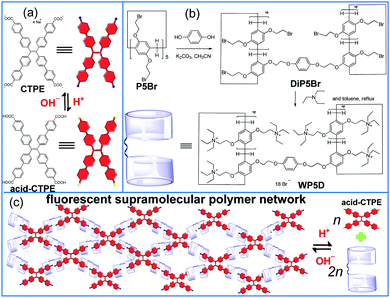
The first water-soluble pillar[5]arene dimer: synthesis and construction of a reversible fluorescent supramolecular polymer network in water†
Yan
Sun
*a,
Jin
Wang
b and
Yong
Yao
*ac
aCollege of Chemistry and Chemical Engineering, Yangzhou University, Yangzhou, Jiangsu 225002, P. R. China. E-mail: yaoyong@zju.edu.cn; ysun@iccas.ac.cn
bResearch and Development Center, China Tobacco Yunnan Industrial Co., Ltd, Kunming, 650231, China
cDepartment of Chemistry, University of Utah, Salt Lake City, 84112, USA
First published on 24th November 2016
Abstract
The first water-soluble pillar[n]arene (n = 5) dimer was successfully synthesized. This dimer can complex with carboxylate anion-functionalized tetraphenyl ethylene to form a reversible fluorescent supramolecular polymer network in water.
Macrocycles,1–5 such as crown ethers,1 cyclodextrins,2 calixarenes3 and cucurbiturils,4 have attracted considerable attention in the past century because of their various host–guest properties and broad applications. In 2008, a new class of macrocycles (i.e., pillar[n]arenes) that consist of hydroquinone units linked by methylene (–CH2–) bridges at their 2,5-positions was reported.6 Subsequently, facile and effective synthetic methods for the preparation of pillar[n]arenes were rapidly developed, and their host–guest properties were studied.7 However, due to the poor solubility of pillar[5]arenes in aqueous media, most investigations of the host–guest properties of pillar[n]arenes have been conducted in organic media, which inhibit the application of pillar[n]arenes in many areas. Therefore, to investigate the host–guest properties and expand the applications of pillar[n]arenes in water, water-soluble pillar[n]arenes were prepared.8 Several water-soluble pillar[n]arenes have been developed by introducing negatively charged groups, such as carboxylate and phosphate anions, or positively charged groups, such as ammonium, pyridinium, and imidazolium salt, at both rims of the pillar[n]arenes.9
The essential function of macrocycle dimers is a cooperative effect. In comparison to monomers, dimers often form stable complexes with guests via intermolecular cooperative binding.10 To the best of our knowledge, the water-soluble pillar[n]arene dimer has not been reported, and the dimer may exhibit properties that are different from those of previously reported water-soluble pillar[n]arenes. Herein, we report the synthesis of the first water-soluble pillar[5]arene dimer (WP5D) and demonstrate that WP5D complexes with carboxylate anion-functionalized tetraphenyl ethylene (CTPE) to form a fluorescent supramolecular polymer network in water that is driven by hydrophobic and electrostatic interactions (Scheme 1a).
WP5D was prepared using a facile approach (Scheme 1b). To prepare WP5D, DiP5Br was prepared by refluxing excess P5Br (10 equiv.) and hydroquinone in CH3CN with K2CO3 as the base for 24 h. Then, due to the treatment of DiP5Br with excess triethylamine (30 equiv.) in toluene, WP5D containing ten triethylammonium groups at both rims was prepared as a light-yellow solid (95% yield).
Based on previous studies, WP5, which possesses positively charged groups, can efficiently complex with negatively charged guest molecules.8 In this communication, we investigated the host–guest interactions between a model WP5 and a guest (Fig. S6, ESI†). Then, we can easily construct a fluorescent supramolecular polymer network in water by mixing WP5D and CTPE in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio.
1 ratio.
