Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Functionalized cationic [4]helicenes with unique tuning of absorption, fluorescence and chiroptical properties up to the far-red range†
I. Hernández
Delgado
a,
S.
Pascal
a,
A.
Wallabregue
a,
R.
Duwald
a,
C.
Besnard
b,
L.
Guénée
b,
C.
Nançoz
c,
E.
Vauthey
c,
R. C.
Tovar
d,
J. L.
Lunkley
d,
G.
Muller
d and
J.
Lacour
 *a
*a
aDepartment of Organic Chemistry, University of Geneva, Quai Ernest Ansermet 30, CH-1211 Geneva 4, Switzerland
bLaboratory of Crystallography, University of Geneva, Quai Ernest Ansermet 24, CH-1211 Geneva 4, Switzerland
cDepartment of Physical Chemistry, University of Geneva, Quai Ernest Ansermet 24, CH-1211 Geneva 4, Switzerland
dDepartment of Chemistry, San José State University, 1 Washington Square, San José, CA 95192-0101, USA
First published on 8th April 2016
Abstract
Unprecedented regioselective post-functionalization of racemic and enantiopure cationic diaza [4]helicenes is afforded. The peripheral auxochrome substituents allow a general tuning of the electrochemical, photophysical and chiroptical properties of the helical dyes (26 examples). For instance, electronic absorption and circular dichroism are modulated from the orange to near-infrared spectral range (575–750 nm), fluorescence quantum efficiency is enhanced up to 0.55 (631 nm) and circularly polarized luminescence is recorded in the red (|glum| ∼ 10−3).
Introduction
Organic helicenes, which are defined as helical derivatives made of ortho-fused aromatic rings,1 commonly feature (chir)optical properties, i.e. absorption, fluorescence, electronic circular dichroism (ECD) and circularly polarized luminescence (CPL) in the blue range of the visible spectrum.2 Such photophysical characteristics have triggered applications in (polarized) blue light emitting diodes for instance3 and to a lesser extent in the field of bio-imaging.4 Accessing the red spectral region (620–750 nm) is essential for applications in microscopy and chemical biology in particular, and this remains a challenge for organic helicenes.5 In fact, only a few cationic aza derivatives, e.g.1–4, exhibit optical properties in this range; these moieties therefore offer limited access to specific low energy absorptions and emissions (Fig. 1, top).6,7 Among these species, cationic helicenes of type 1, sometimes named DMQA (DiMethoxyQuinAcridinium), are of particular interest due to their remarkable chemical (pKR+ ≈ 19)8 and configurational (ΔG‡ of racemization ∼ 42 kcal mol−1) stabilities.9 These derivatives are prepared on the gram-scale in no more than two steps. Starting from a tris(2,6-dimethoxyphenyl)methylium cation and using primary amines as nucleophiles, the protocol involves two consecutive aza ring closures using nucleophilic substitutions of four ortho-OMe groups.9b,d,10 Unsymmetrical derivatives with two different nitrogen substituents can also be afforded using a stepwise process.11 Furthermore a highly reliable resolution procedure is available using (i) addition of (+)-(R)-methyl-p-tolylsulfoxide to the central carbon, (ii) a facile chromatographic separation of the diastereomeric adducts (ΔRf ∼ 0.3 on TLC (SiO2)) and (iii) a final Pummerer fragmentation.12 Applications in the fields of supramolecular chemistry, selective DNA binding or material science have been developed.13 Of importance for the current study, nitrogen substituents (H, alkyl, aryl) have only a negligible impact on the optical properties of cation 1,14 which are remarkably conserved with absorption and emission maxima centered at 616 and 667 nm, respectively. To modulate the photophysics, it was deemed necessary to introduce substituents at positions other than the nitrogen atoms. In fact, in the related triangulenium series, oxa or aza functional groups positioned para to the central cationic charge provoke efficient optical shifts (Δλabs up to 50 nm).9c,15 Still, the introduction of such groups requires the preparation of specific cationic triarylmethylium precursors for each substitution pattern. Herein, the capacity to broadly tune the electrochemical, absorption, fluorescence and chiroptical properties of purely organic cationic