structured superoleophobic surfaces are actually superamphiphobic. The following will show some other examples of superamphiphobic surfaces.Jiang et al. endowed common engineering metals (e.g., zinc, iron, Zn–Fe alloy, aluminum, and nickel) with superamphiphobicity by a simple electrochemical method, as shown in Fig. 6a–c.192 Taking pure zinc sheet as an example, the cleaned zinc plate was directly immersed into the perfluorocarboxylic acid (such as nonadecafluorodecanoic acid) in ethanol solution under ambient conditions. Zinc is a very active metal and it can be easily oxidized to Zn2+ ions by oxygen, especially in acidic solution. After immersing the zinc sheet in perfluorocarboxylic acid solution, the Zn2+ ions were released from the zinc plate. Then, the Zn2+ ions reacted with perfluorocarboxylic acid molecules to produce zinc perfluorocarboxylate, which could slowly adsorb on the zinc substrate. After 10 days of chemical reaction, petal-like nanosheet structures were created on the zinc sheet (Fig. 6a). The length and height of the nanosheets were in the scale of micrometers, while their thickness was only thirty to several hundred nanometers. All the petal-like nanosheets stood on the substrate. The tops of the micrometer-long sheets were interspersed with lots of fine nanostructures, forming a micro/nanoscale hierarchical rough structure. The nanosheet structured surface super-repelled both water and oil without any further surface modification (Fig. 6b). Water droplets on such a surface exhibited the WCA of 158.1° ± 5° (Fig. 6c) and could easily roll off the tilted substrate with the water SA (WSA) of 5°. As for rapeseed oil, the OCA and OSA were 155.6° ± 1° (Fig. 6c) and 10°, respectively. The superamphiphobicity was the direct result of the synergistic effect of the unique surface chemistry and petal-like microstructure. The perfluorocarboxylic acid with high content of –CF3 and –CF2 groups provided very low surface energy, while the rough microstructure could trap air in nanoscale concave cavities and resulted in a steady air cushion which dramatically reduced the contact area between water (oils) and the as-prepared surface, then enlarged the water- and oil-repellence.
 | ||
| Fig. 6 Various superamphiphobic surfaces. (a) Petal-like nanosheet structures on the zinc sheet. (b) Photo of a water droplet and a rapeseed oil droplet on the rough zinc sheet. (c) Shapes of the water droplet (left) and rapeseed oil droplet (right) on the rough zinc sheet. (d) Cross-sectional SEM image and (e) superamphiphobicity of the nanoparticle aerosols self-assembled surface. (f) Textured needle after the self-assembly of nanoparticle aerosols onto its inner and outer surfaces. Inset: The superamphiphobic injector could generate and release nanoliter liquid droplets. (g and h) SEM images of nanocomposite coating. (i and j) Hierarchical rough TiO2/Ti microstructure fabricated by double anodization: (i) low magnification, depicting the microscale structure; (j) high magnification, depicting the nanotubes on the microscale structure. (k) Photo of glycol, glycerol, CH2I2, and hexadecane droplets on the micro/nanostructured TiO2 nanotube arrays. (l) SEM image of PPy–FAS coated fabrics. The inset is the cross-sectional transmission electron microscopy (TEM) image of the coated fiber. (m) Hexadecane (red, left) and water (green, right) droplets on the PPy–FAS coated fabric. (a–c) Reproduced from ref. 192 with permission from ACS, copyright 2008. (d and e) Reproduced from ref. 193 with permission from ACS, copyright 2016. (f) Reproduced from ref. 194 with permission from Wiley, copyright 2017. (g and h) Reproduced from ref. 118 with permission from ACS, copyright 2009. (i–k) Reproduced from ref. 195 with permission from ACS, copyright 2010. (l and m) Reproduced from ref. 196 with permission from RSC, copyright 2011. | ||
Wong et al. recently fabricated flexible and transparent superamphiphobic surfaces by the large-scale omnidirectional self-assembly of nanoparticle aerosols.193 They lit a combustible liquid solution containing hexamethyldisiloxane to generate a flame. With a substrate being placed above the flame, the tailored aerosol of nanoparticles could easily be deposited onto the substrate surface by a highly scalable flame aerosol technique. The resultant nanostructures formed by aerosol deposition of SiO2 nanoparticles looked very much like broccoli plants, having a visible re-entrant cross-section (Fig. 6d). The sample with the self-assembled deposition time of 15 s and further fluorogroup modification exhibited superamphiphobicity with OCAs above 160° for a large number of oils and low-surface-tension liquids (as low as 25 mN m−1), as shown in Fig. 6e. Furthermore, the highly dewetting nanotextures could be created on an extensive set of material surfaces with complex geometries such as curved surfaces and the inner walls of tubes. For example, after the self-assembly of nanoparticle aerosols on the inner and outer surfaces of a commercial hypodermic needle, the textured needle became superamphiphobic (Fig. 6f).194 The obtained superamphiphobic injector was capable of generating and releasing ultrasmall nanoliter droplets (down to 10 nL) of water and oils. There was no contamination of such needle during handling water and oil droplets in air or in liquid environments.
Steele et al. reported a spray coating method to realize superamphiphobicity on various substrates by using nanoparticle-polymer suspensions, as shown in Fig. 6g and h.118 The ZnO particles with diameter of 50 nm were mixed with waterborne perfluoroalkyl methacrylic copolymer (PMC) (30 wt% polymer, 70 wt% water) and acetone, forming a nanocomposite solution. Among them, PMC was a low-surface-energy binder; ZnO nanoparticles were used to create nano-roughness; acetone was the cosolvent. The solutions were sprayed onto solid substrates using an airbrush atomizer, and then dried for 12 h in air. For a hexadecane droplet, the fabricated thin coatings showed OCA of 154° and CA hysteresis of 6°. The hexadecane droplet rolled almost freely on the as-prepared sample surface, as long as the substrate was slightly tilted. No oily trailing stain was left behind, and this was very similar to the process of a water droplet rolling on a lotus leaf. The coatings also exhibited excellent superhydrophobicity but not superoleophobicity. The superamphiphobicity was the result of water droplets and oil droplets on such resultant coatings all being in the Cassie state.
Wang et al. used two routes to build micro/nanoscale hierarchical structures on Ti substrate and successfully obtained superamphiphobic surfaces, as shown in Fig. 6i–k.195 One route was double anodization and another was a combination of laser ablation and anodization. For the former route, a cleaned Ti plate was firstly electrochemically etched in a NaCl solution (0.1 M) at the current density of 500 mA cm−2 for 1 h to form microscale structures (Fig. 6i). Then, the microstructured Ti substrate served as a working electrode and was anodized in an electrolyte (0.5 wt% NH4F, 3% hydrofluoric acid, and ethylene glycol) at 60 V for 1 h, using a graphite slice as a counter electrode. A lot of TiO2 nanotubes were created on the top surface of the microstructure of the Ti substrate during the second anodization process (Fig. 6j). After the modification of 1H,1H,2H,2H-perfluorooctyltrichloro silane, the as-prepared surface showed both superhydrophobicity and superoleophobicity to various liquids (Fig. 6k). For example, the measured CAs and SAs are 167° and <2° for water, 167° and <2° for sea water, 162° and 3° for glycerol, 160° and 3° for liquid paraffin, 156° and 6° for crude oil, 157° and 5° for colza oil, and 155° and 7° for hexadecane, respectively. For the latter route, the only difference from the former route was that the microstructures were generated by laser ablation in the first step. In this case, the microstructures could be designed into different patterns including the line pattern, salient pattern and pit pattern. Superamphiphobicity was also obtained through the latter route.
Wang et al. developed a one-step method to fabricate superhydrophobic and superoleophobic fabrics based on vapour-phase polymerization, as shown in Fig. 6l and m.196 The fabric substrates were firstly dipped into the ethanol solution of FeCl3 and fluorinated alkyl silane (FAS). After extracting the fabrics from the solution and drying them out, the fabrics were further placed into a pyrrole saturated nitrogen atmosphere to form polypyrrole (PPy). Finally, the fabrics became black, revealing that a layer of PPy–FAS was coated on the original fabric surfaces (Fig. 6l). The coated fabrics not only exhibited superamphiphobicity (Fig. 6m) with the CAs of 165° to water and 154° to hexadecane, but also had high electrical conductivity with a surface resistivity of 0.5–0.8 kΩ □−1.
Darmanin et al. obtained a series of electrodeposited polymers from the pre-synthesized fluorinated 3,4-ethylenedioxypyrrole monomers.119 The polymer film showed superoleophobicity and superhydrophobicity with ultralow adhesion to both water and oil droplets. The superamphiphobicity resulted from the nanoscale porosity of the electrodeposited polymer, which was formed during the doping process. Srinivasan et al. sprayed a blend of fluorodecyl POSS and PMMA on the substrate and achieved superhydrophobicity and high oleophobicity.197 The surface microstructures could be systematically changed from randomly distributed spherical microbeads to beads-on-string structures to fibers by varying the polymer molecular weight and the solute concentration. Zhang et al. placed a stainless steel mesh above the flame of a candle to deposit a thin layer of soot.100 The soot was piled with a large amount of carbon nanoparticles with the size ranging from 30 nm to 60 nm, resulting in a porous microstructure. Then, a shell of silica was further deposited on the soot layer by chemical vapor deposition (CVD) of tetraethoxysilane, using ammonia as a catalyst. The silica/soot coated mesh was hydrophilic and became superamphiphobic after tridecafluorotetrahydrooctyl trichlorosilane modification.
Superoleophobic surfaces usually super-repel water droplets as well, because the surface tension of water is much higher than that of various oils; i.e., most of the superoleophobic surfaces (in air) are superamphiphobic.12,109 However, it should be noted that not all the superoleophobic surfaces also have superhydrophobicity. There are also some superoleophobic–superhydrophilic artificial materials.103,205
3.3 Superoleophobic–superhydrophilic surfaces
Recently, through designing and modifying special chemical chains on the surfaces with hierarchical rough microstructures, some surfaces simultaneously showing superoleophobicity and superhydrophilicity in air have been fabricated.103,205 Yang et al. reported a superoleophobic–superhydrophilic coating by the spray-casting of the nanoparticle-polymer suspension.205 Nanoscale SiO2 particles (0.3 g) were firstly dispersed in 100 mL of poly(diallyldimethylammonium chloride) (PDDA) aqueous solution with the concentration of 1 mg mL−1. Then, 20 mL of sodium perfluorooctanoate (PFO) aqueous solution (0.1 M) was further poured in. With constant ultrasonic shaking, the PFO anions could be grafted onto the quaternary ammonium groups of PDDA (Fig. 7a). After removing the water, the dried composite material (PDDA–PFO/SiO2) was ultrasonically dispersed in ethanol. The obtained suspension was sprayed onto various substrates using a spray gun. The PDDA–PFO/SiO2 was tightly coated on the substrate, with the ethanol evaporating completely at room temperature. The PDDA–PFO/SiO2 coating had both oleophobic fluorinated groups and hydrophilic carboxyl/quaternary ammonium groups, endowing the resultant polymer surface with simultaneously oleophobic and hydrophilic properties. In addition, the mixed SiO2 nanoparticles led to a hierarchical rough structure, which significantly amplified the oleophobicity and hydrophilicity. The surface showed a textured microstructure of abundant protrusions and pores, which is a representative morphology of the sprayed coating. When a water droplet and a hexadecane droplet were put on the coated substrate, they initially showed spherical shapes. However, the water droplet slowly collapsed and spread over the sample surface within 9 min, with the WCA decreasing from 165° ± 2° to 0°. This is because that water droplet resulted in a rearrangement of the surface molecular structure, and the capillary effect helped the water to fully wet the space of rough microstructures; in contrast, the oil droplet had almost no change in shape. The OCA remained larger than 155° ± 1° all the time, therefore, the PDDA–PFO/SiO2 coating exhibited simultaneous superhydrophilic and superoleophobic wetting behaviors (Fig. 7b and c). If the coating was sprayed on metal meshes, the meshes with both superhydrophilicity and superoleophobicity could be applied in separating the water/oil mixtures (Fig. 7d and e). During the separation process, water penetrated through the mesh by gravity because of the superhydrophilicity (Fig. 7b), while oils were stopped and retained on the mesh surface because of the superoleophobicity (Fig. 7c). The anti-oil property prevented the rough mesh from fouling or even being blocked by oily liquids, which extended the service life of the separation devices.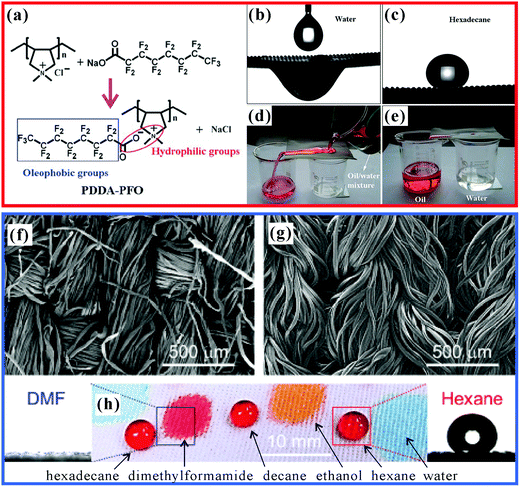 | ||
| Fig. 7 Superoleophobic–superhydrophilic surfaces. (a) Chemical reaction for synthesizing PDDA–PFO. (b) Water droplet wetting and permeating the PDDA–PFO/SiO2-coated mesh film. (c) Hexadecane droplet on the resultant mesh. (d and e) Separating the mixture of water and oil by using the PDDA–PFO/SiO2-coated mesh. (f and g) SEM images of the superoleophobic (f) cotton fabric and (g) polyester fabric. (h) Nonpolar oils beading up and polar liquids permeating through the coated fabric. (a–e) Reproduced from ref. 205 with permission from RSC, copyright 2012. (f–h) Reproduced from ref. 206 with permission from the American Institute of Chemical Engineers, copyright 2014. | ||
Pan et al. changed the intrinsically superoleophilic cotton fabric and polyester fabric surfaces to both superoleophobic and superhydrophilic by a simple fluorosilanization treatment, as shown in Fig. 7f–h.206 (Tridecafluoro-1,1,2,2-tetrahydrooctyl)triethoxysilane was used because this fluorosilanization had a low surface energy (∼11.5 mN m−1), yielding excellent oil resistance to various low-surface-tension hydrocarbons. The combined effect of the intrinsic re-entrant porous microstructure and chemistry coating with ultralow surface energy resulted in the CAs higher than 150° for various nonpolar hydrocarbons (Fig. 7h). For instance, hexane and heptane could easily bead up on the resultant fabrics and these oil droplets were at the Cassie wetting state. In a surprise twist, if a polar liquid droplet (such as water, dimethylformamide and ethanol) were dripped onto the fabric surfaces, the droplet would rapidly spread out and be absorbed by the fabrics, resulting in CAs of about 0° (Fig. 7h). In fact, the intrinsic intact cellulose chains in cotton fabrics could migrate to the outside surface, so both the fluorine and intact cellulose chain covered the surface of the as-prepared fabrics. According to the basic principle of “like dissolves like”, the abundant intact cellulose chains promoted the contact between fabric surface and polar liquids, making the fabrics superhygrophilic for polar liquids.
It is worth noting that these superoleophobic–superhydrophilic surfaces possess both oil-repellent (e.g., fluorinated chains) and hydrophilic (e.g., carboxyl/quaternary ammonium groups, cellulose chains, and other polar groups) chemical compositions. The micro/nanoscale hierarchical rough structures only amplify the surface wettability to superoleophobicity and superhydrophilicity. When an oil droplet is put on the resultant surfaces, the oleophobic chemical compositions and rough microstructures play a dominant role; in contrast, when a water droplet is released onto the resultant surfaces, the hydrophilic compositions will be attracted by the polar liquid (water) and reduce the presence of the fluorinated chains at the extreme surface (surface re-arrangement), leading to superhydrophilicity.205–207
3.4 Slippery liquid-infused porous surfaces
Unlike the aforementioned superoleophobic surfaces, which usually use special rough microstructures to repel oil wetting,3,12,14,109 the Nepenthes pitcher plant (Fig. 8a) uses microstructures to lock-in an intermediary liquid.208 The filled liquid within the rough microstructures generates a continuous trapped liquid cushion that can act as a repellent surface (Fig. 8b). When an insect alights on the rim of a pitcher plant, the insect can hardly stand on and will slide down from the rim to the bottom, which is filled with digestive juices, because the trapped liquid cushion repels the oils on the insect's feet.209 Inspired by the pitcher plant, Wong et al. recently presented a new way to create liquid slippery surfaces, named “slippery liquid-infused porous surface (SLIPS)”.208 The CAs of both oil droplets and water droplets on such surfaces were far smaller than 150° (i.e., the surface was not superoleophobic), but the surface exhibited excellent liquid repellence. Therefore, we are more inclined to categorize the SLIPS into the superoleophobic materials, or we can consider that it is a broader superoleophobic interface. As shown in Fig. 8c, a surface with micro/nanoscale porous structure was pre-infused with a perfluorinated lubricating liquid, resulting in the lubricating fluid being locked in the rough microstructures to form the SLIPS. The SLIPS created a smooth, slippery, and stable interface that significantly decreased the contact line pinning effect for various liquids. The lubricating liquid layer could also resist a very high pressure, so foreign liquids would not easily lodge in the porous microstructures. The SLIPS showed very low adhesion to a variety of liquids with surface tension ranging from 17.2 mN m−1 (n-pentane) to 72.4 mN m−1 (water). All the droplets of various liquids could easily slide on a 5° tilted sample, without leaving any stain. The SLIPS effectively repelled not only pure liquids, but also compound fluids, e.g., crude oil (Fig. 8d) and blood (Fig. 8e); these compound liquids generally wet and contaminate most solid surfaces. In addition, the SLIPS could serve as a slippery, anti-sticking surface for insects, like pitcher plants (Fig. 8f). Other properties like self-healing and optical transparency endow the liquid-repellent SLIPS with wider application prospects, such as biomedical fluid handling, anti-fouling, fuel transport, optical devices, and self-cleaning windows.208 To design a liquid slippery surface, attention should be paid to three criteria:208,210–213 (1) the used materials have porous surface microstructures, which ensure that the lubricating liquid wets and is trapped in the porous microstructures; (2) the adopted lubricating liquid has a stronger ability to wet the solid substrate than the liquid we want to repel; (3) the lubricating liquid must be practically immiscible with the repelled liquids. In addition, the interconnectivity of the micro-pores is also very important to slippery surfaces, although slippery surfaces can be fabricated as long as the above-mentioned three criteria are met. Once the artificial SLIPS is damaged by cutting/abrasion, the interconnectivity of the micro-pores allows the lubricating liquid trapped in the micro-pores surrounding the damaged area to flow freely to the damaged area and re-infuse the physical voids, thereby self-repairing the damaged SLIPS. This process can be spontaneously driven by surface-energy-induced capillary action, without any external force. Therefore, if the micro-pores of the SLIPS are interconnected, the SLIPS will have self-repairing function.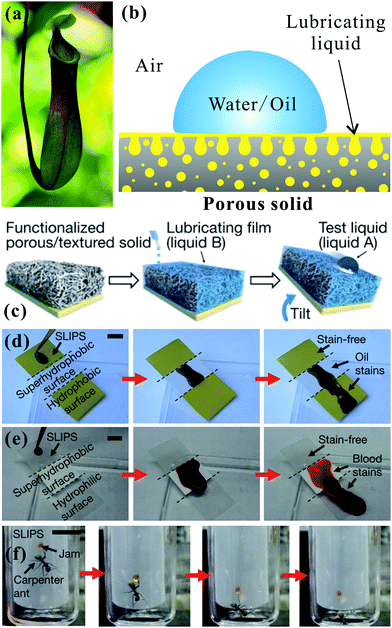 | ||
| Fig. 8 Liquid repellence of slippery surface. (a) Photo of a pitcher plant. (b) Wetting schematic of a SLIPS. (c) Schematic diagram of fabricating a slippery surface. (d) Light crude oil and (e) blood sliding down a substrate composed of three different regions (from top to bottom: SLIPS, superhydrophobic porous Teflon film, and flat hydrophobic surface). A stain was left on the latter two regions. (f) Inability of an ant to stand on or adhere to the SLIPS. The scale bars in (d–f) are 1 cm. Reproduced from ref. 208 with permission from the NPG, copyright 2011. | ||
Recently, Manna and Lynn fabricated slippery surface coatings by a layer-by-layer (LBL) assembly method and oil infusion.210 Sample substrates (e.g., glass, aluminum foil, wire meshes, filter paper, and so on) were firstly immersed in an acetone solution of branched poly(ethyleneimine) (PEI) (0.02 M) for 20 s. Then, the samples were taken out and dipped into two pure acetone baths for 20 s in turn. The substrates were further immersed in an acetone solution of poly(vinyl-4,4-dimethylazlactone) (PVDMA) (0.02 M) for 20 s. Next, the samples were rinsed again by using two pure acetone baths. The abovementioned process was repeated for many cycles to form porous PEI/PVDMA polymer multilayers. Finally, the obtained nanoporous PEI/PVDMA coatings were functionalized by the reaction between surface azlactone groups and n-decylamine to graft hydrophobic groups onto the coating surfaces. After the infusion of oil (e.g., silicon oil) into the nanopores of the coatings, the resultant SLIPS exhibited strong liquid-repellent ability, due to the formation of liquid/liquid interfaces. A variety of chemically complex liquids, such as acidic/alkaline solutions, serum (in cell culture medium), lake water, and ketchup, could easily slide down when the substrate was slightly inclined. Even when the sample was immersed in seawater for 2 months or severely abraded by sandpaper, the slippery property was still maintained. In addition, the authors modified some regions of the original porous PEI/PVDMA multilayers with hydrophobic groups and other regions with hydrophilic groups to obtain a “sticky” patterned oil-infused SLIPS. The sticky regions did not lock silicon oil in the nanopores; thus, these regions could hinder or even stop the sliding of aqueous droplets. The patterned oil-infused SLIPS was successfully applied in extracting small liquid samples from contacting media and guiding the direction of the sliding droplets.
Endoscopes are widely used in medical examination or operations with many advantages, such as reducing the size of the required incision, shortening the surgery time, relieving patients' pain, and accelerating quick post-surgery recovery. However, current endoscopes are easily contaminated by blood and other body fluids during the operation, resulting in an obstructed vision. Nishioka et al. developed a facile method to prepare fluorine-free SLIPS by the self-assembly of methyl group modified silica nanoparticles.211 Plant oil was locked into the microstructures as the lubricant because of its edible and eco-friendly properties. The aggregation of the self-assembled hydrophobic silica nanoparticles generated a micro/nanoscale binary rough structure, which could firmly trap the lubricant even under flowing water. The plant oil SLIPS repelled not only aqueous liquids with differing pH, but also biological fluids such as blood. More specially, the as-prepared SLIPS also showed high transmittance. In addition, it was found that the SLIPS had an anti-coagulation effect. When a bare endoscope was used to observe the abdominal cavity of a mouse, the vision was easily blurred, especially around blood vessels. In contrast, when a SLIPS coated endoscope lens was applied to inspect the internal organs of a mouse, the visibility through the endoscope was very clear all the time. The comparison result was still valid for observing the inner side of the mouse stomach. The SLIPS film did not weaken the visibility after covering the endoscope lens, but it could prevent blood and other body fluids from adhering to the lens surface.
3.5 Controllable oil-adhesion
Controllable liquid adhesion endows the target surfaces with many important applications, such as microdroplet manipulation, microfluidic chips, and cell engineering.51–53,55,80,91,112,124,150,176,214,215 Superhydrophobic surfaces showing controllable adhesion have been widely achieved by various methods.54,99,112,155,177,179,216–220 For example, Li et al. fabricated distinguishing textured superhydrophobic CuO surfaces through the solution-immersion method.177 The fluoroalkylsilane modified surfaces with different topographies exhibited different water adhesion that ranged from low to very high. Our group used the femtosecond laser selective ablation method to build superhydrophobic patterned PDMS surfaces composed of both an ultralow adhesive superhydrophobic domain and an ultrahigh adhesive weak hydrophobic domain.53 The water adhesion could be easily controlled via changing the relative area fraction of laser irradiated (ultralow adhesion) and nonstructured flat (ultrahigh adhesion) domains. Around the same time, a variety of superhydrophobic surfaces with controllable water adhesion were designed using a femtosecond laser.54,179,215,219,221 Lai et al. reported that the adhesion of superhydrophobic nanotube-array surfaces to a certain water droplet could be controlled by the length and diameter of the nanotubes,216 which resulted from the strength change of the “micro-airbag effect”.221Compared to superhydrophobic substrates with different water adhesion, the superoleophobic surfaces showing controllable adhesion to oil droplets are much more practical because both water and oil cannot wet the surfaces;89,124 however, so far, controllable oil-adhesive surfaces are still rarely reported. Yao et al. achieved controllable oil-adhesion on superoleophobic Cu(OH)2 surfaces by changing the surface morphology.89 A ribbed nanoneedle array Cu(OH)2 surface was first obtained by immersing copper foil into ammonia solution (0.03 M) for 36–48 h (Fig. 9a). Then, the rough Cu(OH)2 foils were dipped into the 0.01 M ethanol solution of 1H,1H,2H,2H-perfluorodecanethiol (C2H4C8F17–SH) for different times (Fig. 9b–d). The latter process not only reduced the surface free energy, but also changed the microstructures of the obtained rough Cu(OH)2 surfaces. With increasing the reaction duration from 1 h to 36 h, the nanoneedles became shorter and denser (Fig. 9b–d). The OCA values against an oil (n-hexadecane) droplet were maintained above 150° for different immersing times, revealing superoleophobicity, while the adhesive force between the resultant surface and oil droplet changed from ∼36 μN to 5.9 μN (Fig. 9e). The controllable oil-adhesion mainly resulted from the evolution of surface microstructures. A substrate with shorter and denser nanoneedle array had a smaller contact area with oil droplets, and thus had smaller oil-adhesion. In addition, the authors further found that the oil-adhesion also depended on the hydrostatic pressure (Fig. 9f). Using the original ultralow adhesive superoleophobic surface (reaction time = 36 h) as a study subject, the oil-adhesive force gradually increased from 5.9 μN to 42 μN as the preload force increased from 0 μN to 105 μN. The external hydrostatic pressure made the oil droplet partly penetrate into the space of rough microstructures of the resultant surface, leading to a penetrating Cassie state (transition wetting state). With the preload force increasing, the solid–oil contact area increased; thus, the oil-adhesion was greatly enhanced. Based on the controllability of oil-adhesion, an oil droplet-based microreactor was presented via the superoleophobic surface (see Section 6.4 for details).89
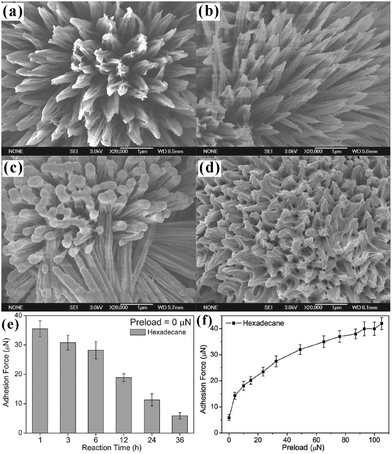 | ||
| Fig. 9 Variation of the oil-adhesion of the superoleophobic Cu(OH)2 nanoneedle structured surfaces with the reaction time (in C2H4C8F17–SH solution) and the applied preload force. (a) SEM image of the obtained Cu(OH)2 nanoneedle structured surface at first. (b–d) SEM images of the resultant rough substrates with different reaction durations: (b) 12 h, (c) 24 h, and (d) 36 h. (e) Relationship between the oil-adhesive force and the reaction time on the resultant surfaces. (f) Oil-adhesive force for the superoleophobic surface (reaction time = 36 h) under different applied preloads. Reproduced from ref. 89 with permission from Wiley, copyright 2011. | ||
Yang et al. fabricated superoleophobic surfaces by depositing polyelectrolyte multilayer films (PMFs) and PFO anions on rough aluminum substrates.124 An aluminum sheet was firstly immersed in an HCl solution (2.5 M) for 10 min and subsequently dipped in boiling water for 30 min. Such treatments resulted in the formation of abundant distorted nanoflakes on the aluminum surface. The rough aluminum surface initially showed superhydrophilicity and superoleophilicity. Interestingly, it switched to superhydrophobic and superoleophobic after depositing PMF layers and PFO anions. The measured CAs of various droplets, including water, rapeseed oil, glycerol and hexadecane on the resultant surface were larger than 150°. With increasing the time of the deposition process of the PMF/PFO layer (i.e., increasing the number of the polyelectrolyte bilayers), they found that the adhesion of the resultant superoleophobic surfaces to various oil droplets increased from low to high; i.e., the liquid droplet changed from the rolling state to the pinned state. The enhancement of oil-adhesion was mainly caused by the change in surface microstructure. With the polyelectrolyte being continually deposited on the rough aluminum substrate, the nanoflakes began to thicken and were finally buried, and the pores were gradually filled. The decrease in surface roughness caused the wetting state to turn from the Cassie state to the transition state, and therefore, the actual solid–oil contact area expanded. As a result, the liquid adhesion gradually increased. They further demonstrated that the controllable oil-adhesive superoleophobic surfaces had the function of selecting and transporting microdroplets.