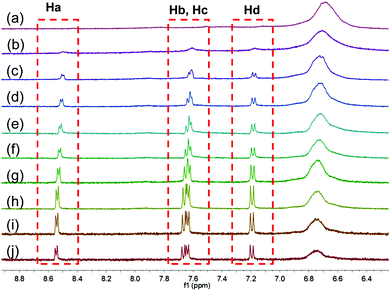
The formation of the supramolecular polymer network was characterized by various techniques including 1H NMR spectroscopy (1H-NMR), two-dimensional diffusion-ordered 1H NMR spectroscopy (DOSY), specific viscosity and scanning electron microscopy (SEM). The 1H NMR spectra of (WP5D)2·CTPE were recorded over a concentration range of 1.00 mM to 200 mM (Fig. 1 and Fig. S7, ESI†). As expected, the proton NMR spectra of (WP5D)2·CTPE are concentration dependent. As the concentration increased, the signals of protons Ha, Hb, Hc, and Hd of CTPE underwent substantial upfield shifts. Moreover, all the signals broadened at a high concentration, which confirmed the formation of high molecular weight aggregates.11
DOSY experiments were performed to investigate the aggregation of (WP5D)2·CTPE supramolecular polymer networks. The measured weight-average diffusion coefficient (D) of (WP5D)2·CTPE in water decreased gradually from 7.42 × 10−10 m2 s−1 to 2.34 × 10−11 m2 s−1 as the concentration of (WP5D)2·CTPE increased from 1.00 mM to 200 mM, which implied that the increase in the average aggregation size led to the transition from supramolecular oligomer species to supramolecular polymer networks (Fig. S8, ESI†). Based on previous studies,12 a significant decrease in the diffusion coefficient results from a high polymerization degree. Therefore, the current measurements indicate the formation of an extended high molecular weight polymeric structure.
Viscosimetry is an efficient method for evaluating the propensity of monomers to self-assemble into large aggregates. Therefore, capillary viscosity measurements were carried out in water using a Cannon Ubbelohde semimicrodilution viscometer. As shown in Fig. 2, the (WP5D)2·CTPE supramolecular polymer networks that were assembled from the monomers exhibited a viscosity transition that is characterized by a change in the slope in the double logarithmic plots of the specific viscosity as a function of the concentration. In the low concentration range, the slopes approximated unity, demonstrating a linear relationship between the specific viscosity and concentration that is characteristic of non-interacting assemblies with a constant size. When the concentration exceeded the critical polymerization concentration (CPC; at approximately 23.0 mM), a sharp increase in viscosity was observed.
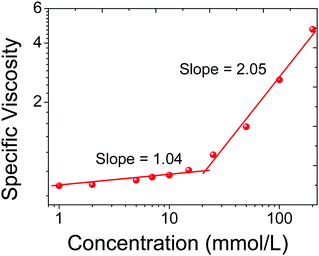 | ||
Fig. 2 Specific viscosity (298 K) of a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar mixture of WP5D and CTPE in water as a function of the concentration. Here, the concentration is the original TPE′ concentration. 1 molar mixture of WP5D and CTPE in water as a function of the concentration. Here, the concentration is the original TPE′ concentration. | ||
Furthermore, a Dynamic Light Scattering (DLS) experiment, which is a technique useful for determining the size distribution profile of a supramolecular system, was performed to study the size distributions of our supramolecular polymer networks. The average hydrodynamic diameters (Dh) of WP5D (50 mM) and CTPE (25 mM) were determined to be ∼4.50 nm and 2.00 nm. After these two components were mixed, the corresponding Dh value increased to 865 nm, which is consistent with the previously mentioned DOSY experiments. This result indicates the formation of supramolecular polymer networks with a high molecular weight (Fig. S9, ESI†).
In addition, SEM studies demonstrated that rod-like fibres can be easily drawn from a highly concentrated solution of (WP5D)2·CTPE supramolecular polymer networks, indicating a significant chain extension that generates the desired high molecular weight polymeric aggregates (Fig. 3). All these images confirmed the formation of a supramolecular polymer network driven by pillar[5]arene-based molecular recognition in water.