helicenes is reported for the first time. Thanks to an unprecedented post-functionalization strategy, a large variety of substituents were introduced regioselectively at the periphery of racemic and enantiopure diaza [4]helicenes 1, in no more than four synthetic steps (Fig. 1, bottom). The functional groups induce major changes in the (photo)physical properties of the chromophores as demonstrated using electrochemical, absorption, and fluorescence studies. This allows a large tuning of the absorption and circular dichroism properties from the orange to near-infrared spectral range (575–750 nm) and an enhancement of fluorescence can be achieved with quantum yields up to 0.55 and cut-off emissions up to the far-red range. CPL activities (|glum| ∼ 10−3) in the red domain were also monitored for some of these dyes.Results and discussion
As already mentioned, species of types 1–4 offer only limited access to specific low energy absorptions and emissions. Care was thus taken to study the viability of a post-functionalization strategy which would give access to a large variety of substituted helicenes using a single (racemic or enantiopure) [4]helicene precursor of type 1.Synthesis
Interestingly and somewhat surprisingly, initial halogenation attempts using classical N-chlorosuccinimide or N-bromosuccinimide reagents (1 equiv.)6b demonstrated a strong nucleophilic reactivity for compound 1 (Fig. 1, R = R′ = nPr). In fact, reactions with the electrophilic agents yielded not only monohalogenated derivatives but also several polyhalogenated adducts.16 Complex mixtures were obtained which could not be purified. Attempts to tame the reactivity after the first halogenation were unsuccessful. To prevent polysubstitution, it became clear that the first introduced substituent should deactivate the nucleophilic character of the central aromatic core. The introduction of strong electron-withdrawing groups such NO2 or CHO was thus envisaged. Accordingly, conditions were found for the selective mononitration of 1 using mild biphasic conditions (HNO3/CH2Cl2, 25 °C, 15 min). Compound 5 was isolated in an excellent yield after a simple filtration (99%, Scheme 1). Satisfactorily, an exclusive regioselectivity was observed with only one of three formally activated positions being nitrated (atom C6 or C8, vide infra). For the formylation, 1 was engaged in a Vilsmeier–Haack reaction using a large excess of phosphorus oxychloride in DMF at 90 °C. Compound 6 was isolated in a good yield (86%), again as a single regioisomer. With 5 and 6 in hand, it was then possible to generate the other functionalized derivatives. Hydrogenation of 5 under heterogeneous catalysis (Pd/C, H2 1 atm) led to the formation of amino derivative 7 (1 h, 99%); the formation was characterized by a visible color change from red to light green. Using modified Eschweiler–Clarke conditions (HCHO, NaBH3CN, acetic acid), 7 was efficiently converted into tertiary bismethylated amine 8 (99%). Primary amino 7 was transformed into azido 9 using the reaction with tert-butyl nitrite and azidotrimethylsilane (99%).17 With this compound in hand, Cu(I)-catalyzed azide–alkyne cycloadditions were performed to yield triazolo derivatives 10a, 10b and 10c (Ar = Ph, p-CF3Ph, p-NMe2Ph, 87–99%).Aldehyde 6 was also easily derivatized. Cyano 11 was obtained through a Schmidt reaction in 70% yield.18,19 Olefination reactions were achieved either via a Knoevenagel condensation with malononitrile (12, 94%) or by Wittig reactions. For these latter transformations, a series of phosphonium salts was prepared with benzylic halides carrying electron-withdrawing and donating substituents. After formation of the ylides and condensation with 6, the resulting alkenes 13a, 13b, 13c, 13e and 13f were isolated with a perfect E selectivity in moderate to excellent yields (55–98%). Reduction of 13b was achieved with Zn in the presence of AcOH to form 13d (98%).20 Furthermore, it was possible to convert the formyl group of 6 into an hydroxyl OH group via a modified Dakin protocol using 3-chloroperbenzoic acid (mCPBA) as the oxidant (14, 78%).21 Compound 14 was alkylated (MeI, K2CO3) to afford 15 in 95% yield.
