Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Ca6La4(SiO4)2(PO4)4O2:Eu2+: a novel apatite green-emitting phosphor for near-ultraviolet excited w-LEDs
Yufei
Xia
a,
Yan-gai
Liu
*a,
Zhaohui
Huang
a,
Minghao
Fang
a,
Maxim S.
Molokeev
 bc and
LeFu
Mei
*a
bc and
LeFu
Mei
*a
aSchool of Materials Science and Technology, Beijing Key Laboratory of Materials Utilization of Nonmetallic Minerals and Solid Wastes, National Laboratory of Mineral Materials, China University of Geosciences, Beijing, 100083, China. E-mail: liuyang@cugb.edu.cn
bLaboratory of Crystal Physics, Kirensky Institute of Physics, SB RAS, Krasnoyarsk 660036, Russia
cDepartment of Physics, Far Eastern State Transport University, Khabarovsk 680021, Russia
First published on 11th April 2016
Abstract
A novel apatite phosphor Ca6La4(SiO4)2(PO4)4O2:Eu2+ was prepared by conventional high-temperature solid-state reaction. Phase purity was examined by XRD and XPS analysis. The crystal structure information, such as space group, cell parameters and atomic coordinates, were refined by the Rietveld method, revealing that Eu2+ occupied the sites of Ca2+ ions. Moreover, low-temperature experiments, including low-temperature PL spectra and low-temperature decay curve, were used to prove the existence of two luminescence centers in Ca6La4(SiO4)2(PO4)4O2:Eu2+. With the increase in doping concentration of Eu2+, the emission wavelength shows a red shift from 498 nm to 510 nm, which is mainly caused by the increase in crystal-field splitting by Eu2+. The optimized concentration of Eu2+ was confirmed to be 0.01, the Rc was calculated to be 20.09 Å and the energy transfer between Eu2+ was demonstrated to be by exchange interaction. Moreover, good thermal stability has been proved by a temperature-dependence experiment; it shows that the phosphor can maintain 55% of emitting intensity at 150 °C compared to that at room temperature. Finally, the Ca6La4(SiO4)2(PO4)4O2:Eu2+ phosphor was fabricated with commercial red (CaAlSiN3:Eu2+) and blue (BAM:Eu2+) phosphor coating on a n-UV chip. This proves that this green phosphor has the potential to be used in a w-LED lamp.
1. Introduction
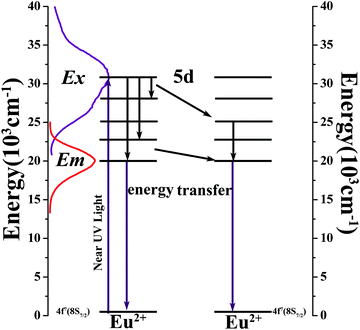
Nichia assembled a white light-emitting diode (w-LED) by means of a blue InGaN LED chip coated with yellow phosphor Y3Al5O12:Ce3+ (YAG:Ce3+) in 1996.1–3 At that time the w-LED was regarded as the next generation light source due to many advantages such as significant power reduction, higher luminous efficiency, longer lifetime and environmental friendliness.4 Accordingly, enormous research efforts have been focused on the preparation of high quality w-LEDs.5,6 Compared with Nichia's assembly method mentioned above, which shows high correlated color temperature (CCT ≈ 7750 K) and a low color rendering index (CRI ≈ 75),7 packaging multicolor phosphors on a near-ultraviolet (n-UV) chips can provide relatively high quality white light because the emission spectra can be distributed over the entire visible range (400–800 nm).8–10 This means that the study of high-performance single-color phosphor is meaningful.Researchers have paid great attention to green-emitting phosphors, for instance the cordierite structure phosphor K0.5Mg1.5Al4Si5O13:Eu2+,11 the garnet structure phosphors Ca2LaZr2Ga3O12:Ce3+,12 Ca2YZr2Al3O12:Ce3+,13 and the commercial green phosphor (Ba,Sr)SiO4:Eu2+. However, there is still room for improvement in some aspects such as wider emission region, better thermal stability and higher quantum efficiency. It is well known that the apatite compound has been utilized as a highly efficient host material for phosphor due to its adjustable crystal structure and its excellent thermal and physicochemical stability.14–16 Recently, our group has reported a series of apatite solid solution phosphors Ca2+xLa8−x(SiO4)6−x(PO4)xO2:Eu2+ (x = 0, 2, 4, and 6) whose emitting color can be adjusted from green to blue by changing the ratio of Ca to La. One of these compounds, Ca6La4(SiO4)2(PO4)4O2:Eu2+, show good potential as a commercial green phosphor. Therefore, we consider it worth investigating the crystal structure and luminescence properties of Ca6La4(SiO4)2(PO4)4O2:Eu2+ in more detail.