 | ||
| Fig. 3 SEM images of a rod-like fibre drawn from a high concentration solution of (WP5D)2·CTPE supramolecular polymer networks in water. | ||
Fluorescence experiments were conducted to investigate the fluorescence response of WP5D to CTPE. Fig. 4a shows the fluorescence spectra of CTPE upon addition of WP5D. CTPE initially exhibited very low fluorescence intensity at 515 nm. However, after the addition of WP5D, the fluorescence intensity increased sharply because the innate torsional conformation of CTPE was restrained by the host–guest interactions (Fig. 4a, blue line, and Fig. 4b, the third vial).13 Then, the addition of H+ induced the protonation of CTPE into acid-CTPE followed by disassociation from the cavity of WP5D. This further increased fluorescence intensity was due to the poor solubility of acid-CTPE in water and is referred to as aggregation-induced emission (Fig. 4a, black line, and Fig. 4b, the fourth vial).14 After filtration, the acid-CTPE precipitates were separated from the solution, and the fluorescence intensity of the solution decreased sharply, which further confirmed the H+ induced disassociation. However, if we added OH− directly to the solution of WP5D and CTPE by treating with H+, the fluorescence emission decreased similarly to polymer networks (Fig. 4a, brown line). The optical images of this process also confirmed the reversible formation of a fluorescent supramolecular polymer network in water by the alternative addition of H+ and OH−.
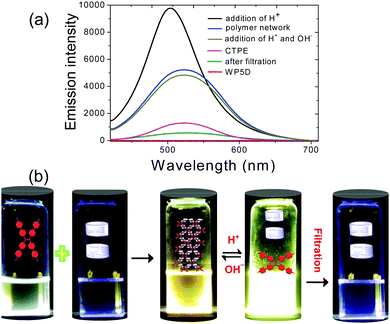 | ||
| Fig. 4 Fluorescence emission spectra (a) and optical images (b) of the formation of a fluorescent supramolecular polymer network in water by alternative addition of H+ and OH−. | ||
In conclusion, we have synthesized the first water-soluble pillar[5]arene dimer and constructed a fluorescent supramolecular binary network due to the complexation of WP5D with CTPE, which was driven by hydrophobic and electrostatic interactions. The intramolecular rotation of the phenyl rings of CTPE was hampered by the addition of WP5D. Therefore, the complex emits strong fluorescence. Due to the pH responsiveness of the pillar[5]arene-based host–guest interactions in water, the fluorescence of the supramolecular binary network can be tuned by changing the solution pH. We believe that the reversible nature of the noncovalent assembly and the special water-soluble property will promise substantial applications in soft material systems.
This work was supported by the National Natural Science Foundation of China (21503185).
Notes and references
- (a) B. Zheng, F. Wang, S. Dong and F. Huang, Chem. Soc. Rev., 2012, 41, 1621–1636 RSC; (b) Z. Ge, J. Hu, F. Huang and S. Liu, Angew. Chem., Int. Ed., 2009, 48, 1798–1802 CrossRef CAS PubMed.
- (a) Y. Wei, D. Sun, D. Yuan, Y. Liu, Y. Zhao, X. Li, S. Wang, J. Dou, X. Wang, A. Hao and D. Sun, Chem. Sci., 2012, 3, 2282–2287 RSC; (b) Q.-W. Zhang, D. Li, X. Li, P. B. White, J. Mecinovic, X. Ma, H. Ågren, R. J. M. Nolte and H. Tian, J. Am. Chem. Soc., 2016, 138, 13541–13550 CrossRef CAS PubMed.
- (a) Y.-X. Wang, Y.-M. Zhang and Y. Liu, J. Am. Chem. Soc., 2015, 137, 4543–4549 CrossRef CAS PubMed; (b) L. Kaufmann, S. R. Kennedy, C. D. Jonesb and J. W. Steed, Chem. Commun., 2016, 52, 10113–10116 RSC; (c) H. Li, L.-L. Tan, P. Jia, Q.-L. Li, Y.-L. Sun, J. Zhang, Y.-Q. Ning, J. Yu and Y.-W. Yang, Chem. Sci., 2014, 5, 2804–2808 RSC.
- (a) X. Ling, S. Saretz, L. Xiao, J. Francescon and E. Masson, Chem. Sci., 2016, 7, 3569–3573 RSC; (b) Y. Jiao, W.-L. Li, J.-F. Xu, G. Wang, J. Li, Z. Wang and X. Zhang, Angew. Chem., Int. Ed., 2016, 55, 8933–8937 CrossRef CAS PubMed.