Finally, a series of carboxyl and carbonyl adducts was generated. Zwitterionic carboxylate 16 was obtained under mild Pinnick–Kraus conditions (76%).22,23 The transformation of green 16 into a corresponding deep purple acyl chloride intermediate (with SOCl2) led to ketones (17a and 17b, 67–75%), esters (17c and 17d, 88–98%), thioester 17e (76%) and amides (17f and 17g, 94–98%) by reactions with Grignard reagents, alcohols, a thiol and amines, respectively. These newly-substituted [4]helicenes, with the exception of 16, were prepared as tetrafluoroborate salts in racemic form and characterized using 1H, 13C, 19F NMR, IR and high-resolution mass spectrometry (see ESI†). Specific compounds were prepared as single enantiomers and details will be given in the following paragraphs. When possible, further structural characterization was achieved using X-ray diffraction analysis.
Solid state structural analysis
Single crystals suitable for diffraction analysis were obtained for racemic 1, 5, 6, 7, 14, 15, 17c, 17e, 17f and 17g by the careful addition of a layer of toluene above solutions of the dyes in dichloromethane (methanol for 14) and a slow mixing of the two phases over one or two weeks (open vial usually). To crystallize 5 and 14, anion exchange metatheses of the BF4− anion to NO3− and Cl− were necessary, respectively. The X-ray structure of 6 is presented in Fig. 2 and the relevant deduced values are reported in Table S1.† As expected for these racemates, all the space groups contain symmetry elements of the second kind. Molecules form stacks along the small unit-cell axes with typical distances between adjacent molecules of the order of 3.8–4.1 Å. Aromatic parts of the adjacent molecules in the stack are most of the time shifted to avoid strong π–π interactions.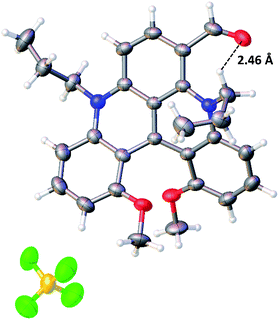 | ||
| Fig. 2 Anisotropic displacement ellipsoids plot at 50 percent probability level of the crystal structure of 6 (only the P enantiomer is shown). | ||
For precursor 1, a helical angle of 41.1° formed by the planes of the terminal methoxyphenyl rings and a helical pitch of 3.19 Å between C(1) and C(13) were measured and confirmed from the previous observations.9b The helicenes with introduced nitro (5) or carbonyl (6, 17c, 17e, 17f and 17g) functional groups possess higher helical angles and pitch (up to 45.9° and 3.26 Å for 6). In the latter case, a C–H⋯O bond between a hydrogen atom from the propyl side chain (NCH2 fragment) and the oxygen of the carbonyl function can be noticed (Fig. 2). This interaction, induced by a probably strong δ+ character of the hydrogen atoms adjacent to the nitrogen, leads to an inwards (syn) conformation of the aldehyde moiety that points towards the exocyclic methylene group.24 This strained geometry might be the reason for the distortion of the helical skeleton. Interestingly, this conformation was evidenced in solution using a 1H NOESY NMR experiment (CD2Cl2, 298 K), which shows a through-space correlation between the H of the formyl group and H(7) (see the ESI, Fig. S3†).
The bond lengths of the carbocations were also extracted from the X-ray data and analyzed (Fig. S1†). An alternation of single and double bonds within the three internal heterocycles could not be evidenced; only a slight single/double bond character of the terminal methoxybenzene rings was noticed. This suggests a strong resonance within the benzonaphthyridinylium scaffold in which the cationic charge is presumably fully delocalized.25 From this section, one can conclude that the substituents introduced at the C(6) position may interact with the pendant alkyl chains and influence the helical core.