As discussed above, the novel apatite phosphors Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.05–0.1) have been synthesized by high-temperature solid state reaction for the first time. The phase purity was examined by XRD and XPS and the crystal structure information was analyzed by Rietveld refinement using XRD profiles. In addition, it is the first time that the low-temperature PL and PLE spectra and decay curves were utilized to demonstrate the inclusion of Eu2+ in the apatite phosphor. Moreover, the luminescence properties, such as PLE and PL spectra, lifetimes and thermal stability, are discussed in detail. Finally, the novel green phosphor Ca6La4(SiO4)2(PO4)4O2:Eu2+ was packaged on a u-UV LED chip (excitation wavelength 385 nm) with commercial red and blue phosphors, demonstrating the potential for it to be utilized in a w-LED.
2. Experiment
2.1 Sample preparation
The apatite phosphors Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.005, 0.01, 0.02, 0.04, 0.06, 0.08, and 0.1) were synthesized by a conventional high-temperature solid-state method. Based on the stoichiometric amounts of reactants, Ca2CO3 (analytical reagent, AR), La2O3 (AR), SiO2 (AR), NH4H2PO4 (AR) and Eu2O3 (AR) were thoroughly ground in an agate mortar for 15 min. The mixed powders were then preheated for 1 h at 1000 °C in a corundum crucible to eliminate H2O and CO2. Subsequently, the mixtures were sintered at 1500 °C in a reductive atmosphere (H2 10%, N2 90%) for 4.5 h. After cooling to room temperature, the sintered samples were ground once again to prepare for further measurements.2.2 Characterization
The X-ray diffraction (XRD) data of the series of phosphors were measured on an X-ray powder diffractometer (D/max-IIIA, Rigaku, Japan) with a stepwise scanning mode over the 2θ range 10°–110° using Cu-Kα radiation (1.5406 Å) with operating voltage 40 kV and current 100 mA. The XRD patterns, used in Rietveld refinement, were acquired with a step size of 0.02° and counting time of 2 s per step. X-ray photoelectron spectroscopy (XPS) measurements were collected with a Kratos Axis Ultra DLD, employing MCP stack & delay-line photoelectron detector with scanned & snapshot spectroscopy modes. The photoluminescence emission (PL) spectrum and the photoluminescence excitation (PLE) spectrum at 298 K and 8 K were measured using a Hitachi F-4600 fluorescence spectrophotometer (Japan) equipped with a 150 W Xe lamp as the excitation source. The temperature-dependent luminescence properties were measured on the same spectrophotometer, which was assembled with a computer-controlled electric furnace and a self-made heating attachment. The room-temperature luminescence decay curves were obtained from a spectrofluorometer (Horiba, Jobin Yvon TBXPS) using tunable pulsed laser irradiation (nano-LED) as the excitation. Quantum efficiency was measured by a Fluoromax-4 spectrofluorometer with an integral sphere at room temperature (Horiba, Jobin Yvon).3. Results and discussion
3.1 Phase formation and structural characteristics
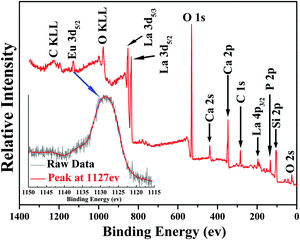
The XRD profiles of Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.005–0.1) phosphors are displayed in Fig. 1. As shown in Fig. 1, all the diffraction spectra do not contain any peaks from impurities or raw materials even at x = 0.1, indicating that pure apatite structure phosphors Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.005–0.1) have been synthesized successfully and that doping of Eu2+ does not cause any impurities or phase transitions.17Fig. 2 depicts the photoelectron survey spectrum of the Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ sample. It contains several photoelectron peaks corresponding to O 2s, Si 2p, P 2p, La 4p3/2, Ca 2p, Ca 2s, O 1s, La 3d5/2, La 3d3/2 and Eu 3d5/2 emissions. In addition, the high-resolution XPS spectrum at the Eu 3d5/2 position is shown in the inset of Fig. 2. The binding energy was measured to be 1127 eV, which matches well with the signal of Eu2+ 3d5/2, demonstrating the existence of Eu2+.18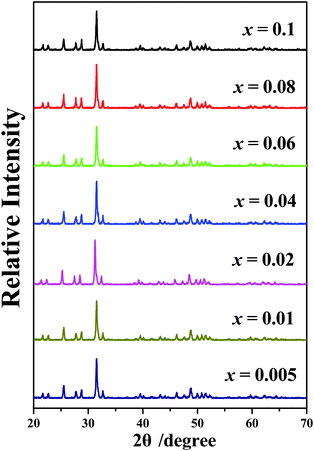 | ||
| Fig. 1 The XRD traces of Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.005–0.1) phosphors. Inset: the high resolution XPS spectrum at Eu 3d. | ||
The crystal structure information of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ was determined by the Rietveld refinement method. The observed (×), calculated (red) and difference (gray) XRD profiles for Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ are illustrated in Fig. 3 and the refinement parameters are presented in Table 1. The refinement results further prove that the novel apatite phosphor is a single phase without any impurity or secondary phase and that Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ crystallizes as a hexagonal structure with space group P63/m. The lattice parameters were determined to be a = b = 9.536 Å, c = 7.026 Å, V = 553.35 Å3 and the refinement R factors finally converged to Rwp = 8.17%, Rexp = 1.28%, Rp = 6.20%, indicating that the refined crystal structure agrees well with the starting model (Ca2La8(SiO4)6). In addition, all atoms positions and occupancy factors for Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ are listed in Table 1.
