The femtosecond-to-second photochemistry of red-shifted fast-closing anion channelrhodopsin PsACR1†
Abstract
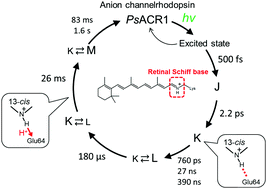
Anion channelrhodopsins (ACRs) are of great interest due to their ability to inhibit electrical signaling in optogenetic experiments. The photochemistry of ACRs is currently poorly understood and an improved understanding would be beneficial for rational design of ACRs with modified properties. Activation/deactivation of ACRs involves a series of photoreactions ranging from femtoseconds to seconds, thus real-time observation is essential to comprehend the full complexity of the photochemical processes. Here we investigate the photocycle of an ACR from Proteomonas sulcata (PsACR1), which is valuable for optogenetic applications due to the red-shifted absorption and action spectra compared to the prototype ACRs from Guillardia theta: GtACR1 and GtACR2, and the fast channel closing properties. From femto-to-submillisecond transient absorption spectroscopy, flash photolysis, and point mutations of acidic residues near the retinal Schiff base (RSB), E64, and D230, we found that the photoisomerization occurs in ∼500 fs independent of the protonation state of E64. Notably, E64 is involved in the rearrangement of the hydrogen-bond network near the RSB after photoisomerization. Furthermore, we suggest that E64 works as a primary proton acceptor during deprotonation of the RSB as has been proposed for GtACR1. Our findings allow for a deeper understanding of the photochemistry on the activation/deactivation of ACRs.
- This article is part of the themed collection: 2017 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...