3.6 Anisotropic oil-wettability
The liquid droplet on an anisotropic textured surface exhibited clearly unequal CAs and SAs in different directions, usually named anisotropic wettability.154,156,214,222–225 This unique property has many important practical applications, such as self-cleaning, liquid pumps, microfluidics, and smart device designs.36–39,43,84,127 Although the anisotropic wettability of a water droplet has been successfully obtained on various textured substrates in atmosphere,32,54,226–234 there are still significant challenges for achieving anisotropic oil-wettability because the organic liquids with lower surface tension have very different wetting behavior, compared to water. Zhao et al. fabricated a microgroove array on Si substrate by the Bosch deep reactive ion etching process, as shown in Fig. 10a and b.125 The height, width and center-to-center spacing of the grooves were 4 μm, 3 μm and 6 μm, respectively. There were many side-by-side nanogrooves with the width of about 300 nm on the side wall (Fig. 10b). After etching, the sample was self-assembled with a molecular layer of tridecafluoro-1,1,2,2-tetrahydrooctyltrichlorosilane through a vapor deposition process. The resultant textured surface was found to exhibit anisotropic wettability for not only water droplets but also oil droplets (Fig. 10c). The static OCA of an oil (hexadecane) droplet on the resultant microgrooves array was 113° in the parallel direction and 162° in the perpendicular direction. The maximum OCA difference reached up to 49°, presenting obvious anisotropic oil-wetting. As for the dynamic aspect, the CA hysteresis in the parallel direction was 19°, while it increased to 66° in the perpendicular direction. The OSA was 8° in the parallel direction and 34° in the perpendicular direction. The discrepant CA hystereses and OSAs suggested that oil droplets preferred to move along the microgrooves (parallel direction).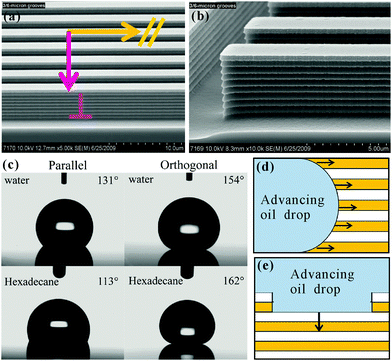 | ||
| Fig. 10 Anisotropic oil-wettability of the microgroove textured Si surface. (a and b) SEM images of the microgroove array. (c) Shapes of water and oil (hexadecane) droplets on the microgroove array in the parallel direction and the perpendicular (orthogonal) direction, respectively. (d and e) Cartoon schematic of an oil droplet moving forward along the (d) parallel and (e) perpendicular directions, respectively. Reproduced from ref. 125 with permission from ACS, copyright 2012. | ||
The anisotropic wettability is the direct result of the anisotropic textured microstructures. When a liquid droplet moved forward along the parallel direction, 50% of the bottom of the droplet was in contact with the solid strips of the microgroove array and another half was held up by air, which was filled in the valley of the microgrooves (Fig. 10d). The solid strips could be wetted by oil preferentially, which reduced the energy barrier for this droplet to advance. The spread of the droplet along the strips was then enhanced. However, in the perpendicular direction, the oil droplet underwent full solid for half the time and air for half the time; that is, the oil droplet “jumped” from one strip to next one (Fig. 10e). The TPCL was generally pinned at the re-entrant microstructures in the sidewall of the microgrooves. With the oil moving forward, the TPCL should first be unpinned from the re-entrant microstructures. The unpinning process of the “stepped” TPCL caused high surface friction;235,236 therefore, the droplet mobility in the perpendicular direction was quite difficult.
3.7 Transparency
Transparency is very important for superoleophobic materials being applied as oil-repellent optical devices, such as goggles, self-cleaning solar panels, touch screens, difficult-to-access windows, and so on.117,120,121,128,237,238 However, most reported superoleophobic substrates are opaque, since the required rough micro/nanoscale structures for achieving superoleophobicity usually lead to strong light scattering. How to create transparent superoleophobic interfaces has always been a great challenge. In theory, the size of the rough microstructures should be smaller than the visible wavelengths to be optically transparent.239–247 Thus, all the length scales of microstructures for achieving transparent superoleophobicity must be less than 300 nm.Deng et al. presented a simple method to obtain transparent superamphiphobic coatings based on candle soot and shell deposition technology.117 A glass slide, as the substrate, was firstly suspended above the flame of a candle (Fig. 11a). The glass slide quickly turned black because a layer of soot was deposited on the glass surface. The soot layer was composed of abundant nanoscale spherical carbon beads. The diameter of the carbon particles was about 35 ± 5 nm. The particles chained together, forming a loose, fractal-like network. Although the soot network was rough enough to achieve superhydrophobicity, it was easily washed off due to the physical and weak particle–particle interaction. Next, the carbon network was switched to the more stable silica shell microstructure by using the soot layer as a template. The soot-coated substrates, a beaker of tetraethoxysilane (TES) and a beaker of ammonia were put in a desiccator together. After 24 hours, a silica shell with thickness of 20 ± 5 nm was coated on every soot particle through the CVD of TES under the catalyst of ammonia (Fig. 11b). The obtained compound carbon@silica network was further calcined at 600 °C for 2 hours to combust and remove the soot particles, leaving only the hollow silica shells on the substrate (Fig. 11c and d). After modifying the coating with low-surface-energy semifluorinated silane, both superhydrophobicity and superoleophobicity were obtained on the resultant surface (inset of Fig. 11e). Water droplets showed a WCA of 165° and a WSA of about 1° on such a surface. When various organic droplets were placed on the surface, the range of the measured OCA values was from 154° (tetradecane) to 162° (diiodomethane). All the oil droplets could easily roll off as soon as the sample was slightly tilted. The measured maximal OSA was 5° for tetradecane droplet (γ = 26 mN m−1). The silica network coating was also highly transparent, besides being superamphiphobic. After removing the soot core by calcination, the rest of the silica network became transparent because the shell thickness was significantly smaller than the wavelength of visible light. Taking the 3 μm-thick coating as an example, the UV-visible spectra revealed that the transmittance decreased less than 10% above 500 nm, compared with the flat bare glass slide (Fig. 11e). The letters on a white paper behind the coated glass slide were very clear (inset of Fig. 11e), revealing excellent optical transparency. Similar to, but beyond Deng's work,117 Wong et al. recently reported a flame spray pyrolysis method to enable the rapid, omnidirectional synthesis of flexible and transparent superamphiphobic microstructures, as shown in Fig. 6d and e.193 The candle was replaced by combustible liquid solutions containing hexamethyldisiloxane. Such a superamphiphobic surface with the deposition time of 15 s could exhibit up to 99.97% transparency.
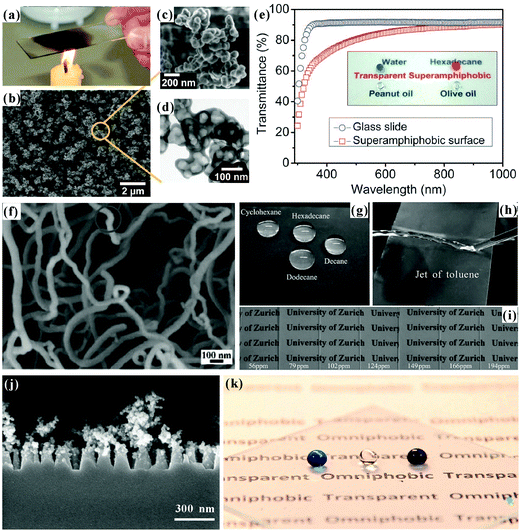 | ||
| Fig. 11 Transparent, superoleophobic surfaces in the atmosphere. (a) A glass slide is held above the flame of a candle to deposit a soot layer, which is composed of abundant nanoscale spherical carbon beads. (b) SEM image of the silica shell coated nanoscale soot particles. (c) High-resolution SEM image of the hollow silica shells after removing the carbon cores (soot particles). (d) TEM image of a cluster of hollow silica shells. (e) UV-visible transmittance spectra of the as-prepared surface and its original glass substrate. The inset shows the photo of a water droplet and three oil droplets on the coated glass slide. (f) SEM image of the TCMS/PFDTS-coated silicone nanofilaments. (g) Droplets of cyclohexane, hexadecane, dodecane and decane on the TCMS/PFDTS-coated surface. (h) Jet flow of toluene bouncing off the TCMS/PFDTS-coated glass slide. (i) Good transparency of a series of TCMS/PFDTS-coated glass slides. (j) Side-view SEM image of the rough glass surface with hierarchical nanostructures (nanopillars@nanobranching). (k) Photo of three liquid droplets (from left to right: oleic acid, water, and hexadecane) on the nanopillars@nanobranching structured glass slide. (a–e) Reproduced from ref. 117 with permission from AAAS, copyright 2012. (f–i) Reproduced from ref. 120 with permission from Wiley, copyright 2011. (j and k) Reproduced from ref. 128 with permission from ACS, copyright 2014. | ||
Zhang et al. fabricated a transparent superoleophobic coating by a simple grow-form approach and subsequent chemical modification.120 Trichloromethylsilane (TCMS) was firstly injected into toluene containing a very small amount of water (56–194 ppm). The hydrolysis of TCMS occurred due to the presence of water. Then, the dispersive TCMS quickly self-assembled into a crosslinked silicone nanofilament coating on the substrate surface (Fig. 11f). The diameters of the nanofilaments were in the range of 50–90 nm and the length reached up to several micrometers. The TCMS coating was further activated by O2 plasma irradiation and then modified with 1H,1H,2H,2H-perfluorodecyltrichlorosilane (PFDTS). The nanofilament skeleton remained intact during plasma irradiation and chemical modification. The water concentration in toluene during the hydrolysis and condensation of TCMS had a significant effect on the surface topography, oil wettability and transparency of the nanofilament coating. The TCMS/PFDTS-coated glass slides showed superoleophobicity and ultralow oil-adhesion when the water concentration ranged from 102 ppm to 194 ppm (Fig. 11g and h). The OCAs remained larger than 150° and OSAs remained smaller than 3° for an oil (hexadecane) droplet on various resultant surfaces. The oil droplet on the sample surfaces was in the Cassie state, because such a rough nanofilament network trapped much air beneath the oil droplet. In addition, the resultant superoleophobic coating showed an antireflective function for various transparent substrates in air (Fig. 11i). The nanofilaments TCMS coating could even improve the transmittance of the glass sheet to about 94% at the wavelength of 600 nm, while that of bare glass slide was only 91.2%. Although the transmittance had a small decrease after PFDTS modification, it retained above 82% for the resultant samples with the nanofilaments growing at water concentration <124 ppm. The letters on a paper behind the superamphiphobic glass slides could be clearly identified. The main reason for exhibiting good transparency is that light scattering was effectively decreased by the uniform growth of the nanofilaments.
Mazumder et al. prepared a superamphiphobic surface made of two-level nanostructures (Fig. 11j).128 An ultrathin layer of metal was firstly deposited on a glass surface. After the subsequent thermal annealing process, the metal film was split to disrupt nanoparticles. Next, a nanopillar structure with height of ∼200 nm and diameter of ∼100 nm was formed on the glass surface by ion etching, using the metal nanoparticles as a mask. The primary nanopillars were further covered with a layer of secondary branching nanostructures with length scale of 10–30 nm through the nanospray CVD method (Fig. 11j). All the samples were finally modified with fluorosilane. Water, oleic acid, and hexadecane droplets showed CAs of 113° ± 3°, 78° ± 2°, and 67 ± 2°, respectively, on the flat glass surface. Those values increased to 172° ± 4°, 163° ± 3°, and 153° ± 2° on the as-prepared rough substrate, meeting the criteria for superhydrophobicity and superoleophobicity (Fig. 11k). In addition, the surface revealed increased transmission (∼93.8% for visible light) and significantly reduced reflection (<0.5%), compared to the intrinsic bare glass (Fig. 11k).
It was reported that most natural and artificial superhydrophobic surfaces are vulnerable to losing their superhydrophobicity for hot water droplets.238,248 For example, the WCA of a hot water droplet (55 °C) on lotus leaves declined significantly to 40°, compared to 152° for a cool droplet (∼25 °C).248 This could be because (a) the condensation of water vapor between the superhydrophobic microstructures and the hot droplet increases the contact surface between liquid and solid materials; (b) the surface tension of water decreases with increasing temperature. Likewise, most of superoleophobic surfaces are also difficult to repel hot organic liquids. Recently, Li et al. generated a uniform silica nanotube coating on glass surfaces and obtained semitransparent superoleophobicity for hot oil droplets.238 The dispersion of PDMS-modified multiwalled carbon nanotubes (MWCNTs) was spray-coated onto a glass slide and then the MWCNT templates were removed by calcinations, leaving a layer of silica nanotube networks covering the glass surface. After lowering the surface energy with 1H,1H,2H,2H-perfluorodecyltrichlorosilane, the resultant coatings showed superoleophobicity to various oils (e.g., n-decane, n-hexadecane, toluene, etc.) and good transparency. Moreover, the transparent coatings still retained superoleophobicity at 75 °C.
3.8 Durability and self-healing
Most superhydrophobic or superoleophobic surfaces have poor durability so that their super-liquid-repellent ability is easily lost after physical rubbing, strong acid/alkali corrosion, strong light irradiation, oxidation in air, and so on.60,215,249–253 The poor durability greatly limits the application areas of those superhydrophobic or superoleophobic materials. Hence, improving the durability of superoleophobic surfaces is critical to their practical applications.117,120,128,206,238,254,255Chen et al. fabricated a kind of superamphiphobic Cu powder through a deposition method and subsequent chemical modification.130 The Al plate was firstly immersed in the 1 mol L−1 CuCl2 solution for 30 s, resulting in a thin Cu layer covering on the Al substrate, due to the chemical substitution reaction. The Cu-coated Al plate was washed with water to remove residual salts, and then the deposited Cu powder was peeled off by ultrasonic cleaning and collected. The separated Cu powder was further modified with perfluorooctanoic acid (CF3(CF2)6COOH) to reduce the surface energy. The powder particles had three typical microstructures: block, dendritic, and coralline-shaped structures with size ranging from several hundred nanometers to several tens of microns. The obtained powder showed both superhydrophobicity and superoleophobicity. The powder could be coated on various kinds of substrates (such as hard Al and glass slide, soft sponge, scouring pad, filter paper and rubber) using low-cost double sided tape or spray adhesives, making those materials superamphiphobic. The superamphiphobicity had no obvious decline even though the coated substrates suffered from immersion in water/acidic/alkaline/saline solutions, falling sand abrasion, UV irradiation, or 50 cycles of stretch-release (rubber substrate), respectively.
Lin et al. developed a simple method to obtain durable and self-healing superamphiphobic fabrics.121 The fluorinated-decyl POSS (FD-POSS, left in Fig. 12a) was firstly dissolved in FAS (right in Fig. 12a) (wt%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) wt% = 1
wt% = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). The viscous solution was further dissolved in ethanol. After ultrasonic treatment for 30 min, they obtained a homogeneous suspension that could be easily coated on a variety of fabrics by coating techniques (e.g., spraying or dip-coating), as show in Fig. 12b and c. The CAs of water, hexadecane, and tetradecane droplets on the FD-POSS/FAS coated fabrics were 171°, 155°, and 152°, respectively, showing both superhydrophobicity and superoleophobicity (Fig. 12d). When the coated fabric was irradiated by plasma to cause artificial damage, its wettability would switch from superamphiphobicity to both superhydrophilicity and superoleophilicity (Fig. 12e). Both water, hexadecane and tetradecane droplets presented CAs of 0° on the sample surface. Fortunately, the original super-liquid-repellency could recover after heating the plasma-irradiated fabrics at 135 °C for 3 min (Fig. 12f). The resultant fabrics could maintain the superoleophobicity even after 40 cycles of alternate plasma irradiation and heating, and maintain the superhydrophobicity for 100 cycles, demonstrating strong self-healing ability. Likewise, the superamphiphobicity would be lost if the coated fabrics were etched by strong alkali (e.g., KOH solution with pH = 14) and would be re-obtained by heat treatment. On the other hand, the FD-POSS/FAS coated fabrics presented no obvious change in WCAs and OCAs (hexadecane) after being damaged through (a) immersion in H2SO4 solution (pH = 1) for 24 h, (b) UV irradiation for 24 h, (c) 200 cycles of standard machine laundry, and (d) 6000 cycles of abrasion damage at the pressure of 12 kPa, respectively. The result revealed that the coating was durable enough to resist most of damage; even though some treatments (i.e., plasma irradiation and strong base corrosion) turned the wettability of the coated fabric into hydrophilic/oleophilic, the damaged fabric could self-heal and re-obtain both superhydrophobicity and superoleophobicity by heating. During the process of damaging the surface composition, polar groups that usually had a high surface free energy were normally introduced. As a result, the surface hydrophobicity/oleophobicity declined. In contrast, heat treatment could enhance the mobility of the FD-POSS molecule. With the molecules rotating and moving, the damage-introduced polar groups are more inclined to be hidden inside the coating layer; meanwhile, the low-surface-energy fluorinated alkyl chain is still exposed to the outside. Therefore, the heat treatment could self-heal the damaged surface chemistry and re-endow the FD-POSS/FAS coated fabrics with superamphiphobicity.
5). The viscous solution was further dissolved in ethanol. After ultrasonic treatment for 30 min, they obtained a homogeneous suspension that could be easily coated on a variety of fabrics by coating techniques (e.g., spraying or dip-coating), as show in Fig. 12b and c. The CAs of water, hexadecane, and tetradecane droplets on the FD-POSS/FAS coated fabrics were 171°, 155°, and 152°, respectively, showing both superhydrophobicity and superoleophobicity (Fig. 12d). When the coated fabric was irradiated by plasma to cause artificial damage, its wettability would switch from superamphiphobicity to both superhydrophilicity and superoleophilicity (Fig. 12e). Both water, hexadecane and tetradecane droplets presented CAs of 0° on the sample surface. Fortunately, the original super-liquid-repellency could recover after heating the plasma-irradiated fabrics at 135 °C for 3 min (Fig. 12f). The resultant fabrics could maintain the superoleophobicity even after 40 cycles of alternate plasma irradiation and heating, and maintain the superhydrophobicity for 100 cycles, demonstrating strong self-healing ability. Likewise, the superamphiphobicity would be lost if the coated fabrics were etched by strong alkali (e.g., KOH solution with pH = 14) and would be re-obtained by heat treatment. On the other hand, the FD-POSS/FAS coated fabrics presented no obvious change in WCAs and OCAs (hexadecane) after being damaged through (a) immersion in H2SO4 solution (pH = 1) for 24 h, (b) UV irradiation for 24 h, (c) 200 cycles of standard machine laundry, and (d) 6000 cycles of abrasion damage at the pressure of 12 kPa, respectively. The result revealed that the coating was durable enough to resist most of damage; even though some treatments (i.e., plasma irradiation and strong base corrosion) turned the wettability of the coated fabric into hydrophilic/oleophilic, the damaged fabric could self-heal and re-obtain both superhydrophobicity and superoleophobicity by heating. During the process of damaging the surface composition, polar groups that usually had a high surface free energy were normally introduced. As a result, the surface hydrophobicity/oleophobicity declined. In contrast, heat treatment could enhance the mobility of the FD-POSS molecule. With the molecules rotating and moving, the damage-introduced polar groups are more inclined to be hidden inside the coating layer; meanwhile, the low-surface-energy fluorinated alkyl chain is still exposed to the outside. Therefore, the heat treatment could self-heal the damaged surface chemistry and re-endow the FD-POSS/FAS coated fabrics with superamphiphobicity.
 | ||
| Fig. 12 Durable and self-healing superoleophobic surfaces. (a) Molecular structure of the used FD-POSS and FAS. (b) SEM image of the FD-POSS/FAS coated polyester fabric. (c) Cross-sectional TEM image of the coating layer. (d) Water, hexadecane, and tetradecane droplets on the resultant fabric. (e) Liquid droplets wetting the plasma irradiated FD-POSS/FAS coated fabric. (f) Liquid droplets on the damaged FD-POSS/FAS coated fabric after 100 cycles of alternate plasma irradiation and heating. (g and h) SEM images of the anodized alumina after double anodizations. The insets depict the shapes of water, glycerol, CH2I2, hexadecane, and rapeseed oil droplets (from I to V) on the fluorinated anodized alumina surface. (i) Variation of the contact angle of a water droplet on the plasma-damaged surface with the healing time at room temperature. (a–f) Reproduced from ref. 121 with permission from Wiley, copyright 2011. (g–i) Reproduced from ref. 132 with permission from RSC, copyright 2011. | ||
From the same research group mentioned above, Lin et al. further found a new coating system that could endow the coated fabrics with robust, self-healing superamphiphobicity through a double dip-coating process.131 The coating was made up of modified silica nanoparticles, poly(vinylidene fluoride-co-hexafluoropropylene), and 1H,1H,2H,2H-perfluorodecyltriethoxysilane (C16H19F17O3Si). The coated fabrics maintained their superhydrophobicity and superoleophobicity even after respectively suffering from 600 cycles of standard laundry, 8000 cycles of abrasion, strong acid/base corrosion, storing under ozone environment, and boiling treatment. Although the coated fabrics became hydrophilic and oleophilic after plasma irradiation, they re-obtained excellent liquid repellence just by being heated for a few minutes or being stored at room temperature. Based on the same design principle, this research group also developed several other self-healing superamphiphobic coatings.256–258
Li et al. fabricated robust superamphiphobic coatings by combining palygorskite (PAL), tetraethoxysilane (TEOS) and 1H,1H,2H,2H-perfluorodecyltriethoxysilane (PFDTES).254 First, PFDTES and TEOS were dissolved in an ethanol–water solution containing abundant nanoscale rod-like PAL. The PFDTES and TEOS were gradually deposited onto the PAL surface, catalyzed by ammonia, forming a fluorinated polysiloxane (POS) modified PAL microstructure (PAL@fluoroPOS) through a hydrolytic co-condensation process. Next, the PAL@fluoroPOS was mixed with PFDTES, and the obtained homogeneous suspensions were spray-coated onto various substrates, forming a superamphiphobic PAL@fluoroPOS/PFDTES coating. The resultant surfaces super-repelled not only cool liquids, but also hot liquids. Furthermore, the superamphiphobic coatings had high mechanical and chemical stability. The only method that resulted in the degeneration of liquid-repellence was plasma treatment, but the damaged coatings could spontaneously repair themselves under a room temperature environment for 24 h and become superamphiphobic again, showing great self-healing ability. The mechanism of self-healing is analogous to Lin's work.121
The self-healing ability of the aforementioned superoleophobic coatings are based on the fluorine migrating from the inside to the outside of the coatings. In addition to such polymer coatings, nanoscale porous structures are also conducive to making the superoleophobic materials have self-healing functions. Wang et al. prepared anodized alumina with micro/nanoscale hierarchical morphology by double anodizations.132 The first anodization endowed the alumina plate with microscale blocks (Fig. 12g), and dense nanopores were further formed on the top surface of the micro-blocks by the second anodization (Fig. 12h). Finally, the perfluorooctyl acid with low surface energy was filled into the nanopores, assisting with a vacuum pump. The fluorinated, rough, anodized alumina showed superamphiphobicity with the CAs of 165° (water), 162° (glycerol), 150° (CH2I2), 155° (rapeseed oil), and 153° (hexadecane), as shown in the insets of Fig. 12g and h. If the sample was irradiated by O2 plasma for 10 s, it would switch to superamphiphilic; i.e., both water droplet and oil droplet would spontaneously wet and spread on the treated surface. This result was caused by the damage of the perfluorooctyl acid on the top surface. However, the WCA and OCA of the plasma treated surface increased to above 160° and above 150° again, respectively, just after being placed at room temperature for two days (Fig. 12i). The self-healing mechanism is similar to Lin's work, but is slightly different.121 Although the low-surface-energy composition (i.e., fluorine) on the top surface was removed by plasma treatment, new perfluorooctyl acid would migrate to the top surface at room temperature, which was thermodynamically derived by minimizing the surface tension. The new introduced fluorinated alkyl chains were not from the inner material but from the nanopores, which looked like the nanoreservoirs of perfluorooctyl acid. The underlying perfluorooctyl acid inside the nanopores was not damaged by short time exposure to O2 plasma. As more and more low-surface-energy fluoride-chains gradually moved to the top, the superamphiphobicity of the resultant sample was finally restored. The abundant nanopores on the anodized alumina substrate were indispensable to endowing the superamphiphobic surface with self-healing ability. For the anodized alumina with only microscale roughness (no nanopores in this case), which was fabricated by just first anodization and perfluorooctyl acid infusion, its superhydrophobicity was not able to recover after plasma irradiation. The better microstructures for achieving self-healing are the microvascular networks that connect the outside surface.