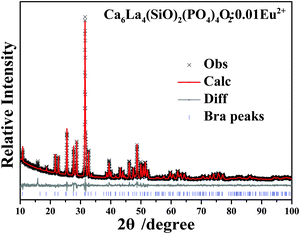 | ||
| Fig. 3 Powder XRD pattern (×) of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ samples with corresponding Rietveld refinement (red) and residual (gray) traces. | ||
| Formula | CLSPO:0.01Eu2+ |
|---|---|
| Space group | P63/m |
| a = b; c (Å) | 9.536(3); 7.026(3) |
| V (Å3) | 553.35(5) |
| R wp (%) | 8.17 |
| R exp (%) | 1.28 |
| R p (%) | 6.20 |
| Atom | Site | x | y | z | Occupancy |
|---|---|---|---|---|---|
| La1 | 6h | 0.227(2) | 0.983(3) | 0.25 | 0.617 |
| Ca1 | 6h | 0.227(2) | 0.983(3) | 0.25 | 0.368 |
| Eu1 | 6h | 0.227(2) | 0.983(3) | 0.25 | 0.005 |
| La2 | 4f | 2/3 | 1/3 | 0.0024(1) | 0.060 |
| Ca2 | 4f | 2/3 | 1/3 | 0.0024(1) | 0.935 |
| Eu2 | 4f | 2/3 | 1/3 | 0.0024(1) | 0.005 |
| Si | 6h | 0.406(8) | 0.375(1) | 1/4 | 1/3 |
| P | 6h | 0.406(8) | 0.375(1) | 1/4 | 2/3 |
| O1 | 6h | 0.598(2) | 0.464(2) | 1/4 | — |
| O2 | 6h | 0.339(2) | 0.493(2) | 1/4 | — |
| O3 | 12i | 0.344(1) | 0.261(1) | 0.072(1) | — |
| O4 | 2a | 0 | 0 | 1/4 | — |
Fig. 4 illustrates the reflectance spectra of the Ca6La4(SiO4)2(PO4)4O2 host and Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ in the range from 200 nm to 800 nm. Obviously, compared with the absorption characteristics of Ca6La4(SiO4)2(PO4)4O2 host, which barely absorbs n-UV light, Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ shows a broad absorption band from 200 nm to 450 nm, demonstrating the as-prepared phosphor can efficiently excited by a UV or n-UV LED chip.10,18 Furthermore, as shown in the inset of Fig. 4, the band gap of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ was estimated to be 3.39 eV.