- (a) K. Jie, Y. Zhou, Y. Yao and F. Huang, Chem. Soc. Rev., 2015, 44, 3568–3587 RSC; (b) I. R. Fernando, M. Frasconi, Y. Wu, W.-G. Liu, M. R. Wasielewski, W. A. Goddard and J. F. Stoddart, J. Am. Chem. Soc., 2016, 138, 10214–10225 CrossRef CAS PubMed; (c) S. K. M. Nalluri, Z. Liu, Y. Wu, K. R. Hermann, A. Samanta, D. J. Kim, M. D. Krzyaniak, M. R. Wasielewski and J. F. Stoddart, J. Am. Chem. Soc., 2016, 138, 5968–5977 CrossRef CAS PubMed; (d) H. Chen, J. Fan, X. Hu, J. Ma, S. Wang, J. Li, Y. Yu, X. Jia and C. Li, Chem. Sci., 2015, 6, 197–202 RSC; (e) J. Ma, Q. Meng, X. Hu, B. Li, S. Ma, B. Hu, J. Li, X. Jia and C. Li, Org. Lett., 2016, 18, 5740–5743 CrossRef CAS PubMed.
- T. Ogoshi, S. Kanai, S. Fujinami, T.-A. Yamagishi and Y. Nakamoto, J. Am. Chem. Soc., 2008, 130, 5022–5023 CrossRef CAS PubMed.
- (a) T. Ogoshi, T. Aoki, K. Kitajima, S. Fujinami, T.-A. Yamagishi and Y. Nakamoto, J. Org. Chem., 2011, 76, 328–331 CrossRef CAS PubMed; (b) N. Sun, X. Xiao and J. Jiang, Polym. Chem., 2015, 6, 5015–5020 RSC; (c) Y. Fang, C. Li, L. Wu, B. Bai, X. Li, Y. Jia, W. Feng and L. Yuan, Dalton Trans., 2015, 44, 14584–14588 RSC; (d) W.-B. Hu, W.-J. Hu, X.-L. Zhao, Y. A. Liu, J.-S. Li, B. Jiang and K. Wen, Chem. Commun., 2015, 51, 13882–13885 RSC; (e) N. Laggoune, F. Delattre, J. Lyskawa, F. Stoffelbach, J. M. Guigner, S. Ruellan, G. Cooke and P. Woisel, Polym. Chem., 2015, 6, 7389–7394 RSC; (f) N. Galanos, E. Gillon, A. Imberty, S. E. Matthewsc and S. Vidal, Org. Biomol. Chem., 2016, 14, 3476–3481 RSC; (g) Y. Chen, L. Rui, L. Liu and W. Zhang, Polym. Chem., 2016, 7, 3268–3276 RSC; (h) K. Eichstaedt, B. Wicher, M. Gdaniecc and T. Połoński, CrystEngComm, 2016, 18, 5807–5810 RSC; (i) X.-B. Hu, Z. Chen, L. Chen, L. Zhang, J.-L. Hou and Z.-T. Li, Chem. Commun., 2012, 48, 10999–11001 RSC; (j) C. Li, Chem. Commun., 2014, 50, 12420–12433 RSC; (k) Y. Wang, G. Ping and C. Li, Chem. Commun., 2016, 52, 9858–9872 RSC; (l) T. Ogoshi, H. Kayama, D. Yamafuji, T. Aoki and T.-A. Yamagish, Chem. Sci., 2012, 3, 3221–3226 RSC; (m) D. Cao, Y. Kou, J. Liang, Z. Chen, L. Wang and H. Meier, Angew. Chem., Int. Ed., 2009, 48, 9721–9723 CrossRef CAS PubMed; (n) T. Ogoshi, R. Sueto, K. Yoshikoshi, K. Yasuhara and T.-A. Yamagishi, J. Am. Chem. Soc., 2016, 138, 8064–8067 CrossRef CAS PubMed; (o) G. Yu, C. Han, Z. Zhang, J. Chen, X. Zhan, B. Zheng, S. Liu and F. Huang, J. Am. Chem. Soc., 2012, 134, 8711–8717 CrossRef CAS PubMed; (p) C. Li, K. Han, J. Li, Y. Zhang, W. Chen, Y. Yu and X. Jia, Chem. – Eur. J., 2013, 19, 11892–11897 CrossRef CAS PubMed; (q) X. Wang, K. Han, J. Li, X. Jia and C. Li, Polym. Chem., 2013, 4, 3998–4003 RSC.