Electronic properties
Cyclic voltammetry (CV) experiments were performed using anhydrous acetonitrile solutions with tetrabutylammonium hexafluorophosphate as the supporting electrolyte. The voltammograms of representative dyes 1, 5, 8, 15 and 17c are presented in Fig. 3. Unsubstituted precursor 1 was used as a reference. As previously reported,9d this compound displays a reversible reduction at −1.23 V versus Fc/Fc+ and a pseudo-reversible oxidation at 0.88 V. In comparison, the CV of nitro-substituted dye 5 reveals a reversible reduction at −0.97 V and a second reduction corresponding to the formation of the carbanion at ∼−1.78 V.26 Only an irreversible oxidation is recorded at high potential (1.30 V) and the increase of scan rate to 2 V s−1 did not allow a reversibility, suggesting a faster chemical step occurring after the oxidation. Ester 17c also displayed a reversible reduction and a pseudo-reversible oxidation, which is very similar to that of 1 except with both reduction and oxidation occurring at higher potentials (−1.09 V and 1.08 V respectively). Such an influence from electron-withdrawing NO2 and CO2Et groups was expected. Electron-rich 15 and 8 were reduced at comparable potentials to 1 but their first oxidation was, on the contrary, considerably facilitated (peaks at 0.64 V and 0.34 V, respectively). Moreover, 8 presented a reversible second oxidation at 0.67 V, marking the strong electronic donating effect of the dimethylamino group. An extension of the analysis to other derivatives was performed. Details can be found in Table 1 and in the ESI (Table S2, Fig. S3†).27 In other instances, complex voltammograms were obtained due to the inherent electroactivity of the substituents and these data are not presented. In a general manner, the introduced moieties have a stronger influence on the first oxidation potentials (Table 1, from 5 to 7: ΔΔE(Ox1) = 0.96 V) than on the first reduction potentials (ΔΔE(Red1) = 0.32 V); this modulation of the electrochemical properties bodes well for tunable optical properties.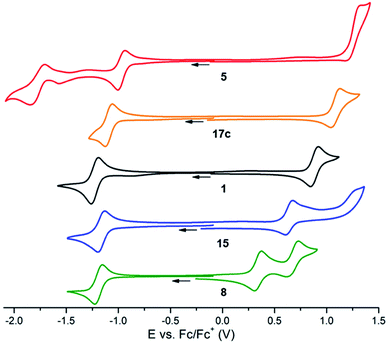 | ||
| Fig. 3 Voltammetric curves of acetonitrile ([TBA][PF6] 10−1 M) solutions of 5 (red), 17c (orange), 1 (black), 15 (blue) and 8 (green) (10−3 M) recorded at a Pt working electrode (ν = 0.1 V s−1). | ||
| Molecule | Functional group (Y) | Reductiona | Oxidationa | ||||
|---|---|---|---|---|---|---|---|
| Red1 | ΔE(Red1)b | Red2 | Ox1 | ΔE(Ox1)b | Ox2 | ||
| a Half-wave potentials values, otherwise noted. b Potential difference compared to the first reduction or oxidation waves of compound 1. c Irreversible process. | |||||||
| 5 | NO2 | −970 | −256 | −1778 | 1304c | 420 | — |
| 6 | CHO | −1059 | −167 | −1269 | 1131 | 247 | — |
| 17c | CO2Et | −1091 | −135 | — | 1084 | 200 | — |
| 17f | CONHPr | −1120 | −106 | −1877 | 1000 | 116 | — |
| 1 | H | −1226 | 0 | — | 884 | 0 | — |
| 9 | N3 | −1094 | −132 | −1834c | 769 | −115 | 1275c |
| 16 | CO2− | −1285 | 59 | — | 711c | −173 | 1006 |
| 15 | OMe | −1164 | −62 | — | 640 | −244 | 1258c |
| 7 | NH2 | −1201 | −25 | — | 354 | −530 | 781c |
| 8 | NMe2 | −1191 | −35 | — | 342 | −542 | 673 |
Electronic absorption
Absorption spectra were recorded in acetonitrile and the results are presented in Fig. 4, data are compiled in Table 2. Unsubstituted precursor 1 was again used as a reference to evaluate the influence of the different functional groups. Dye 1 absorption is characterized using a relatively sharp and moderately intense maximum at 616 nm (full width at half maximum, fwhm = 2460 cm−1, ε = 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1), with a shoulder at higher energy. Upon introduction of electron-withdrawing functions, the lower energy transition undergoes a moderate hypsochromic shift. For instance the strong electron-withdrawing nitro function in 5 leads to a 40 nm blue-shift of the absorption maximum and a slight weakening of the molar extinction coefficient. Weaker aldehyde (6), cyano (11), ketone (17a,b) and ester (17c,e) substituents provoke a less pronounced hypsochromic shift of the absorption with maxima located in the yellow-orange window (∼580–600 nm). Compounds 10a–c and 17f,g display negligible shifts of absorption due to the poor electronic effect brought about by the triazolo and amido functions.