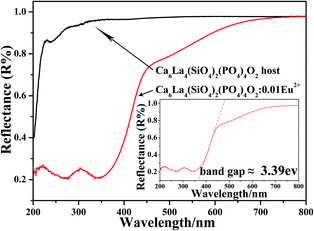 | ||
| Fig. 4 Reflectance spectra of Ca6La4(SiO4)2(PO4)4O2 host and Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+. Inset: the band gap of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+. | ||
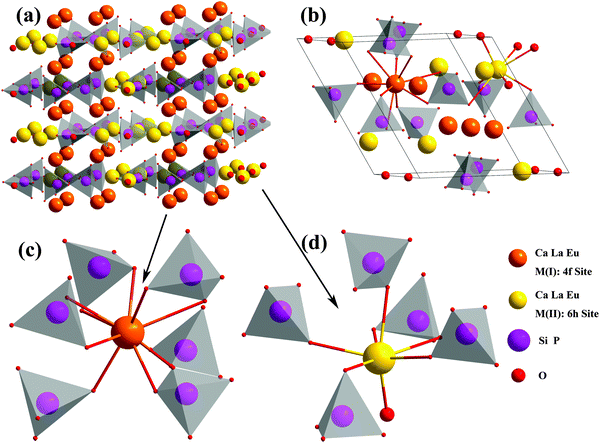
The crystal structure of 2 × 2 × 2 unit cells, the coordination environments of a single unit cell and the coordination environments of cation sites in Ca6La4(SiO4)2(PO4)4O2:Eu2+ are displayed in Fig. 5a–d. As observed from Fig. 5a, Ca6La4(SiO4)2(PO4)4O2 has a layered structure and two types of cation sites included in it, the inner-laminar site labeled M(I) with the local symmetry C3 and the inter-laminar site labeled M(II) with the local symmetry Cs. Furthermore, Fig. 5b–d depicts the coordination environments of these cation sites. The M(I) site (located at position 4f), surrounded by 9 oxygen atoms, forms a mono-caped square antiprism and connects with the tetrahedral PO4/SiO4 groups. On the other hand, the M(II) site (located at position 6h) surrounded by 7 oxygen atoms forms a pentagonal bipyramid and connects with each other through vertices. Furthermore, considering the effective ionic radii (Å) of Ca2+ (RCN=9 = 1.18, RCN=7 = 1.06), La3+ (RCN=9 = 1.21, RCN=7 = 1.10) and Eu2+ ions (RCN=9 = 1.3, RCN=7 = 1.2), Eu2+ is expected to substitute the Ca2+ site in the Ca6La4(SiO4)2(PO4)4O2:Eu2+ crystal structure based on its similar ion radius and valence.19,20
3.2 Photoluminescence characteristics
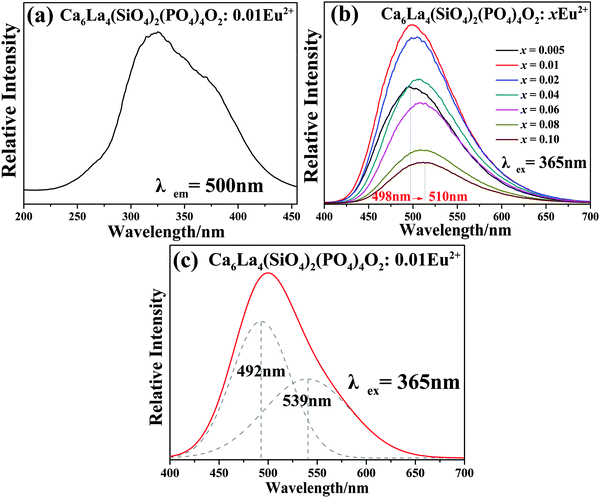
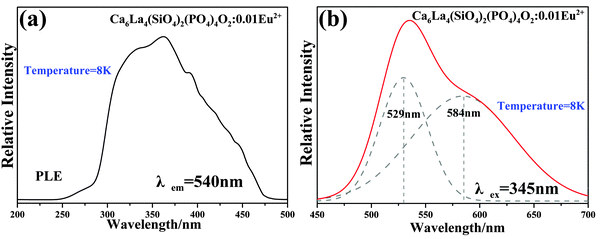
The PLE spectrum of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ (monitored at 500 nm) and the PL spectra of Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.005–0.1) (under 365 nm excitation) are displayed in Fig. 6a and b, respectively. Due to the 4f7 → 4f65d1 transition of Eu2+, the PLE spectrum depicts a broad excitation band ranging from 280 nm to 400 nm with a maximum value at 330 nm, indicating that the Ca6La4(SiO4)2(PO4)4O2:Eu2+ phosphor can be suitably excited by a n-UV LED chip.21 The PL spectrum consists of an asymmetric broad emission band peaking at 500 nm, which is ascribed to the electric dipole-allowed transition of Eu2+ from the lowest level of the 5d excited state to the 4f ground state.22 In general, the asymmetric emission band is due to the different coordination environments of Eu2+. As shown in Fig. 6c, the asymmetric emission band of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ has been resolved into two Gaussian components peaking at 492 nm (I1) and 593 nm (I2). Thus, the low-temperature (8 K) PLE and PL spectra of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ are displayed in Fig. 7a and b to demonstrate the existence of the different Eu2+ sites. Consequently, the PL spectrum at low temperature contains two component peaks located at 529 nm and 584 nm and the shape of PLE spectrum was found to be identical to that of measured at room temperature. These features further prove that Eu2+ occupies two types of Ca2+ sites to form different emitting centers.4 Moreover, due to the existence of multi-emitting centers, the Ca6La4(SiO4)2(PO4)4O2:Eu2+ phosphor shows a wider emission band compared with the BaMgSiO4:Eu2+ and Ca2YZr2Al3O12:Ce3+ green phosphors13,23 and it reaches a similar level as the commercial green phosphor (Ba,Sr)SiO4:Eu2+.In theory, the width of the crystal-field splitting of the Eu2+ 5d level is the main influence on the position of the Eu2+ emitting