4. Underwater superoleophobicity
Fabricating a superoleophobic surface is harder than the fabrication of a superhydrophobic surface, mainly because the surface tensions of oils are much lower, compared to that of water.12,13,72,102,112,117,186 To produce a superoleophobic surface in air, sufficient rough hierarchical micro/nano-structure and re-entrant surface curvature are needed, in addition to the further lowering of the surface free energy by rigorous chemical modification.3,12,72,73,102–108 Recently, inspired by fish scales, an alternative approach to achieving superoleophobicity in an aqueous environment was proposed by Jiang's group.76 It is well-known that fish can swim freely in oil-polluted water without their scales being soiled because of the underwater superoleophobicity of the scales. Such a unique characteristic of fish scales is ascribed to the synergy between the hydrophilic chemical composition and the hierarchical rough surface topography. It has been proved that a surface exhibiting superhydrophilicity in air generally shows superoleophobicity once immersed in water.76,79,94,113,114,129,136,1444.1 From superhydrophilicity to underwater superoleophobicity
Inspired by fish scales and followed the design principle of “from superhydrophilicity to underwater superoleophobicity”, many microfabrication methods have been used to build hierarchical rough microstructures on intrinsic hydrophilic materials to obtain superhydrophilicity in air and then to achieve underwater superoleophobicity; these include laser ablation,7,78,90,94,95,126,129,136 chemical etching,76,77,99,111,113,141,259 electrochemical deposition,80,135,144,145,260,261 self-assembly,91,112,262 spray/dip coating,133,134,263–265 the template method,76,127,170 lithography,76,79,111 electrochemical anodization266 and the hydrothermal method.137Jiang et al. obtained a hydrogel replica of fish scales by a double template replication process.76 The reason for using the hydrogel was that this material is hydrophilic and can retain water molecules. In water, the hydrogel film with fish-scale structures exhibited superoleophobicity, with an OCA of 162.6° ± 1.8°. They further designed three different samples on silicon substrates: smooth silicon surface, microstructured silicon surface, and micro/nanoscale hierarchical rough silicon surface.76 All three samples showed hydrophilicity in an air environment. The microstructured surface and the micro/nanoscale hierarchical rough surface were even superhydrophilic with WCAs being less than 5°. In air, oil droplets such as 1,2-dichloroethane easily spread out on all kinds of silicon surfaces, which demonstrated the superoleophilicity of these samples. However, when the samples were immersed in water, the smooth silicon surface switched to oleophobic with an OCA of 134.8° ± 1.6°, and the microstructured surface and the micro/nanoscale hierarchical rough surface exhibited superoleophobicity with OCAs of 151.5° ± 1.8° and 174.8° ± 2.3°, respectively. Unlike the underwater oleophobicity or even superoleophobicity, these artificial surfaces exhibited significantly different adhesive behaviors. The adhesive force of the smooth surface on an underwater oil droplet was greater than 24.7 ± 1.7 μN. For the microstructured silicon surface, the measured oil-adhesive force was about 10.2 ± 2.4 μN underwater. Interestingly, the adhesive force of the micro/nanoscale hierarchical rough surface was smaller than 1 μN so that the oil droplet could retain its spherical shape without any volume loss during the whole process of being in contact with and leaving the sample surface.
Wang et al. fabricated an inorganic copper oxide coating with underwater low oil-adhesive superoleophobicity.77 A copper sheet was immersed in aqueous ammonia (pH = 10.5) for about 24 h at room temperature until its color changed from bright yellow to black, resulting in a layer of rough CuO microstructure covering the substrate. As a result, there were many microscale cauliflower-like clusters with the size of 1–10 μm forming on the copper sheet. The clusters were decorated with abundant nano-branches (about 100 nm). The resultant rough surface showed superhydrophilicity in air with a WCA of 1.6° ± 0.8°. After immersing in water, the sample became superoleophobic. An oil droplet on the surface (in water) looked like a sphere with OCA of 163.0° ± 4.5°, and easily rolled off when the sample was slightly tilted. The underwater superoleophobic surface also had a very low adhesive force (<1 μN) on various oil droplets.
Our group reported an efficient, one-step method to realize underwater superoleophobicity on a silicon surface by femtosecond laser ablation, as shown in Fig. 13.94 A flat Si wafer showed intrinsic hydrophilicity in air with the WCA of 60° ± 2° (Fig. 13c) and therefore presented oleophobicity under water. The underwater oil droplet (1,2-dichloroethane) on the Si wafer had an almost half-sphere shape with the OCA of 124.6° ± 1° (Fig. 13e). After irradiation by femtosecond laser pulses, an aligned micro-mountain array with the interval of 10 μm was formed on the Si substrate (Fig. 13a).223,267 The rough micro-mountains had a diameter of approximately 6 μm and a height of 2.9 μm. There were abundant protrusions with the size of tens to a few hundreds of nanometers decorating every micro-mountain (Fig. 13b). The surface roughness was about 2.46 μm. The micro/nanoscale hierarchical structure was able to largely enhance the wettability of the silicon surface. In an air environment, the as-prepared rough surface was transformed from ordinary hydrophilic into superhydrophilic. When a 7 μL water droplet was lowered to touch the sample, the droplet spread out within one second, resulting in a very small WCA of only 4° (Fig. 13d). In contrast, when the resultant rough silicon surface was submerged in water, an underwater oil droplet on the surface could retain a spherical shape with OCA reaching up to 159.4° ± 1°, showing underwater superoleophobicity (Fig. 13f). The surface also exhibited ultralow oil-adhesion underwater, with the result that the oil droplet could easily roll down when the surface was slightly inclined at 0.5° (Fig. 13g). The superoleophobicity in water and ultralow oil-adhesion of the as-prepared surface mentioned above can be explained by the underwater version of the Cassie contact state (Fig. 3h). Once the fabricated sample was immersed in water, the valley of the hierarchical micro/nanoscale rough structures was filled by water due to the superhydrophilicity. When an underwater oil microdroplet was placed on this rough surface, there existed a layer of water trapped in the hierarchical microstructures. The trapped water cushion was just between the oil microdroplet and the surface, preventing oil from penetrating into the rough microstructures because water naturally repels oil. In fact, the underwater oil droplet only touched the peaks of the micro-mountain array structures, resulting in a greatly reduced contact area between the underwater oil droplet and the rough substrate. The trapped water endowed the femtosecond laser ablated silicon surface with underwater superoleophobicity, and the small contact area further caused ultralow oil-adhesion. It was tested that the hydrophilicity of the flat silicon surface in air turned into oleophobicity in water, and the oleophobicity was greatly increased to underwater superoleophobicity by femtosecond laser ablated hierarchical rough microstructures.
 | ||
| Fig. 13 Femtosecond laser ablated underwater superoleophobic Si surface. (a and b) SEM images of the rough Si surface irradiated by femtosecond laser. (c and d) Water wettability on the (c) flat and (d) laser-induced rough Si surfaces in air environment. (e and f) Oil wettability on the (e) flat and (f) laser-induced rough Si surfaces in a water environment. (g) Underwater oil droplet rolling on a femtosecond laser ablated surface with tilt angle of 0.5°. Reproduced from ref. 94 with permission from RSC, copyright 2014. | ||
Inspired by fish scales, Lin et al. fabricated a robust underwater superoleophobic surface of hierarchical macromolecule-nanoclay hydrogels.111 The interaction of flexible macromolecules and rigid nanoclays made the trapped water layer on the rough surface microstructures very stable, thereby endowing the as-prepared material with robust underwater superoleophobicity. Wang et al. used the in situ alternating-current deposition method to prepare flower-like conversion films composed of amorphous iron phosphate on carbon steel.260 The hierarchical micro/nanostructured conversion films were superhydrophilic in air and superoleophobic in water. Xu et al. obtained an ion-induced organic/inorganic hybrid film using the typical LBL assembly method.268 The hybrid film was superoleophobic and showed ultralow oil-adhesion even in artificial seawater. Zhang et al. coated sodium silicate and TiO2 nanoparticles on the stainless steel mesh through a LBL assembly method.269 The obtained mesh showed underwater superoleophobicity and had the function of separating water/oil mixtures. Once the separating mesh was polluted, the TiO2 composition would break down and remove the contaminants by UV irradiation and make the contaminated mesh regain the separation ability. Ma et al. reported a novel strategy for realizing substrate-independent underwater superoleophobicity.270 A catechol bearing polymer was synthesized and then coated on various substrates. Next, a nanostructure was formed on the polymer coated substrate by the LBL assembly method. The resultant surfaces exhibited stable superoleophobicity in water. In addition, sticky or rolling underwater superoleophobic surfaces could be prepared by using spin-assisted or dip-assisted LBL processes, respectively. Lai et al. prepared a large scale, pinecone-like TiO2 particle film, based on the one-step electrochemical anodizing method.271 With the film shifting from air atmosphere to underwater, its wettability changed from superhydrophilicity and superoleophilicity to excellent superoleophobicity. Liu et al. fabricated underwater superoleophobic copper wires whose surfaces were covered by hierarchical copper oxide microstructures through an alkali-corrosion process.99 Zhu et al. used a surface-initiated atom transfer radical polymerization process to prepare a superhydrophilic and underwater superoleophobic zwitterionic polyelectrolyte brush–poly(3-(N-2-methacryloxyethyl-N,N-dimethyl)ammonatopropanesultone) grafted poly(vinylidene fluoride) membrane for separating oil from water.272
4.2 Controllable oil-adhesion
Superoleophobic surfaces with very low oil-adhesion have strong anti-oil ability.75,77,94,260,270 In contrast, when the oil-adhesion of a superoleophobic surface is not ultralow, but ultrahigh, that oil droplet can stick to the substrate at any tilted angle; such a surface can be used in no-loss micro-oil-droplet transportation.78,91,144 Currently, multifunctional superoleophobic surfaces with differing oil-adhesion (from low to high) have attracted tremendous attention because these surfaces allow the manipulation of oil droplets on the target materials.78,80,91,112Huang et al. prepared three latex particles (with spherical, cauliflower-like, and single-cavity structures) by emulsion polymerization, as shown in Fig. 14a–f.112 The shape of the latex particles was controlled by the feeding modes of divinylbenzene into the polymerization system. They further obtained three typical films assembled with these three kinds of latex particles. All three resultant films showed superoleophobicity in water. The underwater OCAs of a silicon oil droplet were 146.5° ± 1.6°, 159.5° ± 1.3° and 160.5° ± 2.1° on the substrates assembled from spherical, cauliflower-like, and single-cavity latex particles, respectively. In addition, these films exhibited adjustable underwater oil-adhesion (Fig. 14g–i). The film with spherical latex particles had an ultrahigh adhesive force to an oil droplet (>69.6 μN) (Fig. 14g). After approaching and just contacting the sample surface, an underwater oil droplet had a large shape distortion and finally broke into two parts when it was carried off the film. The film assembled from cauliflower-like latex particles showed a moderate oil-adhesion in water (Fig. 14h). The adhesive force was 43.1 μN. For the surface covered with single-cavity latex particles, the adhesive force to an underwater oil droplet was as low as 9.8 μN (Fig. 14i). The oil droplet hardly adhered to the resultant surface and had almost no shape deformation during the whole process of approaching, contacting, and leaving. The controllable oil-adhesion was ascribed to the continuous change in the wetting state/contact model from underwater Wenzel, to Wenzel–Cassie transitional, to the Cassie regime.
 | ||
| Fig. 14 (a–c) SEM and (d–f) AFM images of the films assembled with three different kinds of latex particles: (a and d) spherical, (b and e) cauliflower-like, and (c and f) single-cavity latex structures. The insets in (a–c) show the shapes of a water droplet on the corresponding films in air. (g–i) An underwater oil droplet sticking or rolling on the three films assembled from (g) spherical, (h) cauliflower-like, and (i) single-cavity latexes. Reproduced from ref. 112 with permission from Wiley, copyright 2011. | ||
The spherical latex particles looked relatively smooth. The underwater oil droplet could wet the rough microstructures of the sample surface assembled from spherical latex particles. The large contact area between the oil droplet and the substrate formed a continuous solid/water/oil TPCL, resulting in a very high CA hysteresis and oil-adhesion.156,218 The oil droplet stuck on the film even through the sample was vertical or turned upside-down (Fig. 14g). The oil droplet on the film was in the underwater Wenzel state. For the film with cauliflower-like latex particles, the underwater oil droplet partly penetrated the space of the irregular nanostructures. The decreased contact area between the oil droplet and the sample surface gave rise to a decrease in oil-adhesion (Fig. 14h). This contact model belonged to the underwater version of the transition state. In comparison, the underwater oil droplet could easily roll off the film covered with single-cavity latex particles, with almost no CA hysteresis (Fig. 14i). This result agreed well with the characteristics of the underwater Cassie state. A water layer was trapped in the cavity, which prevented the oil droplet from fully touching the latex particles. Consequently, the underwater oil microdroplet only contacted the protruding point of the latex cavity, decreasing the oil contact area. The discontinuous TPCL led to an ultralow oil-adhesive force.156,218
Zhang et al. fabricated different microstructured Ni/NiO surfaces showing underwater superoleophobicity by combining electro-deposition and heating techniques.80 The surface microstructures as well as the oil-adhesion could be simply controlled by the annealing temperature. There were many micropores with diameter of 4–11 μm, with close and disorderly distribution on the Ni sample surface, which were electrodeposited for 30 s and subsequently annealed at 500 °C for 5 h. With the annealing temperature increasing to 900 °C and further to 1100 °C, the porous structure gradually degenerated, and a lot of irregular blocks and smooth microscale crystals appeared instead. The adhesive force of the resultant surface on an underwater oil droplet was tuned from ultralow to ultrahigh (1–60 μN), with the contact model regulating from the underwater Cassie state to the underwater transition state and then to the underwater Wenzel state.
Similarly, our group obtained underwater superoleophobic glass surfaces with different micro/nanoscale hierarchical rough structures, induced by femtosecond laser.78 When the average distance (AD, which was determined by the laser scanning speed and the shift of scanning lines) of the laser-pulse-ablation points was relatively small (e.g., AD = 2 μm), a self-organized micro-island array, decorated with abundant nanoscale porous structures and protrusions, was formed on the substrate surface (Fig. 15a–c). There were no typical femtosecond-laser-pulse-induced microcrater-like structures in this case, due to the strong overlap of the adjacent laser pulse irradiated areas. With AD increasing to 4 μm, the laser pulse ablated craters were partly overlapping and connected with each other, resulting in a wave-like pattern (Fig. 15d and e). Apart from the micro-waves, a layer of nanoparticles with diameter of just a few tens of nanometers uniformly covered the whole surface (Fig. 15f). For the as-prepared sample with AD of 6 μm, the surface was covered by nearly complete microcraters decorated with nanoparticles (Fig. 15g–i). All the underwater oil droplets on these surfaces presented almost spherical shapes, revealing underwater superoleophobicity (inset of Fig. 15a, d and g). The corresponding OCAs were as large as 160.5° ± 2° (AD = 2 μm), 158° ± 2.5° (AD = 4 μm), and 154° ± 1.5° (AD = 6 μm), respectively. In contrast, the oil-adhesive properties of the laser ablated surfaces with different parameters varied greatly, compared to the similar underwater superoleophobicity, which could be tuned from very low to ultrahigh by increasing the parameter AD. When the AD increased from 2 μm to 4 μm and then to 6 μm, the underwater OSA increased from 3.5° (inset in Fig. 15b) to 25.5° (inset in Fig. 15e) and then to 90° (insets in Fig. 15h and i). The OSA of 90° expressed that the oil droplet was firmly pinned on the substrate, even when the substrate was vertical or turned upside down. The controllability of oil-adhesion was ascribed to the surface topography variation, which resulted in the wetting behavior changing from the underwater Cassie state to the transition state, and even to the underwater Wenzel state. The femtosecond laser induced underwater superoleophobic surfaces with ultralow oil-adhesion have strong capability for oil resistance in water, while the high oil-adhesive surfaces can be used to transfer small oil droplets, like a “mechanical hand” (see Section 6.4 for details).
 | ||
| Fig. 15 Topography and underwater oil-wettability of the femtosecond laser ablated glass slide surfaces fabricated at different average distances of laser-pulse-ablation points: (a–c) AD = 2 μm, (d–f) AD = 4 μm, and (g–i) AD = 6 μm. Insets in (a, d and g) are the shapes of 8 μL underwater oil droplets on the corresponding surfaces. Insets in (b, e, h and i) reveal an underwater oil droplet rolling or being pinned on the corresponding surfaces. Reproduced from ref. 78 with permission from Springer, copyright 2015. | ||
Besides varying surface microstructures, the oil-adhesion can also be adjusted by regulating the surface chemical composition of underwater superoleophobic materials. Cheng et al. obtained tunable oil-adhesive superoleophobic surfaces underwater, based on a two-step sputtering and self-assembled monolayer process.91 A layer of nanoscale copper particles was firstly covered on the glass substrate via a simple sputtering process. The samples were further modified with an n-alkanoic monolayer by immersion into the ethanol solution containing different n-alkanoic acids (0.001 M) for about 12 h. By simply increasing the chain length of the n-alkanoic acids, the adhesive force of the resultant surfaces on an underwater oil droplet increased from less than 1 μN to larger than 58 μN, revealing that the underwater oil-adhesion was controlled from low to very high.
Chen et al. prepared a thermal-responsive poly(N-isopropylacrylamide) (PNIPAAm) hydrogel whose underwater oil wettability could be reversibly switched between ultralow adhesive superoleophobicity and high adhesive oleophobicity by changing the ambient temperature.142 Ding et al. synthesized an underwater superoleophobic aligned polyaniline (PANI) nanowire film through electrochemical polymerization.144 With the applied potential changing from 0.43 V to −0.2 V or 0.8 V, the 1,2-dibromoethane droplet reversibly switched from the rolling state to the pinned state in water. Cheng et al. grafted the pH-responsive poly(acrylic acid) (PAA) onto a glass substrate by means of plasma polymerization.140 The adhesive force between the resultant film and the underwater oil droplet changed from 21.6 ± 5.0 μN to zero as the pH of the surrounding water medium increased from 1 to 12. Heng et al. fabricated different porous films, which were made up of montmorillonite clay/PAA.273 The underwater oil-adhesion could be controlled by the montmorillonite arrangement in the pore wall.
4.3 Underwater anisotropic oil-wettability
The anisotropic wettability can be realized not only in air, but also in water medium.92,126,127 Recently, our group reported a kind of microgroove array structure on Si substrate through a simple line-by-line femtosecond laser serial scanning process, as shown in Fig. 16a.126 The width and depth of the rough periodic microgrooves were about 8 μm and 5 μm, respectively. The period of the microgroove array could be adjusted by the interval of the scanning lines. Every microgroove was formed by many consecutive micro-craters, which were generated by laser pulse ablation (top inset of Fig. 16a).222,267,274,275 There were abundant irregular particles, with size ranging from tens to several hundreds of nanometers, densely decorating the inner wall and outer rim of the microgrooves. Underwater anisotropic oil-wetting was exhibited after immersing the resultant sample in water. For the underwater microgroove array with the period of 450 μm, the OCA perpendicular to the grooves reached up to 155.5° ± 1.6° (Fig. 16c), while the OCA parallel to the grooves was only 135.7° ± 1.5° (Fig. 16b). The OCA difference revealed that the underwater oil microdroplet preferred to spread along the microgrooves. In addition, the anisotropic oil-wetting could be easily tuned by just changing the interval of the microgroove arrays, with the degree of the wetting anisotropy being changed from 0° to ∼20°. The femtosecond laser ablated rough Si surface exhibited underwater superoleophobicity and ultralow oil-adhesion because water filled in the microgrooves, thus preventing the oil droplet from effectively touching the rough microstructures.94 The contact model of the underwater oil droplet and the laser-induced microgrooves domain could be considered as an underwater Cassie state.76 On the contrary, the oil droplet would be fully in contact with the non-irradiated flat part, which was between two adjacent microgrooves, belonging to the underwater Young state. An energy barrier usually exists between adjacent domains with dissimilar wettabilities, according to Gibbs' criterion.223,233 For the rough microgroove-array surfaces, the energy barrier between the flat domain and the trapped water in the laser-ablated microgrooves prevented the oil droplet from spreading in the perpendicular direction (bottom inset of Fig. 16a), while there was no energy barrier in the parallel direction. The oil was elongated along the microgrooves, so the OCAs along the parallel direction were smaller than those along the perpendicular direction, exhibiting a controllable anisotropy.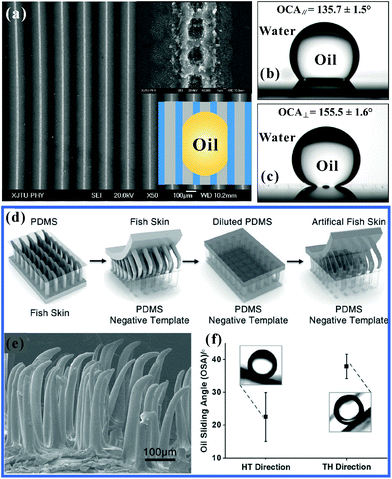 | ||
| Fig. 16 Underwater anisotropic oil-wettability on a femtosecond laser induced Si microgroove array and a PDMS replica of filefish skin. (a) SEM image of the microgroove array fabricated through the line-by-line femtosecond laser scanning method. The top inset shows the SEM image of a single microgroove at high magnification. The bottom inset reveals the restricting effect of the microgroove array on an underwater oil droplet. The energy barrier at the boundary between the untreated flat domain and laser ablated microgrooves prevents the oil droplet from spreading in the perpendicular direction. (b and c) Shapes of an underwater oil droplet on the microgroove structured surface in the (b) parallel and (c) perpendicular directions, respectively. (d) Schematic diagram of obtaining a PDMS replica from filefish skin. (e) Microstructures of the PDMS replica surface. (f) Sliding behavior of an underwater oil droplet on the replica surface. (a–c) Reproduced from ref. 126 with permission from AIP, copyright 2014. (d–f) Reproduced from ref. 127 with permission from Wiley, copyright 2014. | ||
Compared to the two-dimensional anisotropic oil-wettability of the microgroove array structure, directional oil-adhesion (at the one-dimensional level) was also achieved on the asymmetric microstructures. Cai et al. prepared an artificial filefish surface by replicating the N. septentrionalis skin.127 The cleaned filefish skin was first gradient dehydrated by ethanol solution to promote its hardness. Using the pre-treated filefish skin as the template, its PDMS replica was obtained by two rounds of replication (Fig. 16d). The obtained PDMS replica had a very similar surface microstructure to filefish skin (Fig. 16e). Hydrophilic surface composition is necessary to achieve underwater oleophobicity, but PDMS is a typical hydrophobic material with low surface energy.53,54,71,179,221,226 To address this limitation, the artificial PDMS “fish skin” was irradiated by oxygen plasma. With the silanol group (–SiOH) forming on the PDMS surface, the artificial skin switched from hydrophobic to hydrophilic.79,127 When the PDMS fish skin was immersed in water, it showed quasi-superoleophobicity with an OCA of 143° ± 2.1°. Like the natural filefish, the artificial PDMS fish skin also exhibited an anisotropic underwater oleophobicity. Along the direction of the oriented hook-like spines, the OSA was 22.5° ± 7.3°, but the underwater oil droplet stayed on the sample surface until the artificial skin was tilted at 37.9° ± 3.7° in the opposite direction, as shown in Fig. 16f. It was demonstrated that the oil droplet in water was more inclined to move in a certain direction. This kind of anisotropic wettability is usually called unidirectional adhesion/rolling, which was initially observed from butterfly wings.156,226 They further achieved the unidirectional oil-adhesion on commercial cloth corduroy through unidirectional ironing and subsequent hydrophilic polymer modification. The OCA difference along two opposite directions could reach up to 20°. If an oil droplet was sandwiched between two resultant samples exhibiting unidirectional oil-adhesion in water medium, the continuous directional oil transportation would be achieved by repeating the cycles of pressing together and release.
In general, two criteria play a very important role in constructuring underwater anisotropic oleophobic surfaces: high surface energy and anisotropic microtextures. The former ensures that the rough microstructure is fully wetted by water and repels oil after immersion in water. The latter results in the anisotropic static or dynamic oil-wettability on the microtextured substrates. The underwater anisotropic oil-wetting surfaces have many potential applications, such as movement guiding of underwater organic liquids, oil collection, oil transportation, and self-cleaning coatings for marine equipment.92,126,127,160,214
4.4 Transparency
Transparency is very important for the application of underwater superoleophobic materials in underwater optics, such as diving goggles, oil-proof windows of underwater cameras, and even imageable substrates for biologically active cells or tissue. Our group obtained a rough silica glass sheet with both underwater superoleophobicity and good transparent property through a femtosecond laser ablation process.129 Fabrication of the underwater superoleophobic microstructure was inspired by fish scales,76 while the achievement of transparency was inspired by Diphylleia grayi. As shown in Fig. 17a and b, the Diphylleia grayi's petals change from white to transparent if the weather shifts from a sunny day to a rainy day; the cell structure of the petal is very loose. On a sunny day, the intercellular space is filled with air. The diffuse reflection at the interface between the colorless cytolymph and the trapped air results in a white color. Water can displace the trapped air and enter into the intercellular spaces in the rain. The transparent property is improved by the newly generated water–cytolymph (liquid–liquid) interface, so the petals become transparent. We used the femtosecond laser ablation method to form a layer of irregular nanoparticles on silica glass surface (Fig. 17c). After immersing the sample in water, superoleophobicity and very low adhesion to an oil droplet were exhibited. This oil droplet maintained a spherical shape and could easily roll on the surface (Fig. 17d). The measured OCA and OSA were 160.2° ± 1° and 1°.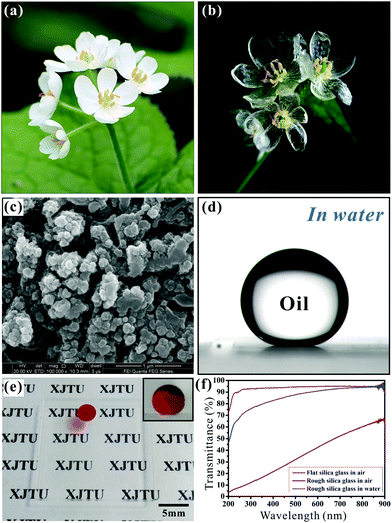 | ||
| Fig. 17 Transparent underwater superoleophobic silica glass surface. (a and b) Petals of Diphylleia grayi (a) on a sunny day and (b) in the rain. (c) SEM image of the femtosecond laser ablated microstructure on a silica glass surface. (d) Oil droplet on the rough silica glass surface in water. (e) Underwater high transparency of the femtosecond laser ablated glass surface. (f) UV-vis spectra of different silica glass: purple line, bare flat silica glass in air; red line, rough silica glass in air; blue line, rough silica glass in water. Reproduced from ref. 129 with permission from RSC, copyright 2015. | ||
The original laser ablated surface was white and looked opaque in air. Nothing could be seen behind the glass sheet. Interestingly, it became transparent just through being shifted into water medium. The letters “XJTU” in a paper behind the resultant sample could be clearly observed, demonstrating that the rough silica glass in water had high transparency (Fig. 17e). The UV-vis spectra further indicated that the transmittance of the underwater rough glass was very close to that of the bare flat silica glass in the visible region (Fig. 17f). When a He–Ne laser beam with the wavelength of 633 nm and the power of 0.96 mW passed through the underwater sample, the transmittance reached to 91.6%. The excellent transparency resulted from the existence of the surrounding water environment. The reflectance, R12, at the interface of two different media (1 and 2) can be expressed as follows:
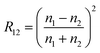 | (12) |
4.5 Durability
Chemical and physical durability are essential to the applications of the underwater superoleophobic surfaces in harsh environments, which will provide the surfaces with important opportunities in different kinds of industrial production and our daily life, such as industrial waste oil cleanup and cell engineering.129,133–135Li et al. fabricated a rough mesh with robust superoleophobicity in water through spraying the mixture of palygorskite powders and polyurethane onto a copper mesh substrate.133 Palygorskite consists of hydrated octahedral layered magnesium aluminum silicate with reactive –OH groups on the surface. It is a typical hydrophilic crystal and has great water-absorbing and water-retaining ability. Waterborne polyurethane was used to enhance the binding force between the substrates and the palygorskite powders coating. The palygorskite powder (0.3 g) was firstly pre-activated with 4 M HCl solution, and then was dispersed in 20 mL of acetone solution of waterborne polyurethane (0.05 g), forming a homogeneous suspension after magnetic stirring. The suspension was coated on a copper mesh with pore diameter of ∼75 μm (200 mesh size) by a spray method and subsequently dried in air. As a result, abundant palygorskite was densely and randomly coated on the copper mesh. The large magnification SEM image revealed that the palygorskite coating was the aggregate of a lot of single fibrils with length of several hundreds of nanometers and diameter of 100 nm. Due to the existence of many hydroxyl groups and hierarchical rough microstructures on the coating surface, the as-prepared rough mesh showed superhydrophilicity and superoleophilicity in air. After immersing in water, the coated mesh became superoleophobic. The OCAs of various oil droplets (e.g., chloroform, kerosene, petroleum ether, rapeseed oil, toluene, and hexane) were greater than 150°. The palygorskite coated mesh surface also exhibited ultralow oil-adhesion in water. For example, an underwater oil droplet of chloroform could easily roll off the inclined sample surface when the tilted angle of the surface was just 8°. Furthermore, it was also found that the coated mesh also super-repelled underwater oils when the mesh was immersed in different corrosive solutions (1 M HCl, 1 M NaOH and 1 M NaCl) and hot water, showing stable underwater superoleophobicity. The advantage of chemical resistance to harsh environments endowed the underwater superoleophobic rough meshes with the ability to efficiently separate the mixtures of oil and various corrosive aqueous solutions.