000 M−1 cm−1), with a shoulder at higher energy. Upon introduction of electron-withdrawing functions, the lower energy transition undergoes a moderate hypsochromic shift. For instance the strong electron-withdrawing nitro function in 5 leads to a 40 nm blue-shift of the absorption maximum and a slight weakening of the molar extinction coefficient. Weaker aldehyde (6), cyano (11), ketone (17a,b) and ester (17c,e) substituents provoke a less pronounced hypsochromic shift of the absorption with maxima located in the yellow-orange window (∼580–600 nm). Compounds 10a–c and 17f,g display negligible shifts of absorption due to the poor electronic effect brought about by the triazolo and amido functions.
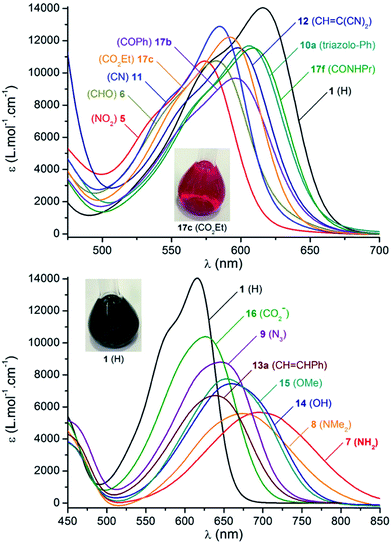 | ||
| Fig. 4 Selected absorption spectra in acetonitrile (10−5 M) at 293 K as a function of substituent Y. Top and bottom diagrams depict spectra presenting hypsochromic and bathochromic shifts compared to ref. 1 respectively. Insets: typical coloration of acetonitrile solutions (10−4 M). | ||
| Molecule | Functional group (Y) | λ max/nm (εmax/L mol−1 cm−1) | fwhm/cm−1 | λ em/nm | Φ | τ /ns | k R /106 s−1 | k NR /106 s−1 |
|---|---|---|---|---|---|---|---|---|
| a Reference: cresyl violet (Φ = 0.54 in methanol), estimated error ± 10%. b Excitation at 470 nm. c With kR = Φ/τ and kNR = (1 − Φ)/τ. | ||||||||
| 1 | H | 616 (14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) 000) |
2460 | 667 | 0.13 | 5.5 | 23.6 | 158.2 |
| 5 | NO2 | 575 (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) 000) |
2440 | 624 | 0.35 | 14.5 | 24.1 | 44.8 |
| 6 | CHO | 582 (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 750) 750) |
2500 | 640 | 0.37 | 16.2 | 22.8 | 38.9 |
| 7 | NH2 | 698 (6560) | 3280 | — | — | — | — | — |
| 8 | NMe2 | 675 (5580) | 3280 | — | — | — | — | — |
| 9 | N3 | 644 (8790) | 2890 | — | — | — | — | — |
| 10a | Triazolo-Ph | 606 (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 670) 670) |
2430 | 670 | 0.19 | 8.5 | 22.4 | 95.3 |
| 10b | Triazolo-p-CF3-Ph | 605 (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 820) 820) |
2370 | 665 | 0.24 | 10.4 | 23.1 | 73.1 |
| 10c | Triazolo-p-NMe2-Ph | 607 (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 520) 520) |
2420 | — | — | — | — | — |
| 11 | CN | 585 (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 880) 880) |
2290 | 631 | 0.55 | 19.1 | 28.8 | 23.6 |
| 12 | CHC(CN)2 | 598 (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 550) 550) |
2540 | 658 | 0.28 | 15.1 | 18.5 | 47.7 |
| 13a | CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-Ph CH-Ph |
638 (6710) | 2910 | — | — | — | — | — |
| 13b | CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-p-NO2-Ph CH-p-NO2-Ph |
638 (8090) | 2810 | — | — | — | — | — |