energy level,24i.e., the lower the lowest crystal-field position, the lower the emission energy.25 The major difference between the 4f and 6h site is that the 6h site has a free oxygen ion, which does not belong to any silicate group. Therefore, the binding strength is not saturated compared with the 4f site resulting in the average covalency of the 6h site being higher than that of the 4f site. Consequently, Eu2+ will have lower emission bands when located at the 6h site than when at the 4f site and the higher energy emission band (492 nm) can be assigned to Eu2+ (4f) while the lower energy emission band (539 nm) is derived from Eu2+ (6h).16
This inferred conclusion can be further demonstrated by an empirical relationship proposed by Van Uitert:15
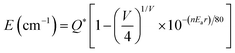 | (1) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1 for Eu2+); V is the activator ion valence (for Eu2+V = 2), n represents the number of anions in the immediate shell about the Eu2+ ion, r is the host cation radius that is replaced by the Eu2+ ion (in Å), and Ea is the electron affinity of the atoms that form anions (in eV). In general, due to the complexity of the local crystal structure, the exact energy levels of Eu2+ at a specific site are hard to calculate precisely.26 However, it can be deduced approximately that the value of E is proportional to the quantity of n and r.4 For the as-prepared phosphors, r is equal because both of the substitutions occurring at cation sites are that Ca2+ is replaced by Eu2+. Therefore, the band peak at 492 nm is ascribed to the 4f65d1 → 4f7 transition of Eu2+ occupying the M(I) site with nine-coordination, whereas the longer wavelength band can be attributed to Eu2+ occupying the M(II) site with seven-coordination.
000 cm−1 for Eu2+); V is the activator ion valence (for Eu2+V = 2), n represents the number of anions in the immediate shell about the Eu2+ ion, r is the host cation radius that is replaced by the Eu2+ ion (in Å), and Ea is the electron affinity of the atoms that form anions (in eV). In general, due to the complexity of the local crystal structure, the exact energy levels of Eu2+ at a specific site are hard to calculate precisely.26 However, it can be deduced approximately that the value of E is proportional to the quantity of n and r.4 For the as-prepared phosphors, r is equal because both of the substitutions occurring at cation sites are that Ca2+ is replaced by Eu2+. Therefore, the band peak at 492 nm is ascribed to the 4f65d1 → 4f7 transition of Eu2+ occupying the M(I) site with nine-coordination, whereas the longer wavelength band can be attributed to Eu2+ occupying the M(II) site with seven-coordination.
In addition, as shown in Fig. 6b, with the increase of doping concentration of Eu2+, the peak emission wavelength shows an obvious red-shift from 498 to 510 nm. The red-shift phenomenon is mainly due to the variation in the crystal-field splitting of Eu2+. In this regard, the crystal-field splitting of Eu2+ can be determined as obeying:1
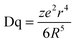 | (2) |
The concentration dependence of the PL intensity of the as-prepared samples Ca6La4(SiO4)2(PO4)4O2:xEu2+ (x = 0.005–0.1) are depicted in Fig. 8. With the increase of Eu2+ content, the PL intensity shows an increase at first and it reaches the maximum value at x = 0.01. Subsequently, the intensity tends to decrease gradually due to the concentration quenching effect. Thus, the optimized concentration of Eu2+ was confirmed to be 0.01.