Waste potato residue is made up of superhydrophilic and superoleophilic starch, cellulose and pectin. In addition, it is a very low-cost material and can easily be obtained. Li et al. also prepared a potato residue powder-coated metal mesh by the spraying method, which is the same as mentioned above.134 The resultant meshes exhibited durable underwater superoleophobicity, even when the sample was immersed in a series of corrosive solutions (1 mol L−1 HCl, 1 mol L−1 NaOH, and 10 wt% NaCl). Our group found that the femtosecond laser induced rough silica glass could maintain superoleophobicity and low oil-adhesion not only in water, but also in strong acid or alkaline aqueous solutions.129 The OCAs remained larger than 150° and the OSAs were smaller than 10° in all cases, with the pH of environmental liquids ranging from 1 to 13. Zhang et al. fabricated a beautiful porous structured copper mesh film by electrodeposition technology.135 The copper mesh substrate was used as the cathode, and a Pt plate (2 × 2 cm2) was the anode. The electrodeposition process was performed in an electrolyte solution of 0.1 M CuSO4 and 1 M H2SO4 at the constant current density of 6 A cm−2 and lasted 5 to 15 s. After electrodeposition, there were abundant porous structures coving the copper wires. The pores were ∼23 μm in size and their sidewalls were decorated with massive nanoparticles. The as-prepared films were superhydrophilic in air and showed superoleophobicity and very low oil-adhesion in water. When the film was immersed in acidic, basic or salt solutions for 24 h, it was found that the underwater low-adhesive superoleophobicity was still there. The underwater superoleophobic films with excellent environmental stability are promising for use in more complex applications.
5. Smart oil-wettability
In recent years, triggered by the peculiar properties and promising functions, various “smart” surfaces that can respond to external stimulation (e.g., light,87,102,136,137,276–282 pH,88,139–141,283,284 temperature,97,143,217,285–288 electrical potential,144,145,289–291 magnetic field,292–294 surface curvature,295 density of surroundings,90 applied force296) to reversibly switch their static or dynamic wettability have been increasingly investigated.16,297 The smart superhydrophobic or superoleophobic surfaces are the most complicated and subtle interfaces in the research field of wettability, which are usually hailed as “the bright pearl on the imperial crown”. The surfaces showing smart wettability are usually obtained by forming rough microstructures on stimuli-responsive material surfaces or modifying rough solid surfaces with stimuli-responsive molecular layers.16,285,298–300 Although many smart superhydrophobic surfaces have been developed,217,276,284,285,292,295 the realization of smart oil-wettability on superoleophobic surface is still in its early stages and further research is needed.5.1 Light response
Our group fabricated a novel wettability-switchable hierarchical rough titanium dioxide (TiO2) layer on a titanium substrate using the one-step femtosecond laser ablation method.136 The synchronicity of microstructure formation and oxidization during femtosecond laser ablation constructed a stratum of rough TiO2 layer on the original substrate (Fig. 18a, b and g). The rough TiO2 surface looked like a micro-mountain array decorated with randomly distributed nanoscale protrusions. Initially, the laser-induced hierarchical rough TiO2 surface showed superhydrophobicity with the WCA of 154.5° ± 2.5° (Fig. 18c). Once immersed in water, the as-prepared TiO2 region can reflect homogeneous light just like a silver mirror. This mirror forms by virtue of the trapped air between water and the substrate, which demonstrated that the contact state between the sample and water belonged to the Cassie model.48,163,164,301,302 If an oil droplet is in contact with the rough surface submerged in water, the underwater oil will rapidly spread out on the sample surface. The OCA of only 4° was measured, indicating the underwater superoleophilicity of the laser-induced TiO2 surface without any treatment (Fig. 18e). Surprisingly, when the sample was irradiated with UV light for 40 min, the WCA in air sharply decreased to 2.5°, which revealed that the wettability of the resultant surface in air was transformed from superhydrophobicity to superhydrophilicity (Fig. 18d). The silver mirror-like reflectance disappeared when the sample was submerged in water because water wetted and filled the interspace of the rough micro/nanoscale structures. The rough surface showed underwater superoleophobicity with the OCA reaching up to 160.5° ± 2°, due to the intrinsic oil-repellency of the filled water (Fig. 18f). The underwater oil droplet easily rolled off as soon as the substrate was slightly inclined. It was found that the underwater oil-wettability showed an opposite switching trend to water-wettability in air. Interestingly, if the sample was further stored in the dark for two days, the original wettabilities such as superhydrophobicity and underwater superoleophilicity would recover (Fig. 18c and e). The above-mentioned wettability switching process could be repeated for many cycles.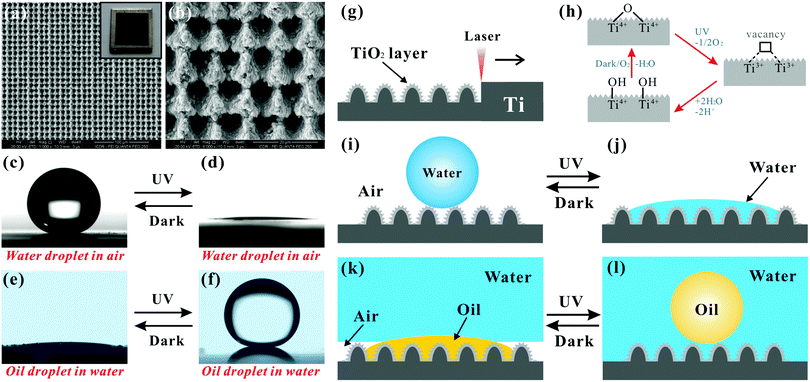 | ||
| Fig. 18 Light-induced switching of underwater superoleophobic–superoleophilic states. (a and b) SEM images of the rough Ti surface after femtosecond laser ablation. (c and d) Water droplet on the rough sample surface after (c) storing in the dark and (d) irradiation with UV light, respectively, in air environment. (e and f) Underwater oil droplet on the rough sample surface after (e) storing in the dark and (f) irradiation with UV light, respectively. (g–l) Schematic illustration of the switchable wettability. Reproduced from ref. 136 with permission from RSC, copyright 2015. | ||
The internal mechanism of the switchable wettability is based on the fact that TiO2 is an excellent photo-responsive material.276,277 The laser induced rough TiO2 layer initially exhibits superhydrophobicity and ultralow water adhesion in air, agreeing well with the Cassie state (Fig. 18i). According to the Cassie wetting state, a trapped air cushion was generated between water and rough microstructures after immersing the sample in water. At this point, if an underwater oil droplet was put on the sample surface, the oil would spread along with the air cushion and displace the trapped air due to the capillary action and water pressure. Therefore, the surface showed underwater superoleophilicity (Fig. 18k). As shown in Fig. 18h, UV light irradiation created pairs of electrons and holes on the rough TiO2 surface.276,277,281,303,304 The photogenerated holes immediately united with lattice oxygen to form highly unstable oxygen vacancies. These oxygen vacancies were inclined to absorb the atmospheric water by dissociation, generating two adjacent Ti–OH groups per vacancy. On account of the high surface energy of the –OH groups, the UV-irradiated rough TiO2 surface was superhydrophilic in air (Fig. 18j). Water could fully wet the rough microstructures of the TiO2 surface when the metal sheet was dipped into water. An underwater oil droplet on such rough TiO2 microstructures is in the underwater Cassie state, so the surface shows underwater superoleophobicity (Fig. 18l). When the substrate was further stored in the dark, the implanted hydroxyl moieties were more easily replaced by ambient oxygen (Fig. 18h), causing the sample to recover its original superhydrophobicity and underwater superoleophilicity (Fig. 18i and k).276,277,281,303,304
The reversible transition of underwater oleophilicity-superoleophobicity on the rough TiO2 surface can also be realized by alternating contamination treatment and UV irradiation. Wang et al. used a simple one-step hydrothermal method to form hierarchical rutile TiO2 flowers on a fluorine-doped tin oxide substrate.137 There were many uniform TiO2 flowers with diameter of ∼6 μm on the resultant surface. Every microscale flower was composed of many tetragonal pillar-liked petals with a rough top and smooth side wall. The submicron petal was an aggregation of many ultrathin nanoneedles. The just prepared surface showed superhydrophilicity and superoleophilicity in air. The CAs were 0° to both water and oil (trichloromethane) droplets. Similar to fish scales, the resultant surface became superoleophobic after immersion in water with an underwater OCA of 155°. This good underwater anti-oil capability was caused by the formation of a three phase (solid/water/oil) system. In this wetting state, a water layer underneath the oil droplet was trapped in the rough TiO2 microstructures. However, when the sample was contaminated with oleic acid (in n-pentane, 0.5 wt%) for 5 s, the as-prepared TiO2 coating would lose its underwater superoleophobicity, with the OCA directly decreasing to 64°. In this case, the oil droplet on the contaminated TiO2 surface was at a heterogeneous wetting state, since some oleic acid spots were also trapped in the hierarchical rough microstructures. The trapped oleic acid spots resulted in a decrease in the contact area between the trapped water and oil droplet as well as the apparent underwater OCA. Interestingly, the oil-wettability could recover to the original underwater superoleophobicity upon UV light (wavelength = 365 ± 10 nm) irradiation for 2 h. TiO2 is a photosensitive material.276,277 The electron–hole pairs were generated and migrated to the TiO2 surface during UV light irradiation. These holes preferred to react with OH− or to adsorb surrounding water molecule to generate highly reactive hydroxyl radicals. The hydroxyl radicals were able to further oxidize and decompose most organic compounds, such as oleic acid, re-endowing the TiO2 surface with underwater superoleophobicity. Furthermore, the rough TiO2 surface still maintained the responsivity after four cycles of oleic acid contamination and UV irradiation recovery.
Zhang et al. prepared TiO2/single-walled carbon nanotube (SWNT) composite coatings through a modified liquid phase deposition process.138 Then, the TiO2/SWNT coatings were modified with 1H,1H,2H,2H-perfluorodecyltrichlorosilane (FDTS) to chemically lower the surface energy. The treated rough coating displayed superoleophobicity with an OCA of 160.4° and an OSA of 5.9° to a silicon oil (γ = 21.5 mN m−1) droplet in air. When the surface was irradiated with UV light (wavelength = 365 nm, intensity = 21 mW cm−2) for 13 h, the composite coatings were thoroughly converted from superoleophobic to superoleophilic. This conversion was ascribed to the FDTS monolayer being decomposed by the photocatalytic action of the TiO2 component.305–312 As the UV illumination time went on, the atomic percent of C–F chemical bond decreased almost linearly and tended to zero after 12 hours. This result revealed that the FDTS monolayer could be completely decomposed. The UV-induced conversion from superoleophobicity to superoleophilicity was also valid for various liquids, such as hexadecane, glycerol, 40% ethanol in water (γ = 30.2 mN m−1), 11.1% ethanol (γ = 46.0 mN m−1), and water. The OCA would sharply reduce to almost zero once the UV illumination time exceeded a threshold. Interestingly, the critical values were different for different liquids. The liquid with higher surface tension needed more irradiation time to become superoleophilic on the resultant surface. As a result, there was a state where the sample surface showed superoleophobicity to one oil droplet but superoleophilicity to another with lower surface tension, by precisely controlling the exposure time of UV light. The distinguishable difference in the surface tensions of these two liquids was even lower than 5 mN m−1. Based on the inverse oil-wettability, the separation of two organic liquids with different surface tensions using a TiO2/SWNT coated metal mesh was demonstrated.
5.2 pH response
Cheng et al. prepared a pH-responsive surface showing switchable oil wettability.139 The surface was superoleophilic in acidic aqueous solutions, but superoleophobic in basic aqueous solutions. The hierarchical microstructures were firstly formed on the copper foil substrates by immersing the samples into the solution of NaOH (2.5 M) and (NH4)2S2O8 (0.1 M), and subsequent heat treatment. As shown in Fig. 19a and b, the treated surface was composed of abundant microflowers with the size of ∼4 μm and nanowires of about 20–80 nm in diameter; all the microflowers stood on the nanowires. The sample was then dipped into a mixed thiol solution of HS(CH2)9CH3 and HS(CH2)10COOH to graft both carboxylic acid and alkyl groups. In water, the OCAs of an underwater oil droplet (octane, 4 μL) on the resultant surface remained constant at about 0° in neutral and even acidic water (pH ≤ 7), showing underwater superoleophilicity (Fig. 19c). However, the OCAs increased with increasing pH in basic water. The OCA reached up to 162° at the water pH of about 12, indicating that the surface switched to underwater superoleophobic (Fig. 19d). After the sample was taken out of the basic water and instantly washed with pure water, it would recover to superoleophilic after immersion into acidic liquid again. Such a reversible transition between underwater superoleophilic and superoleophobic states could be repeated many times. Both carboxylic acid and alkyl groups were grafted on the sample surface during the preparation process. Generally, the carboxylic acid groups are protonated in nonbasic aqueous solutions and deprotonated in basic aqueous solutions. In the neutral or acidic water (pH ≤ 7), the alkyl groups on the sample surface were dominant, resulting in the low surface free energy of the surface (Fig. 19e). The surface showed intrinsic oleophilicity in water. The oil-wettability was further magnified to underwater superoleophilicity by the rough microstructures. In contrast, the surface carboxylic acid groups were deprotonated in basic water (Fig. 19f). The deprotonated state was hydrophilic, since hydrogen bonding generally occurred between the deprotonated carboxylic acid groups and water molecules. As a result, water wetted the sample surface and was trapped among the rough microstructures. Higher pH led to more deprotonated carboxylic acid groups as well as more trapped water. Particularly at pH ≥ 12, the trapped water layer was thick enough, which led to the underwater superoleophobicity due to the formation of a stable solid/water/oil three-phase system (underwater Cassie state). Based on the above, it can be found that the switching ability of underwater oil-wettability was caused by the cooperation between the surface chemistry variation and hierarchical structures on the surface.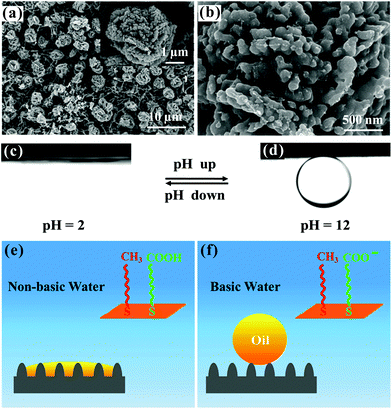 | ||
| Fig. 19 pH-induced switching of underwater superoleophobic–superoleophilic states. (a and b) SEM images of the hierarchical-structured surface at (a) low and (b) high magnification. (c and d) Oil (octane) droplets on the resultant surface in acidic (c, pH = 2) and basic (d, pH = 12) solutions. (e and f) Schematic illustration of the mechanism of the pH-dependent switchable oil-wettability. Reproduced from ref. 139 with permission from ACS, copyright 2013. | ||
Cheng et al. used the plasma polymerization method to graft a layer of pH-responsive PAA on a glass substrate (PAA-G).140 The PAA film exhibited rough nanostructures with thickness of about 64.5 nm. The underwater adhesion of the sample surface to an oil droplet could be reversibly switched by the environmental pH values. As the pH changed from 1.0 to 4.6, 5.0, 8.0 and then to 12.0, the adhesive force on a underwater oil droplet decreased from 21.6 ± 5.0 μN to 15.0 ± 2.9 μN, 2.0 ± 0.6 μN, 1.5 ± 0.4 μN and then to 0 μN. The PAA-G surface would recover high oil-adhesion once the sample was again placed in solutions with low pH value. The switch between high-oil-adhesion and low-oil-adhesion states could cycle at least 10 times. This switchability of oil-adhesion was caused by the pH-dependent chemical composition and surface roughness. When the pH was smaller than the threshold of 4.7, the neighboring carboxylic acid groups on PAA branched chains formed intramolecular hydrogen bonds, resulting in a dehydrated coiled state and a rough surface. The low water content was not enough to fully prevent the oil from wetting the PAA-G substrate. The oil droplet penetrated into and contacted with the rough microstructures of the PAA-G surface, belonging to the underwater Wenzel state. In this case, the resultant surface presented an OCA below 150° and ultrahigh adhesion to an oil droplet in water. When the pH was higher than 4.7, the intermolecular hydrogen bonds between the carboxylic acid groups on the PAA branched chains and water molecules were easier to form on the PAA surface than intramolecular hydrogen bonds. The conformation of PAA became a hydrated stretched state instead of a coiled state. The intermolecular hydrogen bonds gave rise to a high ratio of water content. As a result, a water layer was trapped between the PAA-G surface and the oil droplet, which provided a repulsive force to the oil. This underwater Cassie wetting state exhibited ultralow oil-adhesive superoleophobicity in water.
5.3 Temperature response
Chen et al. prepared a thermal-responsive PNIPAAm hydrogel.142 When the resultant sample was immersed in water, its surface wettability and adhesion to an oil droplet was found to be in response to the temperature of the surrounding water solution. When the temperature was below the LCST (∼32 °C) of PNIPAAm, the as-prepared hydrogel surface showed underwater superoleophobicity and very low oil-adhesion. The OCA and adhesive force to an underwater oil droplet at the solid/water interface were measured to be 151.7° ± 1.6° and 5.8 μN. If the environment temperature increased above the LCST, the wettability state would switch to a weak oleophobicity with high oil-adhesion in water. In this case, the OCA decreased to 127.0° ± 4.6°, while the adhesive force increased to 23.1 μN. These two different wetting states could be reversibly switched for many cycles. There is a balance between hydrophobicity and hydrophilicity in the PNIPAAm hydrogel network, i.e., hydrophobic –CH(CH3)2 and hydrophilic –CONH– groups on the side chains. In general, N–H/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups on the PNIPAAm branched chain and water molecules formed intermolecular hydrogen bonds at room temperature (<LCST), resulting in a hydrated, swollen state of the gel. The repulsive action between trapped water (polar) and oil (nonpolar) molecules endowed the hydrogel with underwater superoleophobicity and low oil-adhesive force. Above the LCST, the N–H and C
O groups on the PNIPAAm branched chain and water molecules formed intermolecular hydrogen bonds at room temperature (<LCST), resulting in a hydrated, swollen state of the gel. The repulsive action between trapped water (polar) and oil (nonpolar) molecules endowed the hydrogel with underwater superoleophobicity and low oil-adhesive force. Above the LCST, the N–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups formed intramolecular hydrogen bonds with those on neighboring branched chains, resulting in a dehydrated collapsed hydrogel. Such sample surface with low water content exhibited weaker oleophobicity and higher oil-adhesion because of the higher affinity between the hydrophobic molecular structure of PNIPAAm and oil.
O groups formed intramolecular hydrogen bonds with those on neighboring branched chains, resulting in a dehydrated collapsed hydrogel. Such sample surface with low water content exhibited weaker oleophobicity and higher oil-adhesion because of the higher affinity between the hydrophobic molecular structure of PNIPAAm and oil.
The thermal-responsive underwater oil-wettability was successfully realized by the cooperation of single PNIPAAm molecules and rough surface topography. However, the switching range of the oil-wettability of the above-mentioned smart surfaces is very limited. Liu et al. recently reported a surface that could switch between underwater superoleophobic and superoleophilic states by adjusting the water temperature.143 Both the underwater oleophilic heptadecafluorodecyltrimethoxysilane (HFMS) and the thermo-responsive PNIPAAm were modified on the surface with silicon nanowire arrays microstructure. A 2 μL underwater oil droplet (1,2-dichloroethane) on the resultant surface showed an OCA of 157° at 20 °C (Fig. 20a). However, the OCA was only about 3° at 60 °C (Fig. 20b). This switching between underwater superoleophobicity (at 20 °C) and underwater superoleophilicity (at 60 °C) could be reversibly repeated for many cycles (Fig. 20c). The grafted PNIPAAm chains were able to conceal or expose the oleophilic HFMS component through a thermo-responsive change in molecular configuration. At room temperature, the PNIPAAm chains preferred to bond with water molecules, forming intermolecular hydrogen bonding (Fig. 20d). The hydrated PNIPAAm chains extend outward and the HFMS was hidden, leading to an underwater superoleophobic surface. With the temperature increasing to 60 °C, the intramolecular hydrogen bonding of PNIPAAm chains was dominant (Fig. 20e). The grafted PNIPAAm chains thereby shrinked and became dehydrated. Therefore, the exposed HFMS chains led to an underwater superoleophilic surface.
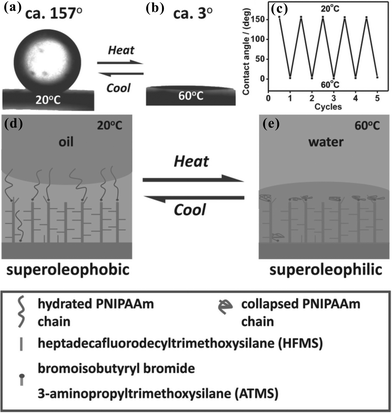 | ||
| Fig. 20 Temperature-induced switching of underwater superoleophobic–superoleophilic states. (a and b) Underwater oil droplet on the resultant substrate at (a) 20 °C and (b) 60 °C. (c) Repeatability of the switchable oil-wettability in water. (d and e) Schematic illustration of the mechanism of the temperature-dependent switchable oil-wettability. Reproduced from ref. 143 with permission from Wiley, copyright 2015. | ||
5.4 Electrical potential response
PANI is a novel conducting polymer. All of its color, conductivity and wettability show a big difference between the electrochemically reduced state and oxidized state.144 In general, there are three stable oxidation states of the PANI surface: the half-oxidized emeraldine base (EB), the fully oxidized pernigraniline form (PNB), and the fully reduced leucoemeraldine form (LEB). The EB form could be doped into the conducting state of the emeraldine salt form (ES) by a proton. Ding et al. prepared a PANI nanowire film through a galvanostatic deposition process.144 The aligned PANI nanowires were very uniform, with the length of 141.3 ± 5.0 nm and the diameter of 45.7 ± 1.7 nm (Fig. 21a). When a voltage of 0.43 V was applied to the PANI nanowire film, the sample surface showed superoleophobicity and ultralow oil-adhesion in 0.1 mol L−1 HClO4 solution (pH = 1.2). The measured OCA of the 1,2-dichloroethane droplet was 161.6° ± 1.5°, while the OSA was less than 3°. As the voltage was tuned to −0.2 V, the sample still maintained underwater superoleophobicity with the OCA of 154.2° ± 2.9°, while the oil droplet stuck tightly to the PANI nanowire film, even though the sample was tilted to any angle. The resultant surface presented a strong oil-adhesive superoleophobic state in aqueous solution. Similarly, the underwater superoleophobicity and ultrahigh oil-adhesion were also achieved at the applied potential of 0.8 V. Therefore, the oil-adhesion of the underwater superoleophobic PANI films could be controlled by adjusting the electrochemical potential (Fig. 21b). By alternately turning off and on the applied potential (−0.2 V), an underwater oil droplet could be circularly passed between two parallel PANI nanowire surfaces, as shown in Fig. 21c.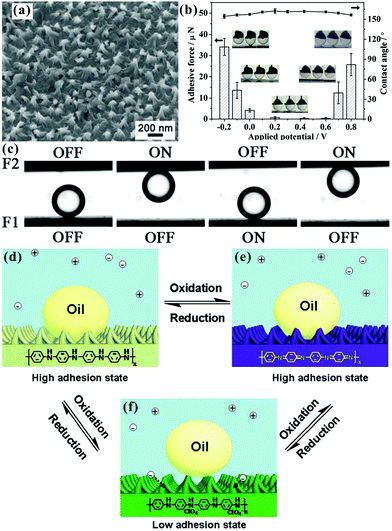 | ||
| Fig. 21 Applied electrical potential induced switchable oil-adhesion between very low and ultrahigh on the underwater superoleophobic PANI nanowire films. (a) SEM image of the PANI nanowire microstructure. (b) OCA and oil-adhesive force on the PANI nanowire film in 0.1 mol L−1 HClO4 solution, with the applied potential ranging from −0.2 V to 0.8 V. (c) Circularly transferring an underwater oil droplet between two PANI nanowire substrates by alternately turning off and on the applied potential (−0.2 V). (d–f) Schematic diagram of the switching mechanism of the oil-adhesion on the PANI nanowire film: (d) LEB state, (e) PNB state, and (f) ES state. Reproduced from ref. 144 with permission from RSC, copyright 2012. | ||
It was found that the state of the PANI nanowire film could transform from ES (Fig. 21f) to LEB (Fig. 21d) or PNB (Fig. 21e) with the electrical potential switching from 0.43 V to −0.2 V or 0.8 V, respectively. The electrochemical change usually results in a flux ion into the PANI backbone. For the PANI nanowire film at the ES state, protonation occurred at the imine nitrogen sites to yield polysemiquinone. As a result, the polaron became unrestricted and left the PANI chains. The anion species (ClO4−) had to act as counter-ions to the N+ sites to maintain charge neutrality. The molecular interaction between the PANI surface and water was enhanced. Therefore, the rough microstructures of the PANI nanowire film were fully wetted by electrolyte solution. An electrolyte solution cushion was trapped in the microstructures of the substrate surface and underneath the placed oil droplet. This underwater Cassie contact state endowed the resultant surface with a high OCA and low oil-adhesion (Fig. 21f). As the potential decreased to −0.2 V or increased to 0.8 V, the PANI form was turned from the ES state to the LEB or PNB state. With the dopant ions (ClO4−) moving out of the PANI backbone, the interaction between the PANI and water molecules was weakened, while the attraction between PANI and oil molecules was strengthened. Therefore, oil partly penetrated into the rough microstructure of the PANI nanowire films (Fig. 21d and e). An underwater transition state formed in the solid/liquid/oil system, giving rise to a high oil-adhesion.