| 13c | CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-p-CF3-Ph CH-p-CF3-Ph |
640 (8810) | 2840 | — | — | — | — | — |
| 13d | CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-p-NH2-Ph CH-p-NH2-Ph |
660 (5600) | 3170 | — | — | — | — | — |
| 13e | CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-m-diCF3-Ph CH-m-diCF3-Ph |
636 (8750) | 2770 | — | — | — | — | — |
| 13f | CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-m-diOMe-Ph CH-m-diOMe-Ph |
644 (7960) | 2860 | — | — | — | — | — |
| 14 | OH | 659 (7450) | 3040 | — | — | — | — | — |
| 15 | OMe | 653 (7750) | 3030 | — | — | — | — | — |
| 16 | CO2− | 626 (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400) 400) |
2840 | 709 | 0.01 | — | — | — |
| 17a | COMe | 593 (9080) | 2430 | 657 | 0.26 | 12.0 | 21.7 | 61.7 |
| 17b | COPh | 598 (9130) | 2450 | 659 | 0.27 | 12.1 | 22.3 | 60.3 |
| 17c | CO2Et | 592 (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200) 200) |
2370 | 653 | 0.33 | 14.0 | 23.6 | 47.9 |
| 17d | CO2Ph | 586 (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700) 700) |
2380 | 641 | 0.39 | 16.3 | 23.9 | 37.4 |
| 17e | COSEt | 596 (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100) 100) |
2370 | 655 | 0.34 | 14.3 | 23.8 | 46.2 |
| 17f | CONHPr | 609 (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500) 500) |
2420 | 667 | 0.19 | 8.3 | 22.9 | 97.6 |
| 17g | CONEt2 | 615 (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300) 300) |
2450 | 678 | 0.13 | 6.2 | 21.0 | 140.3 |
In the series of helicenes carrying electron-withdrawing groups (Fig. 4, top), the shape of the lower energy transition remains remarkably comparable to 1, with fwhm between 2370 and 2540 cm−1. Furthermore, the absorption spectra of chromophores 1 and 5, were recorded in a wide range of solvents (Fig. S4†). 1 displays similar absorption bands in solvents ranging from THF to DMSO (Δ![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif) = 230 cm−1), indicating a small electronic rearrangement between the ground and excited states.28,29 This absence of solvatochromism was also observed for the lower energy transition of 5 (Δ
= 230 cm−1), indicating a small electronic rearrangement between the ground and excited states.28,29 This absence of solvatochromism was also observed for the lower energy transition of 5 (Δ![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif) = 120 cm−1). Electron-withdrawing substituents have therefore only a minor influence and the electronic delocalization is poorly perturbed, leading to limited spectroscopic changes.
= 120 cm−1). Electron-withdrawing substituents have therefore only a minor influence and the electronic delocalization is poorly perturbed, leading to limited spectroscopic changes.