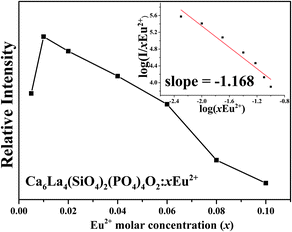 | ||
| Fig. 8 The concentration dependence of the PL spectral intensity. Inset: the curve of log(I/xEu2+) versus log(xEu2+). | ||
In general, concentration quenching can occur in a compound by obeying two mechanisms: exchange interaction and multipolar interaction.25,29,30 In consequence, as shown in Fig. 9, the concentration quenching originates from energy loss at a ‘killer’ center owing to the transfer of excitation energy among activators at a relative higher concentration.17,31 The mechanism of energy transfer between the Eu2+ can be expressed by the following equation:32
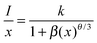 | (3) |
Therefore, as proposed by Blasse, the critical energy transfer distance (Rc) can be calculated as follows:4,13
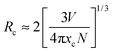 | (4) |
Room temperature decay curves of the as-prepared phosphors, under excitation at 365 nm, were measured and are plotted in Fig. 10. Each decay curve can be well fitted with a second-order exponential function by the following equation:4
I(t) = A1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ1) + A2 exp(−t/τ1) + A2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ2) exp(−t/τ2) | (5) |
| τ* = (A1τ12 + A2τ22)/(A1τ1 + A2τ2) | (6) |
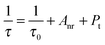 | (7) |
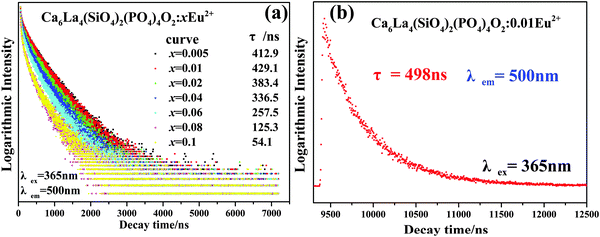 | ||
| Fig. 10 (a) Room-temperature and (b) low-temperature decay curves of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+. | ||
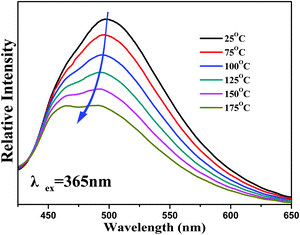
In general, temperature is the factor that has a great influence on the emitting intensity of a phosphor.12 Therefore, thermal stability is one of the important characteristics that needs to be taken into consideration before recommending the phosphor for potential applications.15 The PL spectra of the temperature dependence experiment are shown in Fig. 11. As we can observe from Fig. 11, the luminescence intensity shows a gradual decrease when the sample was heated and the phosphor still maintains 55% of the emitting intensity at 150 °C compared with the initial intensity at room temperature (25 °C). Therefore, it shows better thermal stability than the garnet phosphor Ca2LaZr2Ga3O12:Ce3+ and the commercial green phosphor Ba2SiO4:Eu2+. On the other hand, the full width at half maximum (FWHM) of the PL spectra increases from 56 nm up to 75 nm during this temperature increase. The increase of FWHM can be explained by a configuration coordinate diagram, a physical model, that electron–phonon interaction will enhanced and the population of higher vibration levels will increase when the temperature rises,18 and then, the excited luminescent center is thermally activated and release non-radiative through the crossover between the excited states and the ground states with the help of phonons. In consequence, the luminescence is quenched due to the enhanced population density of phonons, which also causes the broadening of the PL spectra.4,17
On the other hand, the PL spectra at different temperatures show a slight blue-shift, which is attributed to thermally-active phonon-assisted tunneling from the excited states of the low-energy emission band to the excited states of the high-energy emission band.11,12 As mentioned before, there are two emission centers for the Ca6La4(SiO4)2(PO4)4O2:Eu2+ phosphor. The thermal back transfer from lower energy centers to higher energy centers will be promoted at higher temperature.23,35 Consequently, the higher energy emission is strengthened and the blue-shift phenomenon is observed. In addition, the expanded host lattice can also cause a reduction in the crystal-field splitting and lead to a higher energy emission when the temperature increases.4,36
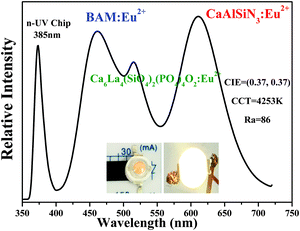
Luminescence efficiency is an important technological parameter for the application of a phosphor. The internal quantum efficiency (QE) of Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ was measured and calculated to be 57.73%. It is higher than that of K0.5Mg1.5Al4Si5O13:Eu2+ (48.3%)11 and Ca2LaZr2Ga3O12:Ce3+ (35.2%), but lower than the commercial green phosphor (Ba,Sr)SiO4 (79%).12 In general, the QE can be further optimized by improving the preparation conditions because the QE depends closely on crystalline defects, particle sizes and the morphology of the phosphor.8,37 To demonstrate the potential application, the electroluminescent spectrum of w-LED lamp is displayed in Fig. 12. The self-made lamp was fabricated by coating Ca6La4(SiO4)2(PO4)4O2:Eu2+, BAM:Eu2+ and CaAlSiN3:Eu2+ phosphors on a n-UV chip (λex = 385 nm). The CIE color coordinates, CCT and Ra of the self-fabricated w-LED lamp were calculated to be (0.37, 0.37), 4253 K and 86, respectively. The relatively high Ra value (86) and appropriate CCT value (4253 K) demonstrate that the Ca6La4(SiO4)2(PO4)4O2:Eu2+ can be a promising candidate for a green-emitting phosphor for the application of w-LEDs.