- (a) T. Ogoshi, M. Hashizume, T.-A. Yamagishi and Y. Nakamoto, Chem. Commun., 2010, 46, 3708–3710 RSC; (b) Y. Yao, Y. Wang and F. Huang, Chem. Sci., 2014, 5, 4312–4316 RSC; (c) Y. Yao, X. Chi, Y. Zhou and F. Huang, Chem. Sci., 2014, 5, 4312–4316 RSC.
- (a) L.-L. Tan, H. Li, Y.-C. Qiu, D.-X. Chen, X. Wang, R.-Y. Pan, Y. Wang, S. X.-A. Zhang, B. Wang and Y.-W. Yang, Chem. Sci., 2015, 6, 1640–1644 RSC; (b) C. D. Ding, Y. Liu, T. Wang and J. Fu, J. Mater. Chem. B, 2016, 4, 2819–2827 RSC; (c) F. Zhang, J. Ma, Y. Sun, I. Boussouar, D. Tian, H. Li and L. Jiang, Chem. Sci., 2016, 7, 3227–3233 RSC; (d) Y. Chang, K. Yang, P. Wei, S. Huang, Y. Pei, W. Zhao and Z. Pei, Angew. Chem., Int. Ed., 2014, 53, 13126–13130 CrossRef CAS PubMed; (e) S. Guo, X. Liu, C. Yao, C. Lu, Q. Chen, X.-Y. Hu and L. Wang, Chem. Commun., 2016, 52, 10751–10754 RSC; (f) Y. Yao, M. Xue, X. Chi, Y. Ma, J. He, Z. Abliz and F. Huang, Chem. Commun., 2012, 48, 6505–6507 RSC; (g) Y. Sun, F. Guo, T. Zuo, J. Hua and G. Diao, Nat. Commun., 2016, 7, 12042, DOI:10.1038/ncomms12042.
- (a) Y. Li, M. Pink, J. A. Karty and A. H. Flood, J. Am. Chem. Soc., 2008, 130, 17293–17295 CrossRef CAS PubMed; (b) Y. Liu and Y. Chen, Acc. Chem. Res., 2006, 39, 681–691 CrossRef CAS PubMed.
- (a) Z. Zhang, C. Han, G. Yu and F. Huang, Chem. Sci., 2012, 3, 3026 RSC; (b) Y. Zhou, K. Jie, B. Shi and Y. Yao, Chem. Commun., 2015, 51, 11112 RSC.
- P. Wei, B. Xia, Y. Zhang, Y. Yu and X. Yan, Chem. Commun., 2014, 50, 3973 RSC.
- (a) R. Chen, H. Jiang, H. Gu, Q. Zhou, J. Wu, D. Chen and J. Zhang, Chem. Commun., 2015, 51, 12220–12223 RSC; (b) X. Yan, M. Wang, T. R. Cook, M. Zhang, M. L. Saha, Z. Zhou, X. Li, F. Huang and P. J. Stang, J. Am. Chem. Soc., 2016, 138, 4580–4588 CrossRef CAS PubMed.
- (a) Y. Hong, J. W. Y. Lamab and B. Z. Tang, Chem. Soc. Rev., 2011, 40, 5361–5388 RSC; (b) Z. Zhao, B. Hea and B. Z. Tang, Chem. Sci., 2015, 6, 5347–5365 RSC; (c) X. Wang, J. Hu, G. Zhang and S. Liu, J. Am. Chem. Soc., 2014, 136, 9890–9893 CrossRef CAS PubMed; (d) C. Li, T. Wu, C. Hong, G. Zhang and S. Liu, Angew. Chem., Int. Ed., 2012, 51, 455–459 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Synthetic procedures and characterization. See DOI: 10.1039/c6cc08452d |
| This journal is © The Royal Society of Chemistry 2017 |