In contrast and not surprisingly, absorption maxima are red-shifted with electron-donating groups (Fig. 4, bottom). The smallest effects are observed for the carboxylato 16, azido 9 and olefinic 13a derivatives that exhibit maxima centered at 626, 638 and 644 nm, respectively. Within the series of compounds 13a to 13f, it was noticed that extra substituents on the styrene fragments have little influence. Only in the case of p-aminophenyl 13d, is a broader transition observed (with a lower intensity and a shift of +20 nm compared to 13a). More noticeable effects are displayed with 14 and 15 that present both similar absorption properties (λabs = 659 and 653 nm respectively). The most dramatically red-shifted absorptions are recorded for dyes 7 and 8, with the absorption red edge extending to the near-infrared (NIR) range, around 800–850 nm. Interestingly, the primary amine function in 7 induces a stronger shift of the absorption towards lower energies compared to its tertiary amine functionalized analogue 8. This bathochromic effect brought on by electron-donating groups is further characterized by a progressive decrease of the molar extinction coefficient from ε = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 to 5580 M−1 cm−1 in compounds 16 to 8. A marked broadening of the transition towards quasi-gaussian profiles also results from the increase in electron-donating strength of the substituents (fwhm = 2840 to 3280 cm−1 from 16 to 8). Such a trend is characteristic of the establishment of an internal charge transfer. As previously, the solvatochromism was studied and compounds 7, 8, 14 and 15 were selected (Fig. S4†). Products 8 and 15 show no variation of absorption maxima and fwhm (Δ
400 to 5580 M−1 cm−1 in compounds 16 to 8. A marked broadening of the transition towards quasi-gaussian profiles also results from the increase in electron-donating strength of the substituents (fwhm = 2840 to 3280 cm−1 from 16 to 8). Such a trend is characteristic of the establishment of an internal charge transfer. As previously, the solvatochromism was studied and compounds 7, 8, 14 and 15 were selected (Fig. S4†). Products 8 and 15 show no variation of absorption maxima and fwhm (Δ![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif) = 196 and 209 cm−1, respectively), and hence a lack of solvatochromism. In contrast, non-methylated analogues 7 and 14 display an inverse solvatochromism characterized by bathochromic and hypochromic shifts upon lowering the polarity of the medium (Δ
= 196 and 209 cm−1, respectively), and hence a lack of solvatochromism. In contrast, non-methylated analogues 7 and 14 display an inverse solvatochromism characterized by bathochromic and hypochromic shifts upon lowering the polarity of the medium (Δ![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif) = 744 and 944 cm−1, respectively). The appearance of solvatochromism with the non-methylated 7 and 14 suggests the occurrence of H-bonding interactions.
= 744 and 944 cm−1, respectively). The appearance of solvatochromism with the non-methylated 7 and 14 suggests the occurrence of H-bonding interactions.
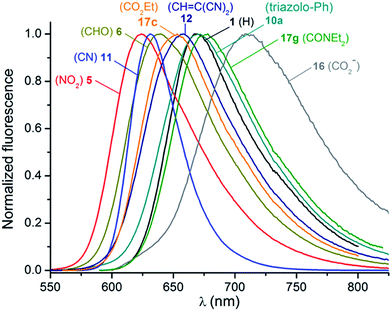
Fluorescence
The emission properties of the [4]helicenes were also recorded in acetonitrile and selected fluorescence spectra are displayed in Fig. 5. 1 shows an emission maximum at 667 nm characterized by a modest fluorescence quantum yield of 0.13 and a lifetime of 5.5 ns, as previously reported.14a The majority of the chromophores functionalized with an electron-withdrawing function exhibit moderate to strong emission in the red optical region with quantum yields ranging from 0.13 (17g) to 0.55 (11). The fluorescence lifetimes are relatively high for organic chromophores in this spectral range, with values between 6.2 ns (17g) and 19.1 ns (11), as presented in Fig. S6.† In this series, the emission spectra are the mirror image of the lower energy absorption bands and show Stokes shifts values of ∼1300–1600 cm−1. In contrast and interestingly, fluorescence of the electron-enriched chromophores was not observed under the current experimental conditions. As displayed in Fig. S7,† the quantum efficiency declines progressively with the concomitant increase in emission wavelength, illustrating the effect of the energy gap law.30 Moreover, the radiative and non-radiative kinetic constants compared in Table 2 highlight the strong increase of de-excitation via non-radiative pathways as the fluorescence maxima are red-shifted while the radiative constants remain in the same range of values (20–30 × 10−6 s−1), assuming a negligibly small triplet yield.Electronic circular dichroism and circularly polarized luminescence