4. Conclusion
A novel apatite structure green phosphor Ca6La4(SiO4)2(PO4)4O2:Eu2+ has been synthesized by a high-temperature solid-state reaction method. The phase purity and elemental valency were examined according XRD and XPS. The crystal structure information was refined by Rietveld refinement using the high quality XRD traces. The refinement parameters, all atom positions and occupancy factors for Ca6La4(SiO4)2(PO4)4O2:0.01Eu2+ were presented and the coordination environments of the two types of cation sites were discussed in detail. In the section on luminescence properties analysis, the asymmetric emission spectrum, which is attributed to the electric dipole-allowed transition of the Eu2+ ions from excited 5d state to the 4f ground state, was resolved into two Gaussian components peaking at 492 nm and 593 nm, demonstrating the existence of two emitting centers. Moreover, this result was further proved by low-temperature luminescence experiments. Based on the coordination environments analysis and an empirical relation proposed by Van Uitert, we propose that the higher energy emission band (492 nm) will be detected when Eu2+ is located at the 4h site and the lower energy emission band (539 nm) is derived from the Eu2+ ion occupying the 6h site. The optimized Eu2+ concentration was confirmed to be x = 0.01, the Rc is calculated to be 20.09 Å and the energy transfer between Eu2+ is of the exchange interaction type. The phosphor shows good thermal stability, it maintain 55% of its emitting intensity at 150 °C compared with room temperature. Finally, the Ca6La4(SiO4)2(PO4)4O2:Eu2+ phosphor was fabricated with commercial red (CaAlSiN3:Eu2+) and blue (BAM:Eu2+) phosphors on a n-UV chip excited at 385 nm; the CIE color coordinates, CCT and Ra of the self-fabricated w-LED lamp were calculated to be (0.37, 0.37), 4253 K and 86, respectively. This proves that this green phosphor has the potential to be used in a w-LED lamp.Acknowledgements
This study was sponsored by the National Natural Science Foundation of China (Grant No. 51472223), the Program for New Century Excellent Talents in University of Ministry of Education of China (Grant No. NCET-12-0951) and the Fundamental Research Funds for the Central Universities (Grant No. 2652015020 and Grant No. 2652015008).References
- C. H. Huang, Y. C. Chiu, Y. T. Yeh, T. S. Chan and T. M. Chen, ACS Appl. Mater. Interfaces, 2012, 4, 6661–6668 CAS.
- S. Ye, F. Xiao, Y. X. Pan, Y. Y. Ma and Q. Y. Zhang, Mater. Sci. Eng., R, 2010, 71, 1–34 CrossRef.
- W. Wang, W. Yang, Z. Liu, H. Wang, L. Wen and G. Li, Sci. Rep., 2015, 5, 11480 CrossRef PubMed.
- W. Xiao, X. Zhang, Z. Hao, G.-H. Pan, Y. Luo, L. Zhang and J. Zhang, Inorg. Chem., 2015, 54, 3189–3195 CrossRef CAS PubMed.
- W. Wang, J. Mater. Chem. C, 2014, 2, 9342–9358 RSC.
- G. Li, W. Wang, W. Yang and H. Wang, Surf. Sci. Rep., 2015, 70, 380–423 CrossRef CAS.
- M. Shang, C. Li and J. Lin, Chem. Soc. Rev., 2014, 43, 1372–1386 RSC.
- J. Chen, Y. Liu, M. Fang and Z. Huang, Inorg. Chem., 2014, 53, 11396–11403 CrossRef CAS PubMed.
- W. Wang, H. Yang and G. Li, J. Mater. Chem. C, 2013, 1, 4070–4077 RSC.
- W. Wang, Y. Lin, W. Yang, Z. Liu, S. Zhiou, H. Qian, F. Gao, L. Wen and G. Li, J. Mater. Chem. C, 2014, 2, 4112–4116 RSC.
- J. Zhou, Z. Xia, M. Chen, M. S. Molokeev and Q. Liu, Sci. Rep., 2015, 5, 12149 CrossRef PubMed.
- J. Zhong, W. Zhuang, X. Xing, R. Liu, Y. Li, Y. Liu and Y. Hu, J. Mater. Chem. C, 2015, 119, 5562–5569 CAS.