Tian et al. developed a method to directionally drive the motion of underwater oil droplets by the combined action of electric field and gradient microstructure.313 A gradient-structured porous polystyrene film was pre-fabricated based on the breath figure process under a humidity field gradient. There were lots of micro-pores forming on the resultant surface. Their size decreased gradually from one edge to another edge on the same sample surface, arranging as a gradient distribution. The WCA of a water droplet on such a surface declined from 120° in the area with larger pores to 98° in the area with smaller pores, while the corresponding OCA of an underwater oil droplet on the surface increased from 34° to 45°. When an oil droplet (liquid paraffin) was put on the gradient-structured film in water, an unbalanced pressure was generated on both ends of the droplet due to the gradient of the porous structured film. Such an unbalanced pressure endowed the oil droplet with an asymmetrical shape and the motion trended toward a certain direction. When an electric field was added, the contact area between the underwater oil droplet and the gradient-structured porous film was decreased. Meanwhile, the viscous drag on the as-prepared surface was efficiently overcome. Once the applied voltage exceeded a threshold, the underwater oil droplet could directionally move from the large pore area to the small pore area on the sample surface. In their experiment, the driving distance of the oil droplet reached up to 5 cm.
5.5 Magnetic field response
Recently, Feng et al. demonstrated a facile method for magnetic-field-driven manipulation and transport of oil-based magnetic fluids.146 A ZnO nanorod array was grown on a glass substrate by a low-temperature hydrothermal method. The as-prepared ZnO nanoarrays consist of uniformly aligned nanorods with diameter of 150–250 nm and length of ∼6 μm. After UV treatment, the resultant ZnO nanoarrays showed superhydrophilicity and superoleophilicity in air with the WCA of 6° and OCA of 9°, respectively. When the surface was placed in water, it became superoleophobic (OCA = 156°) and could easily roll down (OSA < 5°). Two underwater superoleophobic ZnO nanoarrays were then applied to manipulate and transfer oil droplets in water medium. One substrate (top) was suspended above another one (down), allowing them to be face-to-face with an appropriate space. A magnetic fluid droplet (silicon oil of Fe3O4 nanoparticles) was injected onto the down substrate. With a periodic external magnetic field being applied to this system as the actuating force, the oil droplet could quickly respond to the magnetic stimuli and be reversibly transferred between two substrates. There was no adhesion of magnetic oil droplet during the transport process because the ZnO nanorod arrays exhibited ultralow oil-adhesive force to the magnetic oil. In contrast, if two smooth ZnO film coated substrates were used to perform the same transport experiment, an obvious delay in the movement and loss of magnetic oil would be observed.5.6 Density of surrounding water solution response
Based on the underwater superoleophobic surfaces, our group reported a simple way to achieve in situ oil droplet transportation in an aqueous environment just by switching the density of the water solution.90Fig. 22a depicts the setup for transferring the position of an oil droplet in water. It was mainly composed of a water-filled glass container and two parallel underwater superoleophobic sheets (laser ablated Si substrates). The lower sheet was fixed while the upper one could be continuously moved up and down inside the container. Fig. 22b shows the whole transportation process. At the outset, an oil droplet with the volume of 15 μL and the density of 1.26 g cm−3 was located on the surface of the lower sheet (step 1). The underwater oil droplet presented a spherical shape, due to the underwater superoleophobicity of the substrate. Then, the upper sheet was lowered slowly until it touched the top of the oil droplet (step 2). At that moment, we started to increase the density of the aqueous solution in the container by slowly adding sugar water with density of 1.52 g cm−3 into the container. After some time, the oil density was switched from higher than to lower than the surrounding water solution (step 3). With the upper sheet being lifted up, the oil droplet moved upward along with the upper sheet, just like being grappled up by a hand because the buoyancy force of the oil droplet in water was large enough to resist its gravity (step 4). Interestingly, this oil droplet could also be put back on the lower sheet. The upper sheet was moved down until the bottom of the hanging oil droplet just touched the lower sheet (step 5), and deionized water was subsequently added to the container to dilute the aqueous solution (step 6). As the density of the aqueous solution decreased and switched to be much smaller than that of oil, the water solution became clear and transparent once again. Finally, when the upper sheet was lifted again, the oil droplet was released and stayed on the lower sheet successfully (step 7). During the entire process of oil transportation, the upper underwater superoleophobic sheet was employed as a “mechanical hand” for “picking up” the underwater oil droplet from the lower sheet and then “putting it back” just through reversibly regulating the density of the surrounding water solution (Fig. 22c). This simple reversible transportation could be achieved circularly without any oil loss (Fig. 22d).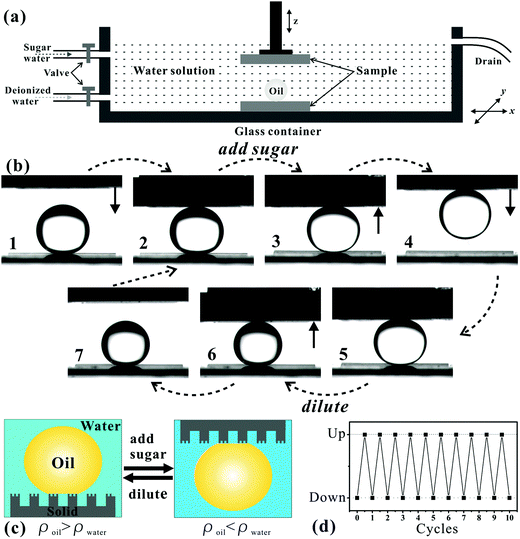 | ||
| Fig. 22 Reversible transportation of underwater oil droplet by underwater superoleophobic surfaces and reversibly switching the density of the surrounding water solution. (a) Schematic of the setup. (b) Operational process of “picking up” and “putting down” an oil droplet. The underwater oil droplet could be picked up by the mechanical hand when the density of oil was lower than that of the aqueous solution, and could also be put down when the density of the oil became higher than that of the aqueous solution again. (c and d) The repeatability of the no-loss transportation of the oil droplet in water. Reproduced from ref. 90 with permission from Wiley, copyright 2014. | ||
6. Applications
As the demand for multi-functional materials with special wettability is increasing, currently, many researchers and engineers are interested in designing and fabricating superoleophobic surfaces that have a broad range of applications. This section mainly focuses on the new and typical functions of superoleophobic surfaces, both in air and in water, including anti-oil ability,75–78 self-cleaning,79,80 oil/water separation,56,81,83–88 oil droplet manipulation,79,80,89–93 chemical shielding,75 anti-blocking,79,94 liquid microlens array,95 oil capture,96 bioadhesion,97 guiding the movement of oil droplet,92,98 and floating on oil.99,1006.1 Anti-oil coatings
Pan et al. prepared a compound coating that was made up of cross-linked PDMS and 50 wt% fluorodecyl POSS.75 After coating the compound onto stainless steel wire meshes by an electrospinning method, the metal meshes showed a rough hierarchical surface structure that had re-entrant curvature at both the macro-level (Fig. 23a) and on the micrometer scale (Fig. 23b). The interaction among the re-entrant curvature, the hierarchical rough microstructure and the low surface energy endowed the coated meshes with excellent superamphiphobicity (Fig. 23c and d). The coated surface virtually repelled all organic and inorganic liquids, including solvents and various acids/bases. In addition, the resultant surface showed low CA hysteresis and ultralow adhesion to essentially all Newtonian liquids. A series of jets of different liquids (e.g., dimethylformamide, toluene, acetic acid, hexadecane, hexylamine, PDMS) could easily bounce on the sample surface, demonstrating excellent anti-oil properties, as shown in Fig. 23d.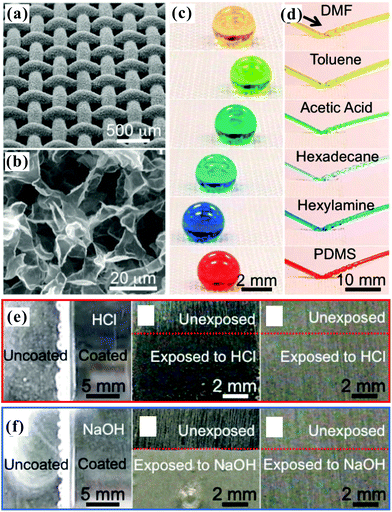 | ||
| Fig. 23 Anti-oil and chemical shielding of the superamphiphobic coating. (a) SEM image of the rough coated metal mesh. (b) SEM image of the electrospun coating on the mesh surface, showing the existence of the re-entrant curvature. (c) Various oil droplets with very high contact angles on the resultant surface. (d) Jets of various liquids bouncing on the resultant surface. (e and f) Immersion of an aluminum plate composed of half untreated domain and half coated domain into the (e) concentrated HCl and (f) concentrated NaOH solutions. Reproduced from ref. 75 with permission from ACS, copyright 2012. | ||
The underwater superoleophobic surfaces with ultralow oil-adhesion have great anti-oil ability in water.76,77 For example, the anti-oil-contamination performance of the femtosecond laser induced rough glass slide (AD = 2 μm, Fig. 15a–c) with ultralow oil-adhesive superoleophobicity underwater was investigated by our group.78 The glass slide was first dipped into a water filled beaker. Then, the dyed oil with red color was deliberately dripped on the sample surface. After taking the treated glass slide out of the beaker, we used an optical microscope to qualitatively analyze the degree of pollution; the nonstructured flat region was seriously polluted. It was found that a lot of red spots and patterns firmly adhered to this area. However, the laser structured rough region remained its initial color. There was no red imprint on the underwater superoleophobic area. The comparison result demonstrated that the laser structured underwater superoleophobic surfaces had the strong ability to repel oil contamination in water.
6.2 Self-cleaning
Since most in-air superoleophobic surfaces usually have superhydrophobicity, so such superamphiphobic surfaces also have self-cleaning ability like ordinary superhydrophobic surfaces. Fig. 24 shows the concept of the self-cleaning behavior of superamphiphobic surfaces with low liquid adhesion. Compared with a general surface, the water/oil droplet on a superamphiphobic surface shows a quasi-spherical shape. If a water/oil droplet is placed on a slightly tilted superamphiphobic surface, the droplet can easily roll away. Similar to the lotus leaf having self-cleaning ability,20,22,54,151,152,162,264 during the rolling process, the droplet will adhere and remove the foreign dirt particles on the material surface because it is easier for dust particles to stick to the liquid droplet than to the solid substrate (Fig. 24b). In this way, superamphiphobic surfaces can be kept clean. In contrast, liquid droplets just pass over the dust on the normal flat surface, leaving the dust particles behind (Fig. 24a).Underwater superoleophobic materials with ultralow oil-adhesion also have an excellent self-cleaning function.79,80 Sun et al. prepared gecko foot-like hierarchical microstructures made of PDMS by combining photolithography and soft lithography.79 After subsequent oxygen plasma treatment, the rough surface showed extreme underwater superoleophobicity, with an OCA larger than 170° and an OSA less than 1°. A soya-bean oil droplet was deliberately put onto the surface as a pollutant in an air environment. The oil quickly adhered and wetted the sample surface. Interestingly, just by immersing the polluted sample into water, the oil was completely removed (Fig. 25), whereas the oil on the untreated flat region was still retained; i.e., the oil was not washed away. This result revealed that underwater superoleophobic surfaces have strong self-cleaning ability. Although both the underwater superoleophobic surface and superhydrophobic lotus leaf have self-cleaning functions, their self-cleaning abilities are caused by different physical mechanisms. Water droplets can easily roll away on a lotus leaf, while taking away the dust particles on the leaf.20,54,128,151,162,267 However, the self-cleaning effect of underwater superoleophobic surface originates from its intrinsic superhydrophilicity, since oil can be removed by the water injection (Fig. 25a).22 Besides the surface tension of the water/oil/air interface, there is another main hydrophilic force to push the oil contamination out of the solid microstructures.79 A higher level of hydrophilicity usually results in a stronger hydrophilic force. Once the oil-polluted sample is gradually immersed in water, the water is injected into the rough microstructures and pushes the oil out, resulting in the oil impurity being cleared, as shown in Fig. 25a.
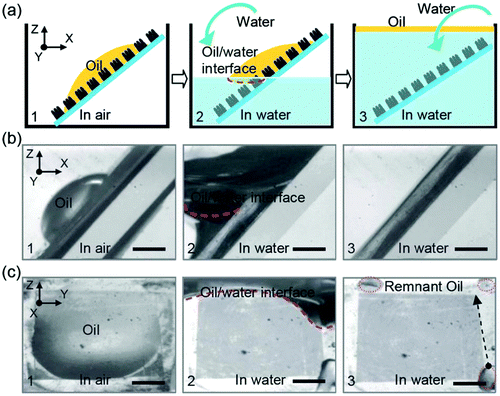 | ||
| Fig. 25 Self-cleaning ability of underwater superoleophobic surfaces. (a) Schematic diagram describing the process of removing oil contamination when a polluted underwater superoleophobic film is dipped into water. (b and c) A series of optical microscopy photos of experimental self-cleaning results observed from (b) side and (c) top-down views, respectively. Reproduced from ref. 79 with permission from RSC, copyright 2011. | ||
6.3 Oil/water separation
With the global energy demand growing, the amount of industrial oily sewage discharge continues to increase and oil spill accidents occur frequently.56,81–83 Nowadays, oil/water separation technology has become a hot research field to protect the environment and reduce economic loss. Recently, meshes or porous materials showing superhydrophobicity–superoleophilicity or superoleophobicity–superhydrophilicity have been successfully applied in oil/water separation devices because water and oil have different interface effects.56,81,84–88 In 2004, Feng et al. first separated the mixtures of water and oil by using a superhydrophobic–superoleophilic mesh film.57 Water was intercepted by the mesh for the superhydrophobicity, but oil fully wetted and quickly permeated through the resultant mesh because of the superoleophilicity, so as to achieve a separation; in fact, oil was removed from the mixture, therefore, the mesh is an “oil removing” material.56–60,301,314–319 However, the oils easily adhere to the mesh and further block it, which significantly decreases the efficiency of the separating materials.114,133,135,139,259,261,266,280,320–322 In addition, such superhydrophobic mesh films are not suited for removing light oils, which have a lower density than water, from the mixed solution.83 This is because water will sink below the light oil, generating a thick water layer underneath the oil and above the rough mesh. The water layer prevents the oil from continuing to touch and pass through the mesh film. Unfortunately, the densities of most of oils are smaller than that of water. In order to overcome the above-mentioned multiple deficiencies, in 2011, Xue et al. developed an underwater superoleophobic mesh and also achieved the function of oil/water separation.321 Nanostructured hydrogel was simply coated on a rough porous stainless steel mesh. The obtained rough mesh exhibited underwater superoleophobicity and had great ability to remove water from the mixture of water and oil. Because oil was repelled by water in rough microstructures and was difficult to really touch the mesh, the hydrogel-coated mesh could not be fouled by oil during the whole oil/water separation process. Such mesh is usually called “water removing” material because water is removed from the mixture.56 Following this strategy, a lot of underwater superoleophobic meshes or porous films were developed and successfully used to separate mixtures of water and oil.113,115,133,135,226,261,266,272,280,282,320,322–328 For example, Sun et al. prepared a graphene oxide (GO) coated rough metal mesh that finally presented underwater superoleophobicity.265 The GO-coated mesh could separate the mixture of bean oil and water that imitated culinary sewage. Jin et al. fabricated a rough Cu(OH)2 copper mesh with both superhydrophilicity and underwater superoleophobicity.113 There were abundant nanowire-hairs covering the mesh surface. Both water-rich immiscible mixtures and dispersed oil–water mixtures were separated by the rough mesh. Zhang et al. prepared a rough nitrocellulose film with microscale and nanoscale multiple pores, which showed superoleophobicity in water.114 The film could even efficiently separate the mixtures of various oils and corrosive liquids. Wen et al. obtained a zeolite-coated metal mesh that exhibited excellent superhydrophilicity and underwater superoleophobicity.328 The mixtures of various oils and water were efficiently separated by the zeolite-coated mesh. Furthermore, the underwater superoleophobicity was very stable, even in harsh environments such as acid and concentrated salt, etc. The stability made the mesh an ideal candidate in practical applications of oil/water separation.Deserts occupy a large area on the earth's surface (Fig. 26a), but are not suited for human beings. Rain droplets can instantly percolate through the sand, revealing the remarkable water absorbing ability of the sand layer. Such superhydrophilicity can possibly endow the sand layer with underwater superoleophobicity; thus, the sand is a promising candidate for application in oil/water separation. Our group found that the surface of sand particles is not smooth, but covered by macro/micro/nano-scale hierarchical rough structures (Fig. 26c–e).83 After immersion in water, the wetted sand layer exhibited underwater quasi-superoleophobicity and ultralow oil-adhesion to both heavy and light oil droplets (Fig. 26b, f and g). The measured oils included 1,2-dichloroethane, chloroform, petroleum ether, paraffin liquid, hexadecane, dodecane, decane, crude oil, diesel, sesame oil, and so on. Based on the underwater superoleophobicity of sand, we designed an oil/water separation device by using a sand layer as a separating membrane. As shown in Fig. 26h, the sand layer was fixed between two plastic tubes. A piece of cloth was put below the sand layer to avoid the loss of sand particles. Before the separation process, there was an essential step that the sand layer needed to be pre-wetted with a little water. Then, the mixture of oil (petroleum ether) and water was poured into the designed device (Fig. 26h). It was found that only the water (dyed blue) quickly permeated through the pre-wetted sand layer, whereas the oil (dyed red) was prevented and remained in the upper tube (Fig. 26i). The experimental results clearly indicate that the oil/water mixture was successfully separated by the pre-wetted sand layer. This separating process was driven by just gravity without any other external force acting on the device. In addition, the separation process could be repeated for many cycles and the designed system could work continuously. The used sand was directly obtained from the desert without any further treatment. This simple, green, almost free, large-scale, highly efficient way for oil–water separation offers a bright prospect for practically solving the pollution problems caused by oily industrial wastewater and oil spills.
 | ||
| Fig. 26 Oil/water separation based on a pre-wetted sand layer. (a) Photo of the desert. (b) Photo of underwater oil droplets on the sand layer. (c–e) SEM images of the sand particles. Shapes of (f) heavy and (g) light oil droplets on the sand layer in water. (h) Pouring the mixture of oil (dyed red) and water (dyed blue) into the designed separation device. (i) After separating the mixture of water and oil. Reproduced from ref. 83 with permission from Wiley, copyright 2016. | ||
6.4 Oil droplet manipulation
Microdroplet manipulation technology has attracted remarkable interest because of its potential applications in liquid transportation, in situ analysis, droplet based microreactors, droplet positioning, and labs-on-chips.16,90,93,162,295,299 In most cases, liquid droplets are controlled and transported by expensive and precision mechanical micro-components, such as micropumps, microvalves, and microchannels.40,64,144 The operational process is very complex and usually causes much droplet loss, due to droplet wetting and pinning.144 The emergence of controllable adhesive superhydrophobic/superoleophobic surfaces provides an alternative solution to manipulating microdroplets.53,78,80,89,91,92,294Yao et al. demonstrated an oil-based microreactor, taking advantage of the controllable oil-adhesive superoleophobic surfaces.89 An oil droplet (10 μL) containing styrene was first put on a substrate that showed superoleophobicity and very low oil-adhesion, while another brown oil droplet containing Br2 was placed on a high oil-adhesive superoleophobic substrate. The droplet containing styrene was then dragged from the substrate by a metal cap and transported to touch the Br2 droplet. These two droplets coalesced as soon as they came in contact with each other. The color of the new merged droplet gradually faded, due to the reaction between Br2 and styrene. The combined droplet was left on the sample surface with high oil-adhesion. The droplet-based microreactor systems will play an important role in enzymatic kinetics, protein crystallization, or other biochemical reactions.329
Our group prepared different hierarchical rough microstructures on glass slide surfaces by femtosecond laser ablation.78 The laser ablated surfaces showed not only superoleophobicity, but also controllable oil-adhesion that ranged from ultralow to ultrahigh in water medium. Underwater no-loss transportation of oil droplets was performed by using the underwater superoleophobic glass with very high oil-adhesion as a “mechanical hand”, as shown in Fig. 27a. A small oil droplet (8 μL) was firstly deposited on an ultralow oil-adhesive superoleophobic substrate in water (step-1), then lowered down a sticky superoleophobic surface (mechanical hand) to touch the oil droplet (step-2). After the mechanical hand just contacted the oil, we started to lift the mechanical hand up. The oil droplet left the original substrate and completely adhered to the mechanical hand for a stronger oil-adhesive force (step-3). The mechanical hand was shifted, which made the suspended oil droplet contact the other flat glass surface that had the highest oil-adhesion (step-4 and step-5). The oil droplet was released and remained on the flat glass surface when the mechanical hand was removed (step-6). The result demonstrated that the oil droplet was successfully transported from a low adhesive substrate to an ultrahigh adhesive surface. Interestingly, the whole transfer process had no oil volume loss due to the very small contact area between the oil droplet and underwater superoleophobic mechanical hand. The fusion of two oil/organic microdroplets was further realized, based on the above-mentioned droplet transfer method (Fig. 27b). No-loss oil transportation can also be carried out by other underwater superoleophobic materials, such as Ni/NiO microstructures,80 copper films,91 and silicon surfaces.92
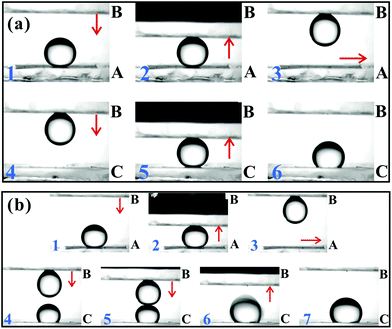 | ||
| Fig. 27 Manipulating oil droplets in a water environment by using underwater superoleophobic surfaces with different oil-adhesion properties. (a) No-loss oil droplet transportation. (b) Fusion of two small oil/organic droplets. Sample A: ultralow oil-adhesive superoleophobic surface in water. Sample B: underwater superoleophobic glass with very high oil-adhesion, which was used as a “mechanical hand”. Sample C: flat glass slide with highest oil-adhesion. The red arrows indicate the direction of movement of corresponding substrates. Reproduced from ref. 78 with permission from Springer, copyright 2015. | ||
It should be noted that the underwater superoleophobic surface with ultrahigh adhesion can only shift an oil droplet from an ultralow oil-adhesive substrate to the target surface with higher oil-adhesion. The transportation process is irreversible. The smart switchable underwater superoleophobic surfaces make it possible to manipulate an oil droplet in situ.90,144 For example, our group used two underwater superoleophobic Si sheets to simply achieve the in situ transportation of small oil droplets in water by switching the density of the water solution (Fig. 22).90 The underwater oil droplet could be “picked up” when the density of oil was smaller than that of the water solution, and be “put down/back” when the density of oil was larger than that of the water solution.
6.5 Chemical shielding (chemical resistance)
Interestingly, the superamphiphobic coating in Fig. 23a that was prepared by Pan et al. exhibited not only great anti-oil ability, but also exceptional chemical shielding function.75 For example, when an aluminum plate composed of half untreated domain and half coated domain was immersed in 12 M concentrated hydrochloric acid (Fig. 23e) and 19 M concentrated sodium hydroxide (Fig. 23f), respectively, the uncoated aluminum surface immediately reacted with the surrounding solution and released gas bubbles, while the coated domain remained calm with no perceivable chemical reactions. After immersion in concentrated HCl or concentrated NaOH for a few minutes, the uncoated aluminum surface was badly damaged. In contrast, the surface topography of the coated aluminum showed no changes on either the microscale or the nanoscale, revealing effective chemical shielding.Vahabi et al. fabricated a free-standing, flexible, superamphiphobic film, which was composed of a polyurethane layer and a layer of fluorinated silica particles.200 The film could be wrapped around spools of different sizes and delivered to the end-users, who can easily paste the film on virtually any substrate (even irregular shapes) and endow those materials surfaces with liquid-repellent ability. The chemical resistance of the as-prepared superamphiphobic film was verified by immersing two aluminum samples into concentrated sulfuric acid. One aluminum sample was covered with the flexible superamphiphobic film and another was covered with just a polyurethane layer (not superamphiphobic). The latter sample turned black quickly, as soon as it came in contact with the sulfuric acid, while the superamphiphobic film coated sample remained unaffected. It was found that the superamphiphobic film showed excellent chemical resistance in a wide variety of aqueous and organic acids and bases.
