H-Bonding controls the emission properties of functionalized carbon nano-dots†
Abstract
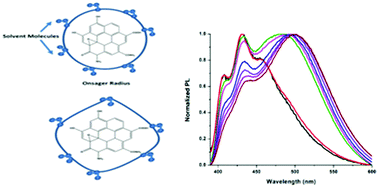
Bathochromic and hypsochromic shifts in the photo-luminescent spectra of doped and functionalized carbon nano-dots (CDs) arise due to the complex interaction between CDs and the solvent molecules around them. Nitrogen-functionalized carbon nano-dots (N-CDs) were synthesized from citric acid and urea using microwave assisted hydrothermal methods. Optical studies (absorption and photoluminescence) from the as-synthesized N-CDs were carried out in polar protic, aprotic and non-polar solvents. When excited at 355 nm, blue photoluminescence (PL) was observed from the N-CDs dispersed in polar aprotic solvents while green emission was observed in polar protic solvents. In addition to the general solvent effect, the analysis of the luminescence spectra in protic solvents suggests that hydrogen bonding plays a crucial role in regulating the photophysical characteristics of the system. Temperature dependent PL studies and time correlated single photon counting experiments in various solvent dispersions of N-CDs support the role of hydrogen bonding. This indicates that these results depend on the specific interactions observed from the N-CDs and can be thought of as the primary driving force which is then followed by solvent properties like dipole moments. Both the Lippert–Mataga model and Kamlet–Taft parameters were used to support the photophysical properties observed from N-CDs.


 Please wait while we load your content...
Please wait while we load your content...