- X. Wang and Y. Wang, J. Mater. Chem. C, 2015, 119, 16208–16214 CAS.
- H. Liu, Y. Luo, Z. Mao, L. Liao and Z. Xia, J. Mater. Chem. C, 2014, 2, 1619 RSC.
- Z. Xia, M. S. Molokeev, W. B. Im, S. Unithrattil and Q. Liu, J. Mater. Chem. C, 2015, 119, 9488–9495 CAS.
- G. Li, D. Geng, M. Shang, Y. Zhang, C. Peng, Z. Cheng and J. Lin, J. Mater. Chem. C, 2011, 115, 21882–21892 CAS.
- J. Chen, Y.-g. Liu, L. Mei, Z. Wang, M. Fang and Z. Huang, J. Mater. Chem. C, 2015, 3, 5516–5523 RSC.
- J. Chen, Y. Liu, L. Mei, H. Liu, M. Fang and Z. Huang, Sci. Rep., 2015, 5, 9673 CrossRef CAS PubMed.
- S. Schmiechen, P. Strobel, C. Hecht, T. Reith, M. Siegert, P. J. Schmidt, P. Huppertz, D. Wiechert and W. Schnick, Chem. Mater., 2015, 27, 1780–1785 CrossRef CAS.
- H. Ji, Z. Huang, Z. Xia, M. S. Molokeev, X. Jiang, Z. Lin and V. V. Atuchin, Dalton Trans., 2015, 44, 7679–7686 RSC.
- N. Guo, H. You, C. Jia, R. Ouyang and D. Wu, Dalton Trans., 2014, 43, 12373–12379 RSC.
- S. Miao, Z. Xia, J. Zhang and Q. Liu, Inorg. Chem., 2014, 53, 10386–10393 CrossRef CAS PubMed.
- W. B. Dai, Y. F. Lei, T. Yu, M. Y. Peng and Q. Y. Zhang, Mater. Res. Bull., 2015, 67, 176–184 CrossRef CAS.
- W. Lv, M. Jiao, Q. Zhao, B. Shao, W. Lu and H. You, Inorg. Chem., 2014, 53, 11007–11014 CrossRef CAS PubMed.
- M. Peng, X. Yin, P. A. Tanner, M. G. Brik and P. Li, Chem. Mater., 2015, 27, 2938–2945 CrossRef.
- Y. Zhang, G. Li, D. Geng, M. Shang, C. Peng and J. Lin, Inorg. Chem., 2012, 51, 11655–11664 CrossRef CAS PubMed.
- K. Fengwen, Z. Yi and P. Mingying, Inorg. Chem., 2015, 54, 1462–1473 CrossRef PubMed.
- H. Lin, J. Xu, Q. Huang, B. Wang, H. Chen, Z. Lin and Y. Wang, ACS Appl. Mater. Interfaces, 2015, 7, 21835–21843 CAS.
- K. A. Denault, J. Brgoch, M. W. Gaultois, A. Mikhailovsky, R. Petry, H. Winkler, S. P. DenBaars and R. Seshadri, Chem. Mater., 2014, 26, 2275–2282 CrossRef CAS.
- C.-H. Huang, P.-J. Wu, J.-F. Lee and T.-M. Chen, J. Mater. Chem., 2011, 21, 10489 RSC.
- R. Yu, J. Wang, Z. Zhao, M. Li, S. Huo, J. Li and J. Wang, Mater. Lett., 2015, 160, 294–297 CrossRef CAS.
- Z. Wang, Z. Xia, M. S. Molokeev, V. V. Atuchin and Q. Liu, Dalton Trans., 2014, 43, 16800–16804 RSC.
- R. Yu, N. Xue, J. Li, J. Wang, N. Xie, H. M. Noh and J. H. Jeong, Mater. Lett., 2015, 160, 5–8 CrossRef CAS.
- J. Chen, Y.-g. Liu, H. Liu, D. Yang, H. Ding, M. Fang and Z. Huang, RSC Adv., 2014, 4, 18234 RSC.
- X. Zhang, J. Wang, L. Huang, F. Pan, Y. Chen, B. Lei, M. Peng and M. Wu, ACS Appl. Mater. Interfaces, 2015, 7, 10044–10054 CAS.
- C. Zhao, Z. Xia and S. Yu, J. Mater. Chem. C, 2014, 2, 6032 RSC.
- S. Miao, Z. Xia, M. S. Molokeev, M. Chen, J. Zhang and Q. Liu, J. Mater. Chem. C, 2015, 3, 4616–4622 RSC.
| This journal is © The Royal Society of Chemistry 2016 |