6.6 Anti-blocking
Pipe blockage, caused by discharging water with a small amount of oily impurities, usually happens both in household kitchens and in industry. The ultralow oil-adhesive underwater superoleophobic surface provides a desirable way to deal with this issue, due to its anti-blocking ability.94Fig. 28a presents a situation where water with oil impurity is flowing in a conventional pipe. The oil impurity droplets in the water flow are prone to sticking to the inside wall of the pipe because the oil droplet can fully touch the flat pipe surface. The adherent oil impurity on the inner wall will hinder water from flowing through the pipe, because the flow tendency of water is disrupted by the adherent oil impurity. The disorganized flow route slows the speed of water flow down, and reduces the efficiency of this drainage system. On the other hand, with the drainage volume of the water–oil mixture increasing, more and more free oil impurities will attach to and merge with the adherent oil droplet. After a while, the adherent oil will accumulate enough and will block the pipe. Fortunately, the blockage can be prevented by using underwater superoleophobic and ultralow oil-adhesive pipes, as shown in Fig. 28b. The inner wall of these pipes allows the oil impurity to maintain a spherical shape and roll forward along with the fluxion of water. Consequently, the flow speed of water will not be impeded or slowed down, and pipe blockage will not increase, due to the timely cleanup of the oil impurity. These drainage pipes can work in a highly effective manner for ages. Such anti-blocking ability of underwater channels can also be broadly applied in other fields. For example, if an artificial blood vessel wall possesses underwater superoleophobicity, the risk of potential fatal thrombosis and blood clots will greatly decrease.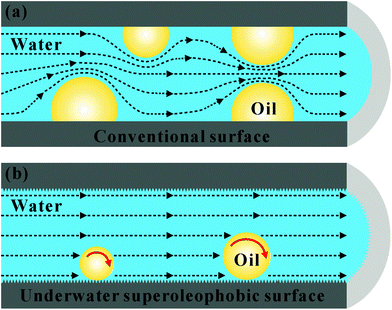 | ||
| Fig. 28 Working principle diagram of the anti-blocking function of the underwater superoleophobic drainage pipe. Water with oil impurity flows in (a) a conventional water pipe; (b) a water pipe with underwater superoleophobic inner wall. The water flow trend is indicated by black dotted lines. The red arrows in (b) show that the oil impurity droplets roll forward with water flow. Reproduced from ref. 94 with permission from RSC, copyright 2014. | ||
6.7 Liquid microlens array
As a basic optical device, lenses play a very important role in eyes, light convergence/divergence, photography, optical communication, microscopy, laser microfabrication, etc.330–333 Liquid lenses are one important form of lenses, which take advantage of the surface tension of the liquid droplet (e.g., water) to form a partial-sphere lens shape.334–336 The liquid lens has many advantages such as economical material, simple preparation process and the easy adjusting of focal length.334–338 Our group developed a novel strategy to prepare an underwater oil microlens array based on an underwater superoleophobic–oleophobic pattern, as shown in Fig. 29.95 A circle array pattern was firstly formed on a glass surface by selective femtosecond laser ablation (Fig. 29b). The pattern was composed of an untreated flat circle array and surrounding laser-induced rough microstructures (Fig. 29c–e). The femtosecond laser induced rough microstructures presented low adhesive superoleophobicity to oil droplets in water, with the OCA of 160.5° ± 2° (inset in Fig. 29d) and the OSA of 1° (inset in Fig. 29e). In contrast, the untreated bare domain only showed intrinsic weak oleophobicity with OCA of 121° ± 3° in water. The designed circle array was indeed a typical heterogeneous topography, composed of both weak underwater oleophobic circular domains and surrounding underwater superoleophobic domains. Next, the resultant sample was immersed in water, and oil droplets were placed on every circular area. An energy barrier generally exists at the boundary between the centric underwater ordinary oleophobic domain and the circumambient underwater superoleophobic domain.54,126 The energy barrier is able to prevent the oil droplet from spreading to the laser-induced region, so the oil droplets are restricted to the untreated circles (Fig. 29a). Surface tension drove the oil droplet to self-form a partial-sphere shape that could act as a lens (Fig. 29f). Since the oil droplet was immersed in water, the underwater oil lens overcame the evaporation problem, which is a common defect of liquid lenses. Moreover, the shape as well as the optical parameters of the oil lens could be simply designed by the diameter of the untreated circles and the oil volume. A uniformly arranged microlens array was further fabricated (Fig. 29f). On account of the great isolation effect of laser-induced superoleophobic domain, the oil lenses could be located very close to each other. The liquid microlens array possessed good imaging ability. When a paper with a letter “A” was put behind the microlens array plane, an array of inverted real images was clearly captured (Fig. 29g).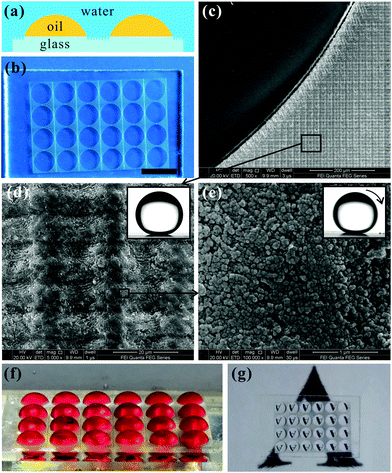 | ||
| Fig. 29 Liquid lens array fabricated by an underwater superoleophobic-oleophobic pattern. (a) Design principle. (b) Photo of a circle array pattern fabricated by the selective femtosecond laser ablation. Scale bar: 5 mm. (c) SEM image of the boundary between the untreated flat domain and the laser-induced rough domain. (d and e) SEM images of the femtosecond laser ablated rough glass slide surface. The insets reveal the static shape and rolling of an underwater oil droplet on the entire laser ablated glass slide surface. (f) Side view of the as-prepared oil lens array. (g) Imaging ability of the designed microlens array. Reproduced from ref. 95 with permission from RSC, copyright 2015. | ||
6.8 Oil capture
One of the paths for removing oil from water is using oleophilic materials to absorb oil pollution.339–344 However, such materials cannot be reused after the first oil capture, due to the enormous difficulty in removing absorbed oil, which causes a huge waste of oil and oleophilic materials. It is thus desirable to develop new oil absorbing materials that can repeatedly capture and release the oil droplet/layer. Jin et al. fabricated a superamphiphobic organosilane network surface through the phase separation reaction and subsequent 1H,1H,2H,2H-perfluorodecyltrichlorosilane grafting.96 Interestingly, the resultant surface exhibited superoleophilicity in water, which was the opposite of superoleophobicity in air. Once an underwater oil droplet contacts the surface, the oil spreads out quickly, resulting in a flat and ultrathin oil film. The as-prepared materials were applied in a designer “oil capture system”. A lot of 1,2-dichloroethane droplets with density larger than that of water were pre-sprayed on the bottom of a water-filled glass container. Next, they moved an organosilane network coated glass tube to absorb the oil droplets. These droplets were gathered and spontaneously ran into the glass tube. The underwater oil droplets were finally captured and collected. Taking advantage of the superamphiphobicity (in air) of the tube wall, the captured oil could be pushed out easily after shifting the tube to the air environment. If a superhydrophobic/superoleophilic surface was used to perform the oil capture experiment, the surface would be totally wetted and polluted by oil. Therefore, it was the integration of superoleophobicity in air and underwater superoleophilicity that endows the resultant materials with repeatable oil capture function.6.9 Bioadhesion
Previously, the surface of blood vessels was thought to be smooth; otherwise, the platelets would firmly adhere to the inner walls of the blood vessels and cause serious thrombosis problems.345 Recently, a study from Han et al. demonstrated that the inner walls of arterial vessels of rats were composed of massive amounts of aligned nanoscale grooves.346 The nanostructures not only optimize the blood fluid dynamics and thereby weaken the turbulence of blood flow, but also prevent platelets from adhering to the blood vessel wall. Furthermore, they found that the vessel surface was superhydrophilic with a WCA of about 0°. Although the underwater oil-wettability was not investigated by them, we assume that there is a strong possibility that the vessel surface will show superoleophobicity in water. The superhydrophilicity and underwater superoleophobicity of blood vessel walls are quite conducive to inhibiting protein adsorption, and thus further inhibiting the adhesion of platelets.Blood-compatible materials and devices are very important in implantation and many other medical treatments.299,345,347,348 However, most implanted materials easily absorb blood proteins and cause platelet activation and adhesion. As a result, blood coagulation and thrombosis usually occur in the repaired human and animal body. The blood compatibility of materials is determined by many factors, e.g., chemical composition, surface morphology, charge, and wettability.44,48,50,349 Particularly, wettability has a significant effect on protein adsorption, platelet activation/adhesion, and blood coagulation.4,24,46,47,49,350 Chen et al. investigated the platelet adhesion on PNIPAAm modified silicon nanowire array (SiNWAs) surfaces (SiNWA-PNIPAAm) in vitro.97 The SiNWA was obtained by chemical etching of the silicon wafer in AgNO3/HF aqueous solution. The PNIPAAm molecular layer was then coated on SiNWA using a surface-initiated atom transfer radical polymerization method. There were many silicon nanoclusters made up of several nanowires on the resultant surface. The average diameter of the nanowires was 67.9 ± 4.3 nm and the length was 25 ± 3.3 μm. The PNIPAAm coating was about 32.3 nm thick. It was found that the resultant SiNWA-PNIPAAm surface had a strong inhibition effect on the activation and adhesion of platelets. The platelet adhesion test was performed in vitro via the platelet-suspension method and inspected by SEM imaging, as shown in Fig. 30a–i. Only a few platelets adhered to the as-prepared surface, both above (37 °C) and below (20 °C) the LCST (∼32 °C) of PNIPAAm (Fig. 30e and i). In contrast, lots of platelets adhered to other surfaces, including smooth silicon wafer (Fig. 30b and f), non-grafted bare SiNWA (Fig. 30c and g), and 37 °C PNIPAAm-grafted smooth silicon wafer (Fig. 30h). When the SiNWA-PNIPAAm sample was immersed in water, its surface showed quasi-superoleophobicity, regardless of whether the temperature was lower or higher than the LCST. The oil-adhesive force was measured to be only 4.6 μN at 20 °C and 3.2 μN at 37 °C, revealing that the oil-adhesion is very low in water. The underwater quasi-superoleophobicity and ultralow oil-adhesion implied that water filled, and was trapped in, the rough microstructures of the SiNWA-PNIPAAm surface after immersion in water. Therefore, the SiNWA-PNIPAAm had a high ratio of water content. In general, the high water content in the hydration layer of the polymer outer chains could prevent the protein or cells from adsorbing (Fig. 30j and k). Therefore, the adhesion of platelets was significantly reduced on the underwater superoleophobic SiNWA-PNIPAAm surfaces.
 | ||
| Fig. 30 Anti-platelet ability of the underwater superoleophobic substrate. (a) Statistical number of adhered platelets on different surfaces at 20 °C and 37 °C, respectively. Si: flat bare silicon; SiNWA: silicon nanowire array; Si-PNIPAAm: PNIPAAm modified smooth silicon; SiNWA-PNIPAAm: PNIPAAm modified silicon nanowire array. (b–i) SEM images of the adhered platelets on different substrates: (b and f) Si, (c and g) SiNWA, (d and h) Si-PNIPAAm, and (e and i) SiNWA-PNIPAAm. The platelets in (b–e) were cultured at 20 °C, while the platelets in (f–i) were cultured at 37 °C. (j and k) Schematic illustration of the anti-platelet ability of the underwater quasi-superoleophobic SiNWA-PNIPAAm surfaces at (j) 20 °C and (k) 37 °C. Reproduced from ref. 97 with permission from ACS, copyright 2009. | ||
6.10 Guiding the movement of oil droplets
Wu et al. used the femtosecond laser to ablate a nickel substrate in sucrose solution and fabricated hierarchical microcone arrays on the nickel surface.98 The ablated surface showed superhydrophilicity and superoleophilicity in air, and superoleophobicity in water. A hollow “Y” shaped path that was like a flat canal was designed. The middle part of the path was a bare flat nickel surface without any treatment. The untreated nickel was weakly oleophobic underwater. Both sides of the path were irradiated by femtosecond laser, forming two underwater superoleophobic thick lines. In water, oil droplets were limited to the middle path and were only able to slide along the set path between two laser induced lines on both sides. The remarkable underwater superoleophobicity of the two side lines provided enough energy barrier to avoid the oil droplet derailment. When two oil droplets were placed on the two ends of the “Y” shaped path, respectively, the droplets could travel along the “Y” shaped line, and finally mixed together with the sheet being slightly inclined.6.11 Floating on oil
Most of the earth's surface is covered by liquid, such as oceans, lakes and rivers, and humans constantly think about how to float and travel on the liquid surface. In ancient times, our ancestors mastered the shipbuilding technology. They used the fabricated boats for long-distance travel, transportation, and fishing. The boats generally had a large volume and were made from lightweight materials like wood. In recent years, miniaturization has led to new advantages over past bulky devices, such as low cost, occupying less space, easy portability, and so on. Therefore, the micro-devices that can float and move on liquid surfaces have many significant potential applications such as in water environment monitoring, sensing pollution, supporting the feet of aquatic microrobots.71,99,100,351,352 In nature, the lotus leaf can float stably on the water surface, even with a heavy frog resting on it (Fig. 31a). Our recent experiment revealed that the superhydrophobicity of the upper surface not only gives the lotus leaf self-cleaning ability, but also enhances its loading capacity; i.e., it is the superhydrophobicity of the upper surface that endows the lotus leaf with very large loading capacity.71 The former keeps the lotus leaf clean, while the latter lets the lotus leaf float on the water surface and its upper surface always faces the sky. Both effects benefit its growth by allowing it to receive more sunlight and maximizing photosynthesis. Unlike the leaf-like micro-boat of the lotus leaf, the water strider can also float and even jump on the water surface without sinking, but it has only six superhydrophobic legs (Fig. 31c).158,159 In addition, it is found that even the small objects whose densities are higher than that of water can float on the surface of water. Fig. 31b and d show the photos of a metal coin and a needle floating on the water surface, respectively. | ||
| Fig. 31 Superhydrophobic or superoleophobic micro-boats floating on water or oil surfaces. (a–d) Small objects floating on water surface: (a) lotus leaf with a frog resting on it, (b) a metal coin, (c) a water strider, and (d) a metal needle. (e) Femtosecond laser induced microstructure of the upper surface of the artificial superhydrophobic PDMS micro-boat. Upper inset: a water droplet on the micro-boat surface; bottom inset: the high-magnification SEM image of the rough microstructure. (f) Artificial PDMS micro-boat floating on water surface with 5.58 g weights on its surface. (g) Cross-sectional profile of the micro-boat on the water surface. The boundary between the water surface and air is highlighted by yellow dotted lines. (h) Schematic diagram of a loaded superhydrophobic micro-boat on the water surface. (i and j) Artificial micro-boat (“oil strider”) with four superoleophobic feet floating on the oil-contaminated water surface: (i) the microstructure of the pan-shaped feet surface, (j) the photo of the oil strider. The inset shows a peanut oil droplet on the superoleophobic feet surface. (k and l) Artificial micro-boat (“oil strider”) with four underwater superoleophobic legs floating on the water/oil interface: (k) the microstructure of the wire legs, (l) the photo of the oil strider. The inset shows an oil droplet (1,2-dichloroethane) on the as-prepared copper wires. (a, b and e–h) Reproduced from ref. 71 with permission from IOP, copyright 2014. (i and j) Reproduced from ref. 100 with permission from ACS, copyright 2014. (k and l) Reproduced from ref. 99 with permission from ACS, copyright 2012. | ||
Our group has made an artificial planar superhydrophobic PDMS micro-boat that has a superhydrophobic upper surface like a lotus leaf.71 The upper surface of a PDMS wafer (with the thickness of 0.73 mm and the diameter of 4 cm) was fully ablated by femtosecond laser to form hierarchical rough microstructures (Fig. 31e). A water droplet on such a rough surface showed a WCA of 156° ± 2° (inset of Fig. 31e) and a small WSA (<4°). The micro-boat could float on the water surface effortlessly, even though it was loaded with some heavy objects, demonstrating a strong loading capacity (Fig. 31f). Although the maximum buoyancy force estimated from the same water volume of the micro-boat was only 0.92 g, the micro-boat could load 5.58 g weights. Interestingly, the micro-boat kept floating even if its top was below the water level, with a curved side water surface (Fig. 31g). Such a water/air meniscus has been also observed by other research groups.100,353 The curved convex water surface was caused by the water surface tension and the superhydrophobicity of the laser-induced rough PDMS surface. Therefore, the loading capacity was greatly increased because the curved water surface resulted in a very large total displacement. To reveal the underlying mechanism of the superhydrophobic micro-boat's loading capacity, we designed five different types of leaf-like micro-boats and compared their loading capacity. Experimental results demonstrated that the wettability of the micro-boat's lower surface had little impact on its loading capacity, while the superhydrophobicity of its upper surface, especially the superhydrophobicity at the edge of its upper surface, was responsible for the micro-boat's large loading capacity on the water surface. This effect is called the “superhydrophobic edge effect”.
As shown in Fig. 31h, the lifting forces exerted on the superhydrophobic micro-boat include the surface tension (Fσ) and the buoyancy force (Fb) acting on the boat's bottom. The maximum total lifting force is equal to the following:351,352
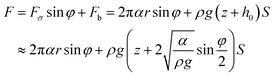 | (13) |
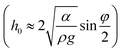 ,351,352 and S is the bottom area of the superhydrophobic micro-boat. For the wire-like micro-boats, the Fb can be neglected because of the small surface area. The lifting force that makes such a micro-boat float on water mainly comes from the surface tension. For example, surface tension endows the water strider with the ability to move on the water surface, since its legs are pretty thin.158 In contrast, for the planar leaf-like micro-boats, the perimeter 2πr ≪ 1 m, Fσ is negligible compared to Fb. Higher CA usually signifies higher φ because both of them describe the surface wettability. Therefore, the above formula reveals that the higher the hydrophobicity of the upper surface of planar micro-boats is, the larger the loading capacity that can be obtained. Superhydrophobicity of the upper surface can make φ large enough and greatly improve the loading capacity of the artificial micro-boat. Our study clarifies the buoyancy generation mechanism of the leaf-like superhydrophobic micro-boat, and is helpful in the design and manufacture of super-buoyant aquatic devices.
,351,352 and S is the bottom area of the superhydrophobic micro-boat. For the wire-like micro-boats, the Fb can be neglected because of the small surface area. The lifting force that makes such a micro-boat float on water mainly comes from the surface tension. For example, surface tension endows the water strider with the ability to move on the water surface, since its legs are pretty thin.158 In contrast, for the planar leaf-like micro-boats, the perimeter 2πr ≪ 1 m, Fσ is negligible compared to Fb. Higher CA usually signifies higher φ because both of them describe the surface wettability. Therefore, the above formula reveals that the higher the hydrophobicity of the upper surface of planar micro-boats is, the larger the loading capacity that can be obtained. Superhydrophobicity of the upper surface can make φ large enough and greatly improve the loading capacity of the artificial micro-boat. Our study clarifies the buoyancy generation mechanism of the leaf-like superhydrophobic micro-boat, and is helpful in the design and manufacture of super-buoyant aquatic devices.
Like the superhydrophobic micro-boat floating on water surface, superoleophobicity enables some micro-boats and micro-devices to float and work on oil surfaces. For example, Zhang et al. coated stainless steel meshes with a superamphiphobic layer by the processes of burning in a candle flame, silica shell deposition and then fluoroalkylsilane modification.100 The deposited soot nanoparticles with carbon cores and silica shells formed a loose network on the mesh wire surface (Fig. 31i). The resultant rough mesh showed the OCAs of 155° ± 3° to an n-hexadecane droplet and 165° ± 4° to a peanut oil droplet (inset of Fig. 31i). Both those oil droplets had OSAs less than 10° on the mesh. The disk-like superamphiphobic mesh could float on various organic liquid surfaces. The loading capacity of the artificial mesh micro-boat exceeded 15 times its weight on the n-hexadecane surface. They further designed a model device using four pan-shaped superamphiphobic meshes as feet, like an “oil strider”. The artificial oil strider not only floated on an oil-polluted surface, but also moved freely on the oil surface, guided by applying a permanent magnet (Fig. 31j). Inspired by the unique ability of the water strider to walk and jump on the surface of water, Liu et al. made an artificial “oil strider” that had four underwater superoleophobic metal “legs”.99 The legs were copper wires, which were coated by hierarchical rough copper oxide microstructures (Fig. 31k). The rough wires were fabricated through an alkali-corrosion process and exhibited underwater superoleophobicity (inset of Fig. 31k). The metal oil strider could freely float on the oil/water interface; i.e., the oil strider was underwater but on the oil surface (Fig. 31l).
7. Conclusions and future perspectives
This review systematically summarizes the recent developments of superoleophobic surfaces. Much attention is focused on the design principles, fabrication, characteristics, functions, and important applications of various superoleophobic surfaces. The deepening understanding and the constant breakthroughs of such surfaces are very meaningful for most fundamental research and a great number of commercial applications. In the past decade, many important achievements on superoleophobicity have been accomplished, such as presenting the concept of re-entrant surface curvature and opening the branch of underwater superoleophobicity. However, there are still many challenges that need to be addressed. First and foremost, more research is required to further understand the forming principle of the superoleophobic system. The fundamental theory plays a very important guiding role in fabrication and applications of superoleophobic surfaces. Secondly, the current fabrication technologies still need to be improved and perfected because most of the methods are only limited to the laboratory and are difficult to industrialize. Simple, rapid, efficient, low-cost technologies for constructuring superoleophobic interfaces are eagerly awaited. Thirdly, the poor mechanical and chemical durability severely restricts the wide applications of fabricated superoleophobic surfaces. Many surfaces easily lose their superoleophobicity after several cycles of use or being applied in harsh environments. Although a few stable and self-repairing superoleophobic surfaces were developed, there is an unmet need to design more durable superoleophobic surfaces. The underlying determinants for realizing stability need to be studied carefully, such as substrate materials, surface morphology, and surface chemistry. Last but not least, superoleophobicity by itself does not fully satisfy the needs of practical application. Endowing superoleophobic surfaces with multiple functions and smart properties is the major trend in this research field.The development trend of superoleophobicity may see an explosive growth towards the design, realization, and application of more complicated and subtle superoleophobic surfaces. With more and more scientists and engineers devoted to achieving superoleophobicity, the future of superoleophobic surfaces is bright and exciting, due to their great commercialization value and potential.
Acknowledgements
This work is supported by the National Science Foundation of China under the Grant no. 51335008, 61275008, and 61475124, the Special-funded programme on national key scientific instruments and equipment development of China under the Grant no. 2012YQ12004706, China Postdoctoral Science Foundation under the Grant no. 2016M600786, the Collaborative Innovation Center of Suzhou Nano Science and Technology and the International Joint Research Center for Micro/Nano Manufacturing and Measurement Technologies.References
- Y. Tian, B. Su and L. Jiang, Adv. Mater., 2014, 26, 6872–6897 CrossRef CAS PubMed.
- L. Wen, Y. Tian and L. Jiang, Angew. Chem., Int. Ed., 2015, 54, 3387–3399 CrossRef CAS PubMed.
- H. Bellanger, T. Darmanin, E. Taffin de Givenchy and F. Guittard, Chem. Rev., 2014, 114, 2694–2716 CrossRef CAS PubMed.
- Y. Lee, S. H. Park, K. B. Kim and J. K. Lee, Adv. Mater., 2007, 19, 2330–2335 CrossRef CAS.
- F. Chen, D. Zhang, Q. Yang, J. L. Yong, G. Du, J. Si, F. Yun and X. Hou, ACS Appl. Mater. Interfaces, 2013, 5, 6777–6792 CAS.
- J. N. Wang, Y. L. Zhang, Y. Liu, W. Zheng, L. P. Lee and H. B. Sun, Nanoscale, 2015, 7, 7101–7114 RSC.
- J. L. Yong, F. Chen, Q. Yang and X. Hou, Soft Matter, 2015, 11, 8897–8906 RSC.
- X. Yao, Y. Song and L. Jiang, Adv. Mater., 2011, 23, 719–734 CrossRef CAS PubMed.
- K. Liu, X. Yao and L. Jiang, Chem. Soc. Rev., 2010, 39, 3240–3255 RSC.
- P. Roach, N. J. Shirtcliffe and M. I. Newton, Soft Matter, 2008, 4, 224–240 RSC.
- H. Teisala, M. Tuominen and J. Kuusipalo, Adv. Mater. Interfaces, 2014, 1, 1300026 CrossRef.
- Z. Chu and S. Seeger, Chem. Soc. Rev., 2014, 43, 2784–2798 RSC.
- Z. Xue, M. Liu and L. Jiang, J. Polym. Sci., Part B: Polym. Phys., 2012, 50, 1209–1224 CrossRef CAS.
- T. Jiang, Z. Guo and W. Liu, J. Mater. Chem. A, 2015, 3, 1811–1827 CAS.
- J. Genzer and K. Efimenko, Biofouling, 2006, 22, 339–360 CrossRef CAS PubMed.
- F. Xia and L. Jiang, Adv. Mater., 2008, 20, 2842–2858 CrossRef CAS.
- X.-M. Li, D. Reinhoudt and M. Crego-Calama, Chem. Soc. Rev., 2007, 36, 1350–1368 RSC.
- Zen Yoshimitsu, Akira Nakajima, Toshiya Watanabe and Kazuhito Hashimoto, Langmuir, 2002, 18, 5818–5822 CrossRef CAS.
- T. Darmanin and F. Guittard, J. Mater. Chem. A, 2014, 2, 16319–16359 CAS.
- P. Ragesh, V. A. Ganesh, S. V. Nair and A. S. Nair, J. Mater. Chem. A, 2014, 2, 14773–14797 CAS.
- Y. Zhang, Y. Chen, L. Shi, J. Li and Z. Guo, J. Mater. Chem., 2012, 22, 799–815 RSC.
- S. Nishimoto and B. Bhushan, RSC Adv., 2013, 3, 671–690 RSC.
- K. Liu and L. Jiang, Annu. Rev. Mater. Res., 2012, 42, 231–263 CrossRef CAS.
- L. Shen, B. Wang, J. Wang, J. Fu, C. Picart and J. Ji, ACS Appl. Mater. Interfaces, 2012, 4, 4476–4483 CAS.
- M. Barberoglou, V. Zorba, E. Stratakis, E. Spanakis, P. Tzanetakis, S. H. Anastasiadis and C. Fotakis, Appl. Surf. Sci., 2009, 255, 5425–5429 CrossRef CAS.
- X. Gao, X. Yan, X. Yao, L. Xu, K. Zhang, J. Zhang, B. Yang and L. Jiang, Adv. Mater., 2007, 19, 2213–2217 CrossRef CAS.
- M. J. Kreder, J. Alvarenga, P. Kim and J. Aizenberg, Nat. Rev. Mater., 2016, 1, 15003 CrossRef CAS.
- M. Nosonovsky and V. Hejazi, ACS Nano, 2012, 6, 8488–8491 CrossRef CAS PubMed.
- J. Lv, Y. Song, L. Jiang and J. Wang, ACS Nano, 2014, 8, 3152–3169 CrossRef CAS PubMed.
- F. Shi, J. Niu, J. Liu, F. Liu, Z. Wang, X. Q. Feng and X. Zhang, Adv. Mater., 2007, 19, 2257–2261 CrossRef CAS.
- H. Dong, M. Cheng, Y. Zhang, H. Wei and F. Shi, J. Mater. Chem. A, 2013, 1, 5886–5891 CAS.
- Y. Wu, Q. Wei, M. Cai and F. Zhou, Adv. Mater. Interfaces, 2015, 2, 1400392 CrossRef.
- G. McHale, M. I. Newton and N. J. Shirtcliffe, Soft Matter, 2010, 6, 714–719 RSC.
- N. J. Shirtcliffe, G. McHale, M. I. Newton and Y. Zhang, ACS Appl. Mater. Interfaces, 2009, 1, 1316–1323 CAS.
- G. McHale, N. J. Shirtcliffe, C. R. Evans and M. I. Newton, Appl. Phys. Lett., 2009, 94, 064104 CrossRef.
- G.-H. Kim, B.-H. Lee, H. Im, S.-B. Jeon, D. Kim, M.-L. Seol, H. Hwang and Y.-K. Choi, RSC Adv., 2016, 6, 41914–41918 RSC.
- S. Wang, T. Wang, P. Ge, P. Xue, S. Ye, H. Chen, Z. Li, J. Zhang and B. Yang, Langmuir, 2015, 31, 4032–4039 CrossRef CAS PubMed.
- J. Songok, M. Tuominen, H. Teisala, J. Haapanen, J. Makela, J. Kuusipalo and M. Toivakka, ACS Appl. Mater. Interfaces, 2014, 6, 20060–20066 CAS.
- A. Ghosh, R. Ganguly, T. M. Schutzius and C. M. Megaridis, Lab Chip, 2014, 14, 1538–1550 RSC.
- B. P. Casavant, E. Berthier, A. B. Theberge, J. Berthier, S. I. Montanez-Sauri, L. L. Bischel, K. Brakke, C. J. Hedman, W. Bushman, N. P. Keller and D. J. Beebe, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 10111–10116 CrossRef CAS PubMed.
- B. Zhao, J. S. Moore and D. J. Beebe, Science, 2001, 291, 1023–1026 CrossRef CAS PubMed.
- A. Vitale, M. Quaglio, S. L. Marasso, A. Chiodoni, M. Cocuzza and R. Bongiovanni, Langmuir, 2013, 29, 15711–15718 CrossRef CAS PubMed.
- T.-i. Kim and K. Y. Suh, Soft Matter, 2009, 5, 4131–4135 RSC.
- K. W. Kwon, S. S. Choi, S. H. Lee, B. Kim, S. N. Lee, M. C. Park, P. Kim, S. Y. Hwang and K. Y. Suh, Lab Chip, 2007, 7, 1461–1468 RSC.
- H. X. Ren, X. Chen, X. J. Huang, M. Im, D. H. Kim, J. H. Lee, J. B. Yoon, N. Gu, J. H. Liu and Y. K. Choi, Lab Chip, 2009, 9, 2140–2144 RSC.
- E. P. Ivanova, J. Hasan, H. K. Webb, G. Gervinskas, S. Juodkazis, V. K. Truong, A. H. Wu, R. N. Lamb, V. A. Baulin, G. S. Watson, J. A. Watson, D. E. Mainwaring and R. J. Crawford, Nat. Commun., 2013, 4, 2838 Search PubMed.
- Q. Huang, L. Lin, Y. Yang, R. Hu, E. A. Vogler and C. Lin, Biomaterials, 2012, 33, 8213–8220 CrossRef CAS PubMed.
- A. Ranella, M. Barberoglou, S. Bakogianni, C. Fotakis and E. Stratakis, Acta Biomater., 2010, 6, 2711–2720 CrossRef CAS PubMed.
- E. Fadeeva, V. K. Truong, M. Stiesch, B. N. Chichkov, R. J. Crawford, J. Wang and E. P. Ivanova, Langmuir, 2011, 27, 3012–3019 CrossRef CAS PubMed.
- E. Stratakis, A. Ranella and C. Fotakis, Biomicrofluidics, 2011, 5, 13411 CrossRef CAS PubMed.
- X. Liu, Y. Liang, F. Zhou and W. Liu, Soft Matter, 2012, 8, 2070–2086 RSC.
- M. Wang, C. Chen, J. Ma and J. Xu, J. Mater. Chem., 2011, 21, 6962–6967 RSC.
- J. L. Yong, F. Chen, Q. Yang, D. Zhang, G. Du, J. Si, F. Yun and X. Hou, J. Phys. Chem. C, 2013, 117, 24907–24912 CAS.
- J. L. Yong, Q. Yang, F. Chen, D. Zhang, U. Farooq, G. Du and X. Hou, J. Mater. Chem. A, 2014, 2, 5499–5507 CAS.
- J. Li, Z. Jing, F. Zha, Y. Yang, Q. Wang and Z. Lei, ACS Appl. Mater. Interfaces, 2014, 6, 8868–8877 CAS.
- Z. Xue, Y. Cao, N. Liu, L. Feng and L. Jiang, J. Mater. Chem. A, 2014, 2, 2445–2460 CAS.
- L. Feng, Z. Zhang, Z. Mai, Y. Ma, B. Liu, L. Jiang and D. Zhu, Angew. Chem., Int. Ed., 2004, 43, 2012–2014 CrossRef CAS PubMed.
- S. Wang, Y. Song and L. Jiang, Nanotechnology, 2007, 18, 015103 CrossRef.
- Y. Yu, H. Chen, Y. Liu, V. Craig, L. H. Li and Y. Chen, Adv. Mater. Interfaces, 2014, 1, 1300002 CrossRef.
- J. L. Yong, Y. Fang, F. Chen, J. Huo, Q. Yang, H. Bian, G. Du and X. Hou, Appl. Surf. Sci., 2016, 389, 1148–1155 CrossRef CAS.
- N. J. Shirtcliffe, G. McHale and M. I. Newton, Langmuir, 2009, 25, 14121–14128 CrossRef CAS PubMed.
- A. Nakajima, NPG Asia Mater., 2011, 3, 49–56 CrossRef.
- D. Zahner, J. Abagat, F. Svec, J. M. Frechet and P. A. Levkin, Adv. Mater., 2011, 23, 3030–3034 CrossRef CAS PubMed.
- H. Mertaniemi, V. Jokinen, L. Sainiemi, S. Franssila, A. Marmur, O. Ikkala and R. H. Ras, Adv. Mater., 2011, 23, 2911–2914 CrossRef CAS PubMed.
- A. R. Parker and C. R. Lawrence, Nature, 2001, 414, 33–34 CrossRef CAS PubMed.
- L. Zhai, M. C. Berg, F. C. Cebeci, Y. Kim, J. M. Milwid, M. F. Rubner and R. E. Cohen, Nano Lett., 2006, 6, 1213–1217 CrossRef CAS PubMed.
- R. P. Garrod, L. G. Harris, W. C. Schofield, J. Mcgettrick, L. J. Ward, D. O. Teare and J. P. Badyal, Langmuir, 2007, 23, 689–693 CrossRef CAS PubMed.
- F. De Angelis, F. Gentile, F. Mecarini, G. Das, M. Moretti, P. Candeloro, M. L. Coluccio, G. Cojoc, A. Accardo, C. Liberale, R. P. Zaccaria, G. Perozziello, L. Tirinato, A. Toma, G. Cuda, R. Cingolani and E. Di Fabrizio, Nat. Photonics, 2011, 5, 682–687 CrossRef CAS.
- Q. X. Zhang, Y. X. Chen, Z. Guo, H. L. Liu, D. P. Wang and X. J. Huang, ACS Appl. Mater. Interfaces, 2013, 5, 10633–10642 CAS.
- B.-B. Xu, Y.-L. Zhang, W.-Y. Zhang, X.-Q. Liu, J.-N. Wang, X.-L. Zhang, D.-D. Zhang, H.-B. Jiang, R. Zhang and H.-B. Sun, Adv. Opt. Mater., 2013, 1, 56–60 CrossRef.
- J. L. Yong, Q. Yang, F. Chen, D. Zhang, G. Du, J. Si, F. Yun and X. Hou, J. Micromech. Microeng., 2014, 24, 035006 CrossRef.
- A. Tuteja, W. Choi, M. Ma, J. M. Mabry, S. A. Mazzella, G. C. Rutledge, G. H. McKinley and R. E. Cohen, Science, 2007, 318, 1618–1622 CrossRef CAS PubMed.
- A. Tuteja, W. Choi, J. M. Mabry, G. H. McKinley and R. E. Cohen, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 18200–18205 CrossRef CAS PubMed.
- http://www.surface-tension.de/ .
- S. Pan, A. K. Kota, J. M. Mabry and A. Tuteja, J. Am. Chem. Soc., 2013, 135, 578–581 CrossRef CAS PubMed.
- M. Liu, S. Wang, Z. Wei, Y. Song and L. Jiang, Adv. Mater., 2009, 21, 665–669 CrossRef CAS.
- X. Liu, J. Zhou, Z. Xue, J. Gao, J. Meng, S. Wang and L. Jiang, Adv. Mater., 2012, 24, 3401–3405 CrossRef CAS PubMed.
- J. L. Yong, F. Chen, Q. Yang, U. Farooq, H. Bian, G. Du and X. Hou, Appl. Phys. A: Mater. Sci. Process., 2015, 119, 837–844 CrossRef CAS.
- D. Wu, S. Z. Wu, Q. D. Chen, S. Zhao, H. Zhang, J. Jiao, J. A. Piersol, J. N. Wang, H. B. Sun and L. Jiang, Lab Chip, 2011, 11, 3873–3879 RSC.
- E. Zhang, Z. Cheng, T. Lv, L. Li and Y. Liu, Nanoscale, 2015, 7, 19293–19299 RSC.
- B. Wang, W. Liang, Z. Guo and W. Liu, Chem. Soc. Rev., 2015, 44, 336–361 RSC.
- Z. Chu, Y. Feng and S. Seeger, Angew. Chem., Int. Ed., 2015, 54, 2328–2338 CrossRef CAS PubMed.
- J. L. Yong, F. Chen, Q. Yang, H. Bian, G. Du, C. Shan, J. Huo, Y. Fang and X. Hou, Adv. Mater. Interfaces, 2016, 3, 1500650 CrossRef.
- K. Li, J. Ju, Z. Xue, J. Ma, L. Feng, S. Gao and L. Jiang, Nat. Commun., 2013, 4, 2276 Search PubMed.
- C. F. Wang, F. S. Tzeng, H. G. Chen and C. J. Chang, Langmuir, 2012, 28, 10015–10019 CrossRef CAS PubMed.
- W. Zhang, Z. Shi, F. Zhang, X. Liu, J. Jin and L. Jiang, Adv. Mater., 2013, 25, 2071–2076 CrossRef CAS PubMed.
- S. J. Gao, Z. Shi, W. B. Zhang, F. Zhang and J. Jin, ACS Nano, 2014, 8, 6344–6352 CrossRef CAS PubMed.
- G. Ju, M. Cheng and F. Shi, NPG Asia Mater., 2014, 6, e111 CrossRef CAS.
- X. Yao, J. Gao, Y. Song and L. Jiang, Adv. Funct. Mater., 2011, 21, 4270–4276 CrossRef CAS.
- J. L. Yong, Q. Yang, F. Chen, H. Bian, G. Du, U. Farooq and X. Hou, Adv. Mater. Interfaces, 2015, 2, 1400388 CrossRef.
- Z. Cheng, H. Liu, H. Lai, Y. Du, K. Fu, C. Li, J. Yu, N. Zhang and K. Sun, ACS Appl. Mater. Interfaces, 2015, 7, 20410–20417 CAS.
- G. Li, Z. Zhang, P. Wu, S. Wu, Y. Hu, W. Zhu, J. Li, D. Wu, X. Li and J. Chu, RSC Adv., 2016, 6, 37463–37471 RSC.
- B. Su, S. Wang, Y. Song and L. Jiang, Soft Matter, 2011, 7, 51445149 RSC.
- J. L. Yong, F. Chen, Q. Yang, D. Zhang, U. Farooq, G. Du and X. Hou, J. Mater. Chem. A, 2014, 2, 8790 CAS.
- J. L. Yong, Q. Yang, F. Chen, G. Du, C. Shan, U. Farooq, J. Wang and X. Hou, RSC Adv., 2015, 5, 40907–40911 RSC.
- M. Jin, J. Wang, X. Yao, M. Liao, Y. Zhao and L. Jiang, Adv. Mater., 2011, 23, 2861–2864 CrossRef CAS PubMed.
- L. Chen, M. Liu, H. Bai, P. Chen, F. Xia, D. Han and L. Jiang, J. Am. Chem. Soc., 2009, 131, 10467–10472 CrossRef CAS PubMed.
- G. Li, Y. Lu, P. Wu, Z. Zhang, J. Li, W. Zhu, Y. Hu, D. Wu and J. Chu, J. Mater. Chem. A, 2015, 3, 18675–18683 CAS.
- X. Liu, J. Gao, Z. Xue, L. Chen, L. Lin, L. Jiang and S. Wang, ACS Nano, 2012, 6, 5614–5620 CrossRef CAS PubMed.
- J. Zhang, X. Deng, H. J. Butt and D. Vollmer, Langmuir, 2014, 30, 10637–10642 CrossRef CAS PubMed.
- K. Tsujii, T. Yamamoto, T. Onda and S. Shibuichi, Angew. Chem., Int. Ed., 1997, 36, 1011–1012 CrossRef CAS.
- J. L. Yong, F. Chen, Q. Yang, Y. Fang, J. Huo and X. Hou, Chem. Commun., 2015, 51, 9813–9816 RSC.
- H. Zhao, K. Y. Law and V. Sambhy, Langmuir, 2011, 27, 5927–5935 CrossRef CAS PubMed.
- M. Im, H. Im, J.-H. Lee, J.-B. Yoon and Y.-K. Choi, Soft Matter, 2010, 6, 1401–1404 RSC.
- L. Yuan, T. Wu, W. Zhang, S. Ling, R. Xiang, X. Gui, Y. Zhu and Z. Tang, J. Mater. Chem. A, 2014, 2, 6952–6959 CAS.
- A. Ahuja, J. A. Taylor, V. Lifton, A. A. Sidorenko, T. R. Salamon, E. J. Lobaton, A. P. Kolodner and T. N. Krupenkin, Langmuir, 2008, 24, 9–14 CrossRef CAS PubMed.
- T. L. Liu and C. J. Kim, Science, 2014, 346, 1096–1100 CrossRef CAS PubMed.
- A. K. Kota, Y. Li, J. M. Mabry and A. Tuteja, Adv. Mater., 2012, 24, 5838–5843 CrossRef CAS PubMed.
- A. K. Kota, G. Kwon and A. Tuteja, NPG Asia Mater., 2014, 6, e109 CrossRef CAS.
- A. K. Kota, J. M. Mabry and A. Tuteja, Surf. Innovations, 2013, 1, 71–83 CrossRef CAS.
- L. Lin, M. Liu, L. Chen, P. Chen, J. Ma, D. Han and L. Jiang, Adv. Mater., 2010, 22, 4826–4830 CrossRef CAS PubMed.
- Y. Huang, M. Liu, J. Wang, J. Zhou, L. Wang, Y. Song and L. Jiang, Adv. Funct. Mater., 2011, 21, 4436–4441 CrossRef CAS.
- F. Zhang, W. B. Zhang, Z. Shi, D. Wang, J. Jin and L. Jiang, Adv. Mater., 2013, 25, 4192–4198 CrossRef CAS PubMed.
- X. Gao, L. P. Xu, Z. Xue, L. Feng, J. Peng, Y. Wen, S. Wang and X. Zhang, Adv. Mater., 2014, 26, 1771–1775 CrossRef CAS PubMed.
- K. He, H. Duan, G. Y. Chen, X. Liu, W. Yang and D. Wang, ACS Nano, 2015, 9, 9188–9198 CrossRef CAS PubMed.
- Y. Cai, Q. Lu, X. Guo, S. Wang, J. Qiao and L. Jiang, Adv. Mater., 2015, 27, 4162–4168 CrossRef CAS PubMed.
- X. Deng, L. Mammen, H. J. Butt and D. Vollmer, Science, 2012, 335, 67–70 CrossRef CAS PubMed.
- A. Steele, I. Bayer and E. Loth, Nano Lett., 2009, 9, 501–505 CrossRef CAS PubMed.
- T. Darmanin and F. Guittard, J. Am. Chem. Soc., 2009, 131, 7928–7933 CrossRef CAS PubMed.
- J. Zhang and S. Seeger, Angew. Chem., Int. Ed., 2011, 50, 6652–6656 CrossRef CAS PubMed.
- H. Wang, Y. Xue, J. Ding, L. Feng, X. Wang and T. Lin, Angew. Chem., Int. Ed., 2011, 50, 11433–11436 CrossRef CAS PubMed.
- Y. Hu, Q. Yang, F. Chen, H. Bian, Z. Deng, G. Du, J. Si, F. Yun and X. Hou, Appl. Surf. Sci., 2014, 292, 285–290 CrossRef CAS.
- A. K. Kota, G. Kwon, W. Choi, J. M. Mabry and A. Tuteja, Nat. Commun., 2012, 3, 1025 CrossRef PubMed.
- J. Yang, H. Song, B. Chen, H. Tang and C. Li, RSC Adv., 2014, 4, 14227–14232 RSC.
- H. Zhao and K. Y. Law, Langmuir, 2012, 28, 11812–11818 CrossRef CAS PubMed.
- J. L. Yong, F. Chen, Q. Yang, U. Farooq, H. Bian, G. Du and X. Hou, Appl. Phys. Lett., 2014, 105, 071608 CrossRef.
- Y. Cai, L. Lin, Z. Xue, M. Liu, S. Wang and L. Jiang, Adv. Funct. Mater., 2014, 24, 809–816 CrossRef CAS.
- P. Mazumder, Y. Jiang, D. Baker, A. Carrilero, D. Tulli, D. Infante, A. T. Hunt and V. Pruneri, Nano Lett., 2014, 14, 4677–4681 CrossRef CAS PubMed.
- J. L. Yong, F. Chen, Q. Yang, G. Du, C. Shan, H. Bian, U. Farooq and X. Hou, J. Mater. Chem. A, 2015, 3, 9379–9384 CAS.
- F. Chen, J. Song, Y. Lu, S. Huang, X. Liu, J. Sun, C. J. Carmalt, I. P. Parkin and W. Xu, J. Mater. Chem. A, 2015, 3, 20999–21008 CAS.
- H. Zhou, H. Wang, H. Niu, A. Gestos and T. Lin, Adv. Funct. Mater., 2013, 23, 1664–1670 CrossRef CAS.
- X. Wang, X. Liu, F. Zhou and W. Liu, Chem. Commun., 2011, 47, 2324–2326 RSC.
- J. Li, L. Yan, H. Li, W. Li, F. Zha and Z. Lei, J. Mater. Chem. A, 2015, 3, 14696–14702 CAS.
- J. Li, D. Li, Y. Yang, J. Li, F. Zha and Z. Lei, Green Chem., 2016, 18, 541–549 RSC.
- E. Zhang, Z. Cheng, T. Lv, Y. Qian and Y. Liu, J. Mater. Chem. A, 2015, 3, 13411–13417 CAS.
- J. L. Yong, F. Chen, Q. Yang, U. Farooq and X. Hou, J. Mater. Chem. A, 2015, 3, 10703–10709 CAS.
- H. Wang and Z. Guo, Appl. Phys. Lett., 2014, 104, 183703 CrossRef.
- M. Zhang, T. Zhang and T. Cui, Langmuir, 2011, 27, 9295–9301 CrossRef CAS PubMed.
- Z. Cheng, H. Lai, Y. Du, K. Fu, R. Hou, C. Li, N. Zhang and K. Sun, ACS Appl. Mater. Interfaces, 2014, 6, 636–641 CAS.
- Q. Cheng, M. Li, F. Yang, M. Liu, L. Li, S. Wang and L. Jiang, Soft Matter, 2012, 8, 6740–6743 RSC.
- M. Li, B. Wang, L. Heng and L. Jiang, Adv. Mater. Interfaces, 2014, 1, 1400298 CrossRef.
- L. Chen, M. Liu, L. Lin, T. Zhang, J. Ma, Y. Song and L. Jiang, Soft Matter, 2010, 6, 2708–2712 RSC.
- H. Liu, X. Zhang, S. Wang and L. Jiang, Small, 2015, 11, 3338–3342 CrossRef CAS PubMed.
- C. Ding, Y. Zhu, M. Liu, L. Feng, M. Wan and L. Jiang, Soft Matter, 2012, 8, 9064–9068 RSC.
- M. Liu, X. Liu, C. Ding, Z. Wei, Y. Zhu and L. Jiang, Soft Matter, 2011, 7, 4163–4165 RSC.
- H. Feng, X. Xu, W. Hao, Y. Du, D. Tian and L. Jiang, Phys. Chem. Chem. Phys., 2016, 18, 16202–16207 RSC.
- T. Darmanin and F. Guittard, Mater. Today, 2015, 18, 273–285 CrossRef CAS.
- B. Bhushan, Philos. Trans. R. Soc., A, 2009, 367, 1445–1486 CrossRef CAS PubMed.
- M. J. Liu, Y. M. Zheng, J. Zhai and L. Jiang, Acc. Chem. Res., 2010, 43, 368–377 CrossRef CAS PubMed.
- Y. L. Zhang, Q. D. Chen, Z. Jin, E. Kim and H. B. Sun, Nanoscale, 2012, 4, 4858–4869 RSC.
- W. Barthlott and C. Neinhuis, Planta, 1997, 202, 1–8 CrossRef CAS.
- V. Zorba, E. Stratakis, M. Barberoglou, E. Spanakis, P. Tzanetakis, S. H. Anastasiadis and C. Fotakis, Adv. Mater., 2008, 20, 4049–4054 CrossRef CAS.
- L. Feng, Y. Zhang, J. Xi, Y. Zhu, N. Wang, F. Xia and L. Jiang, Langmuir, 2008, 24, 4114–4119 CrossRef CAS PubMed.
- L. Feng, S. H. Li, Y. S. Li, H. J. Li, L. J. Zhang, J. Zhai, Y. L. Song, B. Q. Liu, L. Jiang and D. B. Zhu, Adv. Mater., 2002, 14, 1857–1860 CrossRef CAS.
- D. Wu, J.-N. Wang, S.-Z. Wu, Q.-D. Chen, S. Zhao, H. Zhang, H.-B. Sun and L. Jiang, Adv. Funct. Mater., 2011, 21, 2927–2932 CrossRef CAS.
- Y. Zheng, X. Gao and L. Jiang, Soft Matter, 2007, 3, 178–182 RSC.
- W. Barthlott, T. Schimmel, S. Wiersch, K. Koch, M. Brede, M. Barczewski, S. Walheim, A. Weis, A. Kaltenmaier, A. Leder and H. F. Bohn, Adv. Mater., 2010, 22, 2325–2328 CrossRef CAS PubMed.
- X. Gao and L. Jiang, Nature, 2004, 432, 36 CrossRef CAS PubMed.
- D. L. Hu, B. Chan and J. W. Bush, Nature, 2003, 424, 663–666 CrossRef CAS PubMed.
- K. Liu and L. Jiang, Nano Today, 2011, 6, 155–175 CrossRef CAS.
- L. Zhang, Z. Zhou, B. Cheng, J. M. Desimone and E. T. Samulski, Langmuir, 2006, 22, 8576–8580 CrossRef CAS PubMed.
- Y.-L. Zhang, H. Xia, E. Kim and H.-B. Sun, Soft Matter, 2012, 8, 11217 RSC.
- I. A. Larmour, S. E. J. Bell and G. C. Saunders, Angew. Chem., 2007, 119, 1740–1742 CrossRef.
- Y. Zhao, Y. Tang, X. Wang and T. Lin, Appl. Surf. Sci., 2010, 256, 6736–6742 CrossRef CAS.
- N. J. Shirtcliffe, G. McHale, M. I. Newton, C. C. Perry and F. B. Pyatt, Appl. Phys. Lett., 2006, 89, 104106 CrossRef.
- R. S. Seymour and S. K. Hetz, J. Exp. Biol., 2011, 214, 2175–2181 CrossRef PubMed.
- R. Helbig, J. Nickerl, C. Neinhuis and C. Werner, PLoS One, 2011, 6, e25105 CAS.
- R. Hensel, A. Finn, R. Helbig, H.-G. Braun, C. Neinhuis, W.-J. Fischer and C. Werner, Adv. Mater., 2014, 26, 2029–2033 CrossRef CAS PubMed.
- A. B. D. Cassie and S. Baxter, Trans. Faraday Soc., 1944, 40, 546 RSC.
- Q. Cheng, M. Li, Y. Zheng, B. Su, S. Wang and L. Jiang, Soft Matter, 2011, 7, 5948–5951 RSC.
- R. N. Wenzel, Ind. Eng. Chem., 1936, 28, 988–994 CrossRef CAS.
- R. J. Good, J. Am. Chem. Soc., 1952, 74, 5041–5042 CrossRef CAS.
- G. Whyman, E. Bormashenko and T. Stein, Chem. Phys. Lett., 2008, 450, 355–359 CrossRef CAS.
- E. Bormashenko, Colloids Surf., A, 2009, 345, 163–165 CrossRef CAS.
- E. Bormashenko, J. Phys. Chem. C, 2009, 113, 17275–17277 CAS.
- M. Liu and L. Jiang, Adv. Funct. Mater., 2010, 20, 3753–3764 CrossRef CAS.
- J. Li, X. Liu, Y. Ye, H. Zhou and J. Chen, J. Phys. Chem. C, 2011, 115, 4726–4729 CAS.
- Z. Cheng, M. Du, H. Lai, N. Zhang and K. Sun, Nanoscale, 2013, 5, 2776–2783 RSC.
- J. L. Yong, Q. Yang, F. Chen, D. Zhang, G. Du, H. Bian, J. Si, F. Yun and X. Hou, Appl. Surf. Sci., 2014, 288, 579–583 CrossRef CAS.
- S. Wang and L. Jiang, Adv. Mater., 2007, 19, 3423–3424 CrossRef CAS.
- S.-M. Lee and T. H. Kwon, J. Micromech. Microeng., 2007, 17, 687–692 CrossRef CAS.
- S. Tian, L. Li, W. Sun, X. Xia, D. Han, J. Li and C. Gu, Sci. Rep., 2012, 2, 511 Search PubMed.
- H. Y. Erbil and C. E. Cansoy, Langmuir, 2009, 25, 14135–14145 CrossRef CAS PubMed.
- K. Li, X. Zeng, H. Li, X. Lai, C. Ye and H. Xie, Appl. Surf. Sci., 2013, 279, 458–463 CrossRef CAS.
- A. Marmur, Soft Matter, 2012, 8, 6867–6870 RSC.
- G. Whyman and E. Bormashenko, Langmuir, 2011, 27, 8171–8176 CrossRef CAS PubMed.
- S. S. Chhatre, W. Choi, A. Tuteja, K. C. Park, J. M. Mabry, G. H. McKinley and R. E. Cohen, Langmuir, 2010, 26, 4027–4035 CrossRef CAS PubMed.
- M. Guo, B. Ding, X. Li, X. Wang, J. Yu and M. Wang, J. Phys. Chem. C, 2010, 114, 916–921 CAS.
- G.-R. Choi, J. Park, J.-W. Ha, W.-D. Kim and H. Lim, Macromol. Mater. Eng., 2010, 295, 995–1002 CrossRef CAS.
- W. Choi, A. Tuteja, S. Chhatre, J. M. Mabry, R. E. Cohen and G. H. McKinley, Adv. Mater., 2009, 21, 2190–2195 CrossRef CAS.
- J. Yang, Z. Zhang, X. Men, X. Xu and X. Zhu, New J. Chem., 2011, 35, 576–580 RSC.
- H. Meng, S. Wang, J. Xi, Z. Tang and L. Jiang, J. Phys. Chem. C, 2008, 112, 11454–11458 CAS.
- W. S. Y. Wong, G. Liu, N. Nasiri, C. Hao, Z. Wang and A. Tricoli, ACS Nano, 2017, 11, 587–596 CrossRef CAS PubMed.
- W. S. Y. Wong, G. Liu and A. Tricoli, Small, 2017, 13, 1603688 CrossRef PubMed.
- D. Wang, X. Wang, X. Liu and F. Zhou, J. Phys. Chem. C, 2010, 114, 9938–9944 CAS.
- H. Wang, Y. Xue and T. Lin, Soft Matter, 2011, 7, 8158–8161 RSC.
- S. Srinivasan, S. S. Chhatre, J. M. Mabry, R. E. Cohen and G. H. McKinley, Polymer, 2011, 52, 3209–3218 CrossRef CAS.
- K. Ellinas, M. Chatzipetrou, I. Zergioti, A. Tserepi and E. Gogolides, Adv. Mater., 2015, 27, 2231–2235 CrossRef CAS PubMed.
- S. Wooh, H. Huesmann, M. N. Tahir, M. Paven, K. Wichmann, D. Vollmer, W. Tremel, P. Papadopoulos and H. J. Butt, Adv. Mater., 2015, 27, 7338–7343 CrossRef CAS PubMed.
- H. Vahabi, W. Wang, S. Movafaghi and A. K. Kota, ACS Appl. Mater. Interfaces, 2016, 8, 21962–21967 CAS.
- Y. Lu, J. Song, X. Liu, W. Xu, Y. Xing and Z. Wei, ACS Sustainable Chem. Eng., 2013, 1, 102–109 CAS.
- S. P. P. Kobaku, A. K. Kota, D. H. Lee, J. M. Mabry and A. Tuteja, Angew. Chem., Int. Ed., 2012, 51, 10109–10113 CrossRef CAS PubMed.
- Z. He, M. Ma, X. Lan, F. Chen, K. Wang, H. Deng, Q. Zhang and Q. Fu, Soft Matter, 2011, 7, 6435–6443 RSC.
- H. Zhou, H. Wang, H. Liu and T. Lin, Sci. Rep., 2013, 3, 2964 CrossRef PubMed.
- J. Yang, Z. Zhang, X. Xu, X. Zhu, X. Men and X. Zhou, J. Mater. Chem., 2012, 22, 2834–2837 RSC.
- S. Pan, R. Guo and W. Xu, AIChE J., 2014, 60, 2752–2756 CrossRef CAS.
- S. Taleb, T. Darmanin and F. Guittard, ACS Appl. Mater. Interfaces, 2014, 6, 7953–7960 CAS.
- T. S. Wong, S. H. Kang, S. K. Tang, E. J. Smythe, B. D. Hatton, A. Grinthal and J. Aizenberg, Nature, 2011, 477, 443–447 CrossRef CAS PubMed.
- H. F. Bohn and W. Federle, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 14138–14143 CrossRef CAS PubMed.
- U. Manna and D. M. Lynn, Adv. Mater., 2015, 27, 3007–3012 CrossRef CAS PubMed.
- S. Nishioka, M. Tenjimbayashi, K. Manabe, T. Matsubayashi, K. Suwabe, K. Tsukada and S. Shiratori, RSC Adv., 2016, 6, 47579–47587 RSC.
- M. Röhrig, M. Mail, M. Schneider, H. Louvin, A. Hopf, T. Schimmel, M. Worgull and H. Hölscher, Adv. Mater. Interfaces, 2014, 1, 1300083 CrossRef.
- N. Vogel, R. A. Belisle, B. Hatton, T. S. Wong and J. Aizenberg, Nat. Commun., 2013, 4, 2167 Search PubMed.
- M. J. Hancock, K. Sekeroglu and M. C. Demirel, Adv. Funct. Mater., 2012, 22, 2223–2234 CrossRef CAS PubMed.
- Y. Fang, J. L. Yong, F. Chen, J. Huo, Q. Yang, H. Bian, G. Du and X. Hou, Appl. Phys. A: Mater. Sci. Process., 2016, 122, 827 CrossRef.
- Y. Lai, X. Gao, H. Zhuang, J. Huang, C. Lin and L. Jiang, Adv. Mater., 2009, 21, 3799–3803 CrossRef CAS.
- C. Li, R. Guo, X. Jiang, S. Hu, L. Li, X. Cao, H. Yang, Y. Song, Y. Ma and L. Jiang, Adv. Mater., 2009, 21, 4254–4258 CrossRef CAS.
- X. D. Zhao, H. M. Fan, X. Y. Liu, H. Pan and H. Y. Xu, Langmuir, 2011, 27, 3224–3228 CrossRef CAS PubMed.
- D. Zhang, F. Chen, Q. Yang, J. L. Yong, H. Bian, Y. Ou, J. Si, X. Meng and X. Hou, ACS Appl. Mater. Interfaces, 2012, 4, 4905–4912 CAS.
- M. Jin, X. Feng, J. Xi, J. Zhai, K. Cho, L. Feng and L. Jiang, Macromol. Rapid Commun., 2005, 26, 1805–1809 CrossRef CAS.
- J. L. Yong, F. Chen, Q. Yang, D. Zhang, H. Bian, G. Du, J. Si, X. Meng and X. Hou, Langmuir, 2013, 29, 3274–3279 CrossRef CAS PubMed.
- F. Chen, D. Zhang, Q. Yang, X. Wang, B. Dai, X. Li, X. Hao, Y. Ding, J. Si and X. Hou, Langmuir, 2011, 27, 359–365 CrossRef CAS PubMed.
- E. Mele, S. Girardo and D. Pisignano, Langmuir, 2012, 28, 5312–5317 CrossRef CAS PubMed.
- W. Li, G. Fang, Y. Li and G. Qiao, J. Phys. Chem. B, 2008, 112, 7234–7243 CrossRef CAS PubMed.
- M. Morita, T. Koga, H. Otsuka and A. Takahara, Langmuir, 2005, 21, 911–918 CrossRef CAS PubMed.
- J. L. Yong, Q. Yang, F. Chen, D. Zhang, G. Du, H. Bian, J. Si and X. Hou, RSC Adv., 2014, 4, 8138–8143 RSC.
- J. Y. Chung, J. P. Youngblood and C. M. Stafford, Soft Matter, 2007, 3, 1163–1169 RSC.
- K. Sekeroglu, U. A. Gurkan, U. Demirci and M. C. Demirel, Appl. Phys. Lett., 2011, 99, 63703 CrossRef PubMed.
- C. Py, P. Reverdy, L. Doppler, J. Bico, B. Roman and C. N. Baroud, Phys. Rev. Lett., 2007, 98, 156103 CrossRef PubMed.
- D. Zhang, F. Chen, Q. Yang, J. Si and X. Hou, Soft Matter, 2011, 7, 8337–8342 RSC.
- S. Neuhaus, N. D. Spencer and C. Padeste, ACS Appl. Mater. Interfaces, 2012, 4, 123–130 CAS.
- S. Z. Wu, D. Wu, J. Yao, Q. D. Chen, J. N. Wang, L. G. Niu, H. H. Fang and H. B. Sun, Langmuir, 2010, 26, 12012–12016 CrossRef CAS PubMed.
- S.-G. Park, J. H. Moon, H. C. Jeon and S.-M. Yang, Soft Matter, 2012, 8, 4567–4570 RSC.
- J. N. Wang, R. Q. Shao, Y. L. Zhang, L. Guo, H. B. Jiang, D. X. Lu and H. B. Sun, Chem. – Asian J., 2012, 7, 301–304 CrossRef CAS PubMed.
- A. T. Paxson and K. K. Varanasi, Nat. Commun., 2013, 4, 1492 CrossRef PubMed.
- W. Xu and C. H. Choi, Phys. Rev. Lett., 2012, 109, 024504 CrossRef PubMed.
- K. Golovin, D. H. Lee, J. M. Mabry and A. Tuteja, Angew. Chem., Int. Ed., 2013, 52, 13007–13011 CrossRef CAS PubMed.
- B. Li, J. Zhang, Z. Gao and Q. Wei, J. Mater. Chem. A, 2016, 4, 953–960 CAS.
- X. Deng, L. Mammen, Y. Zhao, P. Lellig, K. Müllen, C. Li, H.-J. Butt and D. Vollmer, Adv. Mater., 2011, 23, 2962–2965 CrossRef CAS PubMed.
- J. T. Han, S. Y. Kim, J. S. Woo and G.-W. Lee, Adv. Mater., 2008, 20, 3724–3727 CrossRef CAS.
- P. A. Levkin, F. Svec and J. M. Frechet, Adv. Funct. Mater., 2009, 19, 1993–1998 CrossRef CAS PubMed.
- Y. Chen, Y. Zhang, L. Shi, J. Li, Y. Xin, T. Yang and Z. Guo, Appl. Phys. Lett., 2012, 101, 033701 CrossRef.
- Y. L. Zhang, J. N. Wang, Y. He, Y. He, B. B. Xu, S. Wei and F. S. Xiao, Langmuir, 2011, 27, 12585–12590 CrossRef CAS PubMed.
- J. Bravo, L. Zhai, Z. Wu, R. E. Cohen and M. F. Rubner, Langmuir, 2007, 23, 7293–7298 CrossRef CAS PubMed.
- Y. Li, F. Liu and J. Sun, Chem. Commun., 2009, 2730–2732 RSC.
- R. G. Karunakaran, C. H. Lu, Z. Zhang and S. Yang, Langmuir, 2011, 27, 4594–4602 CrossRef CAS PubMed.
- L. Xu, R. G. Karunakaran, J. Guo and S. Yang, ACS Appl. Mater. Interfaces, 2012, 4, 1118–1125 CAS.
- Y. Liu, X. Chen and J. H. Xin, J. Mater. Chem., 2009, 19, 5602–5611 RSC.
- T. Verho, C. Bower, P. Andrew, S. Franssila, O. Ikkala and R. H. Ras, Adv. Mater., 2011, 23, 673–678 CrossRef CAS PubMed.
- C.-H. Xue and J.-Z. Ma, J. Mater. Chem. A, 2013, 1, 4146–4161 CAS.
- Z. Guo, F. Zhou, J. Hao and W. Liu, J. Am. Chem. Soc., 2005, 127, 15670–15671 CrossRef CAS PubMed.
- H. S. Lim, J. H. Baek, K. Park, H. S. Shin, J. Kim and J. H. Cho, Adv. Mater., 2010, 22, 2138–2141 CrossRef CAS PubMed.
- B. Deng, R. Cai, Y. Yu, H. Jiang, C. Wang, J. Li, L. Li, M. Yu, J. Li, L. Xie, Q. Huang and C. Fan, Adv. Mater., 2010, 22, 5473–5477 CrossRef CAS PubMed.
- B. Li and J. Zhang, Chem. Commun., 2016, 52, 2744–2747 RSC.
- J. Ou, W. Hu, S. Liu, M. Xue, F. Wang and W. Li, ACS Appl. Mater. Interfaces, 2013, 5, 10035–10041 CAS.
- H. Wang, H. Zhou, A. Gestos, J. Fang and T. Lin, ACS Appl. Mater. Interfaces, 2013, 5, 10221–10226 CAS.
- H. Wang, H. Zhou, A. Gestos, J. Fang, H. Niu, J. Ding and T. Lin, Soft Matter, 2013, 9, 277–282 RSC.
- H. Zhou, H. Wang, H. Niu, J. Fang, Y. Zhao and T. Lin, Adv. Mater. Interfaces, 2015, 2, 1400559 CrossRef.
- Z. Cheng, H. Lai, Y. Du, K. Fu, R. Hou, N. Zhang and K. Sun, ACS Appl. Mater. Interfaces, 2013, 5, 11363–11370 CAS.
- Z. Wang, L. Zhu, W. Li and H. Liu, ACS Appl. Mater. Interfaces, 2013, 5, 10904–10911 CAS.
- B. Wang and Z. Guo, Chem. Commun., 2013, 49, 9416–9418 RSC.
- Z. Cheng, C. Ding, H. Liu, Y. Zhu and L. Jiang, Nanoscale, 2014, 6, 190–194 RSC.
- M. A. Gondal, M. S. Sadullah, M. A. Dastageer, G. H. McKinley, D. Panchanathan and K. K. Varanasi, ACS Appl. Mater. Interfaces, 2014, 6, 13422–13429 CAS.
- D. Ge, L. Yang, C. Wang, E. Lee, Y. Zhang and S. Yang, Chem. Commun., 2015, 51, 6149–6152 RSC.
- Y.-Q. Liu, Y.-L. Zhang, X.-Y. Fu and H.-B. Sun, ACS Appl. Mater. Interfaces, 2015, 7, 20930–20936 CAS.
- L. Li, Z. Liu, Q. Zhang, C. Meng, T. Zhang and J. Zhai, J. Mater. Chem. A, 2015, 3, 1279–1286 CAS.
- J. L. Yong, Q. Yang, F. Chen, D. Zhang, H. Bian, Y. Ou, J. Si, G. Du and X. Hou, Appl. Phys. A: Mater. Sci. Process., 2013, 111, 243–249 CrossRef CAS.
- L. P. Xu, J. Zhao, B. Su, X. Liu, J. Peng, Y. Liu, H. Liu, G. Yang, L. Jiang, Y. Wen, X. Zhang and S. Wang, Adv. Mater., 2013, 25, 606–611 CrossRef CAS PubMed.
- L. Zhang, Y. Zhong, D. Cha and P. Wang, Sci. Rep., 2013, 3, 2326 CrossRef PubMed.
- W. Ma, H. Xu and A. Takahara, Adv. Mater. Interfaces, 2014, 1, 1300092 CrossRef.
- Y.-K. Lai, Y.-X. Tang, J.-Y. Huang, F. Pan, Z. Chen, K.-Q. Zhang, H. Fuchs and L.-F. Chi, Sci. Rep., 2013, 3, 3009 CrossRef PubMed.
- Y. Zhu, F. Zhang, D. Wang, X. F. Pei, W. Zhang and J. Jin, J. Mater. Chem. A, 2013, 1, 5758–5765 CAS.
- L. Heng, T. Guo, B. Wang, Y. Zhang and L. Jiang, RSC Adv., 2015, 5, 102378 RSC.
- M. Y. Shen, C. H. Crouch, J. E. Carey and E. Mazur, Appl. Phys. Lett., 2004, 85, 5694–5696 CrossRef CAS.
- J. Bonse, S. Baudach, J. Krüger, W. Kautek and M. Lenzner, Appl. Phys. A: Mater. Sci. Process., 2014, 74, 19–25 CrossRef.
- R. Wang, K. Hashimoto, A. Fujishima, M. Chikuni, E. Kojima, A. Kitamura, M. Shimohigoshi and T. Watanabe, Nature, 1997, 388, 431–432 CrossRef CAS.
- K. Liu, M. Cao, A. Fujishima and L. Jiang, Chem. Rev., 2014, 114, 10044–10094 CrossRef CAS PubMed.
- D. Wang, Y. Liu, X. Liu, F. Zhou, W. Liu and Q. Xue, Chem. Commun., 2009, 7018–7020 RSC.
- Y. Liu, Z. Lin, W. Lin, K. S. Moon and C. P. Wong, ACS Appl. Mater. Interfaces, 2012, 4, 3959–3964 CAS.
- D. Tian, X. Zhang, Y. Tian, Y. Wu, X. Wang, J. Zhai and L. Jiang, J. Mater. Chem., 2012, 22, 19652–19657 RSC.
- J. Yang, Z. Zhang, X. Men, X. Xu and X. Zhu, Langmuir, 2010, 26, 10198–10202 CrossRef CAS PubMed.
- Y. Sawai, S. Nishimoto, Y. Kameshima, E. Fujii and M. Miyake, Langmuir, 2013, 29, 6784–6789 CrossRef CAS PubMed.
- Y. Gao, M. Cheng, B. Wang, Z. Feng and F. Shi, Adv. Mater., 2010, 22, 5125–5128 CrossRef CAS PubMed.
- F. Xia, L. Feng, S. Wang, T. Sun, W. Song, W. Jiang and L. Jiang, Adv. Mater., 2006, 18, 432–436 CrossRef CAS.
- T. Sun, G. Wang, L. Feng, B. Liu, Y. Ma, L. Jiang and D. Zhu, Angew. Chem., Int. Ed., 2004, 43, 357–360 CrossRef CAS PubMed.
- G. Ju, M. Cheng, M. Xiao, J. Xu, K. Pan, X. Wang, Y. Zhang and F. Shi, Adv. Mater., 2013, 25, 2915–2919 CrossRef CAS PubMed.
- L. Xu, X. Lu, M. Li and Q. Lu, Adv. Mater. Interfaces, 2014, 1, 1400011 CrossRef.
- S. S. Chhatre, A. Tuteja, W. Choi, A. Revaux, D. Smith, J. M. Mabry, G. H. McKinley and R. E. Cohen, Langmuir, 2009, 25, 13625–13632 CrossRef CAS PubMed.
- X.-D. Zhao, H.-M. Fan, J. Luo, J. Ding, X.-Y. Liu, B.-S. Zou and Y.-P. Feng, Adv. Funct. Mater., 2011, 21, 184–190 CrossRef CAS.
- M. Im, D.-H. Kim, J.-H. Lee, J.-B. Yoon and Y.-K. Choi, Langmuir, 2010, 26, 12443–12447 CrossRef CAS PubMed.
- L. Xu, Q. Ye, X. Lu and Q. Lu, ACS Appl. Mater. Interfaces, 2014, 6, 14736–14743 CAS.
- X. Hong, A. X. Gao and L. Jiang, J. Am. Chem. Soc., 2007, 129, 1478–1479 CrossRef CAS PubMed.
- Z. Cheng, L. Feng and L. Jiang, Adv. Funct. Mater., 2008, 18, 3219–3225 CrossRef CAS.
- Y. Zhao, J. Fang, H. Wang, X. Wang and T. Lin, Adv. Mater., 2010, 22, 707–710 CrossRef CAS PubMed.
- D. Wu, S. Z. Wu, Q. D. Chen, Y. L. Zhang, J. Yao, X. Yao, L. G. Niu, J. N. Wang, L. Jiang and H. B. Sun, Adv. Mater., 2011, 23, 545–549 CrossRef CAS PubMed.
- S. Zhao, H. Xia, D. Wu, C. Lv, Q.-D. Chen, K. Ariga, L.-Q. Liu and H.-B. Sun, Soft Matter, 2013, 9, 4236–4240 RSC.
- F. Guo and Z. Guo, RSC Adv., 2016, 6, 36623–36641 RSC.
- T. Chen, H. Liu, H. Yang, W. Yan, W. Zhu and H. Liu, RSC Adv., 2016, 6, 43937–43949 RSC.
- T. Sun and G. Qing, Adv. Mater., 2011, 23, H57–H77 CrossRef CAS PubMed.
- Z. Hu, X. Zhang, Z. Liu, K. Huo, P. K. Chu, J. Zhai and L. Jiang, Adv. Funct. Mater., 2014, 24, 6381–6388 CrossRef CAS.
- X. Zhou, Z. Zhang, X. Xu, F. Guo, X. Zhu, X. Men and B. Ge, ACS Appl. Mater. Interfaces, 2013, 5, 7208–7214 CAS.
- C.-H. Xue, Y.-R. Li, J.-L. Hou, L. Zhang, J.-Z. Ma and S.-T. Jia, J. Mater. Chem. A, 2015, 3, 10248–10253 CAS.
- N. Sakai, A. Fujishima, T. Watanabe and K. Hashimoto, J. Phys. Chem. B, 2003, 107, 1028–1035 CrossRef CAS.
- G. Caputo, C. Nobile, T. Kipp, L. Blasi, V. Grillo, E. Carlino, L. Manna, R. Cingolani, P. D. C. And and A. Athanassiou, J. Phys. Chem. C, 2008, 112, 701–714 CAS.
- X. Chen and A. Selloni, Chem. Rev., 2014, 114, 9281–9282 CrossRef CAS PubMed.
- M. Dahl, Y. Liu and Y. Yin, Chem. Rev., 2014, 114, 9853–9889 CrossRef CAS PubMed.
- F. Zuo, L. Wang, T. Wu, Z. Zhang, B. Dan and P. Feng, J. Am. Chem. Soc., 2010, 132, 11856–11857 CrossRef CAS PubMed.
- S. Livraghi, S. Maurelli, M. C. Paganini, M. Chiesa and E. Giamello, Angew. Chem., Int. Ed., 2011, 50, 8038–8040 CrossRef CAS PubMed.
- G. Liu, L.-C. Yin, J. Wang, P. Niu, C. Zhen, Y. Xie and H.-M. Cheng, Energy Environ. Sci., 2012, 5, 9603–9610 CAS.
- A. Naldoni, M. Allieta, S. Santangelo, M. Marelli, F. Fabbri, S. Cappelli, C. L. Bianchi, R. Psaro and V. D. Santo, J. Am. Chem. Soc., 2012, 134, 7600–7603 CrossRef CAS PubMed.
- Q. Xiang, J. Yu and M. Jaroniec, J. Am. Chem. Soc., 2012, 134, 6575–6578 CrossRef CAS PubMed.
- T. R. Gordon, M. Cargnello, T. Paik, F. Mangolini, R. T. Weber, P. Fornasiero and C. B. Murray, J. Am. Chem. Soc., 2012, 134, 6751–6761 CrossRef CAS PubMed.
- D. Tian, L. He, N. Zhang, X. Zheng, Y. Dou, X. Zhang, Z. Guo and L. Jiang, Adv. Funct. Mater., 2016, 26, 7986–7992 CrossRef CAS.
- Z. Wang, Y. Xu, Y. Liu and L. Shao, J. Mater. Chem. A, 2015, 3, 12171–12178 CAS.
- S. Li, J. Huang, M. Ge, C. Cao, S. Deng, S. Zhang, G. Chen, K. Zhang, S. S. Al-Deyab and Y. Lai, Adv. Mater. Interfaces, 2015, 2, 1500220 CrossRef.
- Y. Cao, X. Zhang, L. Tao, K. Li, Z. Xue, L. Feng and Y. Wei, ACS Appl. Mater. Interfaces, 2013, 5, 4438–4442 CAS.
- J. Song, S. Huang, Y. Lu, X. Bu, J. E. Mates, A. Ghosh, R. Ganguly, C. J. Carmalt, I. P. Parkin, W. Xu and C. M. Megaridis, ACS Appl. Mater. Interfaces, 2014, 6, 19858–19865 CAS.
- J. Y. Huang, S. H. Li, M. Z. Ge, L. N. Wang, T. L. Xing, G. Q. Chen, X. F. Liu, S. S. Al-Deyab, K. Q. Zhang, T. Chen and Y. K. Lai, J. Mater. Chem. A, 2015, 3, 2825–2832 CAS.
- B. Cortese, D. Caschera, F. Federici, G. M. Ingo and G. Gigli, J. Mater. Chem. A, 2014, 2, 6781–6789 CAS.
- Q. Liu, A. A. Patel and L. Liu, ACS Appl. Mater. Interfaces, 2014, 6, 8996–9003 CAS.
- Z. Xue, S. Wang, L. Lin, L. Chen, M. Liu, L. Feng and L. Jiang, Adv. Mater., 2011, 23, 4270–4273 CrossRef CAS PubMed.
- Z. Cheng, J. Wang, H. Lai, Y. Du, R. Hou, C. Li, N. Zhang and K. Sun, Langmuir, 2015, 31, 1393–1399 CrossRef CAS PubMed.
- W. Zhang, Y. Zhu, X. Liu, D. Wang, J. Li, L. Jiang and J. Jin, Angew. Chem., Int. Ed., 2014, 53, 856–860 CrossRef CAS PubMed.
- Y. Dong, J. Li, L. Shi, X. Wang, Z. Guo and W. Liu, Chem. Commun., 2014, 50, 5586–5589 RSC.
- G. J. Dunderdale, C. Urata, T. Sato, M. W. England and A. Hozumi, ACS Appl. Mater. Interfaces, 2015, 7, 18915–18919 CAS.
- M. Tao, L. Xue, F. Liu and L. Jiang, Adv. Mater., 2014, 26, 2943–2948 CrossRef CAS PubMed.
- R. Yang, P. Moni and K. K. Gleason, Adv. Mater. Interfaces, 2015, 2, 1400489 CrossRef.
- Q. Wen, J. Di, L. Jiang, J. Yu and R. Xu, Chem. Sci., 2013, 4, 591–595 RSC.
- H. Song, D. L. Chen and R. F. Ismagilov, Angew. Chem., Int. Ed., 2006, 45, 7336–7356 CrossRef CAS PubMed.
- Y. L. Sun, W. F. Dong, R. Z. Yang, X. Meng, L. Zhang, Q. D. Chen and H. B. Sun, Angew. Chem., Int. Ed., 2012, 51, 1558–1562 CrossRef CAS PubMed.
- X. Li, Y. Ding, J. Shao, H. Tian and H. Liu, Adv. Mater., 2012, 24, OP165–OP169 CAS.
- F. Chen, Z. Deng, Q. Yang, H. Bian, G. Du, J. Si and X. Hou, Opt. Lett., 2014, 39, 606–609 CrossRef CAS PubMed.
- X. H. Lee, I. Moreno and C. C. Sun, Opt. Express, 2013, 21, 10612–10621 CrossRef PubMed.
- C. Li and H. Jiang, Appl. Phys. Lett., 2012, 100, 231105 CrossRef PubMed.
- A. O. Ashtiani and H. Jiang, Appl. Phys. Lett., 2013, 103, 111101 CrossRef.
- Y. S. Lu, H. Tu, Y. Xu and H. Jiang, Appl. Phys. Lett., 2013, 103, 261113 CrossRef PubMed.
- Z. Ding and B. Ziaie, Appl. Phys. Lett., 2009, 94, 081111 CrossRef.
- X. Jin, D. Guerrero, R. Klukas and J. F. Holzman, Appl. Phys. Lett., 2014, 105, 031102 CrossRef.
- Q. Zhu, Q. Pan and F. Liu, J. Phys. Chem. C, 2011, 115, 17464–17470 CAS.
- Y. Liu, J. Ma, T. Wu, X. Wang, G. Huang, Y. Liu, H. Qiu, Y. Li, W. Wang and J. Gao, ACS Appl. Mater. Interfaces, 2013, 5, 10018–10026 CAS.
- A. Zhang, M. Chen, C. Du, H. Guo, H. Bai and L. Li, ACS Appl. Mater. Interfaces, 2013, 5, 10201–10206 CAS.
- S. J. Choi, T. H. Kwon, H. Im, D. I. Moon, D. J. Baek, M. L. Seol, J. P. Duarte and Y. K. Choi, ACS Appl. Mater. Interfaces, 2011, 3, 4552–4556 CAS.
- Y. Gao, Y. S. Zhou, W. Xiong, M. Wang, L. Fan, H. Rabiee-Golgir, L. Jiang, W. Hou, X. Huang, L. Jiang, J. F. Silvain and Y. F. Lu, ACS Appl. Mater. Interfaces, 2014, 6, 5924–5929 CAS.
- X. Huang, B. Sun, D. Su, D. Zhao and G. Wang, J. Mater. Chem. A, 2014, 2, 7973–7979 CAS.
- T. Sun, G. Qing, B. Su and L. Jiang, Chem. Soc. Rev., 2011, 40, 2909–2921 RSC.
- Y. Mao, Q. Sun, X. Wang, Q. Ouyang, L. Han, L. Jiang and D. Han, Appl. Phys. Lett., 2009, 95, 013704 CrossRef.
- B. Chang, M. Zhang, G. Qing and T. Sun, Small, 2015, 11, 1097–1112 CrossRef CAS PubMed.
- X. Zhang, L. Wang and E. Levänen, RSC Adv., 2013, 3, 12003–12020 RSC.
- I. Banerjee, R. C. Pangule and R. S. Kane, Adv. Mater., 2011, 23, 690–718 CrossRef CAS PubMed.
- J. Y. Huang, Y. K. Lai, F. Pan, L. Yang, H. Wang, K. Q. Zhang, H. Fuchs and L. F. Chi, Small, 2014, 10, 4865–4873 CrossRef CAS PubMed.
- J. Zhao, X. Zhang, N. Chen and Q. Pan, ACS Appl. Mater. Interfaces, 2012, 4, 3706–3711 CAS.
- X. Zhang, J. Zhao, Q. Zhu, N. Chen, M. Zhang and Q. Pan, ACS Appl. Mater. Interfaces, 2011, 3, 2630–2636 CAS.
- E. Bormashenko, A. Musin, R. Grynuov and R. Pogreb, Colloid Polym. Sci., 2015, 293, 567–572 CAS.
| This journal is © The Royal Society of Chemistry 2017 |