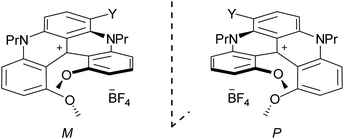
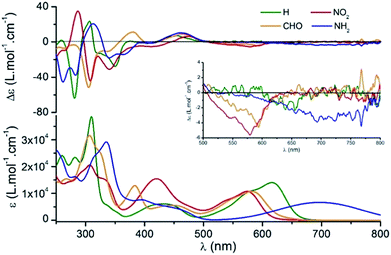
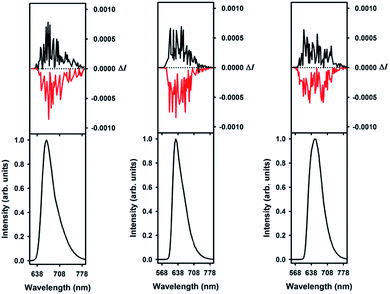
Using M-1 and P-1 as precursors instead of the racemate,12b compounds 5, 6 and 7 were prepared as single enantiomers (Fig. 6). In Fig. 7 the UV-Vis-NIR electronic circular dichroism (ECD) spectra of the M enantiomers are displayed. The spectra of the P antipodes are reported in the ESI (Fig. S8†). The four dyes present pronounced ECD bands in the UV range with a Cotton effect near their most intense higher energy transitions. As predicted by Elm and co-workers,31 the functionalized helicenes feature stronger circular dichroism in the visible region than the parent 1. Chromophores M-5 and M-6 unambiguously display absorption of circularly polarized light between 400 and 600 nm, corresponding to the first and second low energy absorption transitions. Compound M-7 constitutes a rare example of a purely organic helicene exhibiting ECD in the far-red and NIR region of the electromagnetic spectrum.6c,d,32The circularly polarized luminescence (CPL) spectra were recorded for both enantiomers of 1, 5 and 6 and are presented in Fig. 8. Although a very weak CPL was measured, opposite trend signals were observed for the P-(+) and M-(−) enantiomers in the same wavelength as the corresponding unpolarized fluorescence. The glum values are +0.0013/−0.0010, +0.0016/−0.0017, and +0.0009/−0.0008 at the vicinity of the maximum emission wavelength for the P-(+)/M-(−) helices of 1, 5, and 6, respectively. These CPL intensities are in the same order as previously reported chiral organic dyes and bodipys33 but present the originality to be located in the red range, an unusual spectral region for helicene-like chromophores.2 To our knowledge, only helicene derivatives benefiting from the presence of transition metal fragments have achieved similar outputs.34
Conclusions
In summary, direct post-functionalization routes to a large variety of substituted cationic diaza [4]helicenes have been achieved. More than twenty new chromophores were prepared in moderate to excellent yields (55–99%). Structural solid-state analyses have revealed higher helical angles and pitches upon introduction of the substituents (up to 45.9° and 3.26 Å). Moreover, a strongly resonant π-system was evidenced by the non-alternant bond lengths of the conjugated core. Furthermore a broad tuning of the electrochemical and optical properties can be achieved by the selection of the peripheral substituent. While electron-withdrawing functional groups led to highly fluorescent derivatives emitting in the red domain, electron-donating residues allowed an extension of the low-energy absorption band towards the far-red and NIR regions. Thanks to these properties, ECD and CPL spectra were recorded at unusual wavelengths for purely organic helicenes, in the far-red spectral range. Some of these compounds should be well-suited for high-contrast imaging applications using confocal microscopy in the transparency window of biological media.13b,35Acknowledgements
We thank the University of Geneva and the Swiss National Science Foundation for financial support. We acknowledge the contributions of the Sciences Mass Spectrometry (SMS) platform at the Faculty of Sciences, University of Geneva. G. M. thanks the NIH, Minority Biomedical Research Support (grant 1 SC3 GM089589-07) and the Henry Dreyfus Teacher-Scholar Award for financial support, whereas RCT thanks the SJSU RISE program (NIH grant 5R25GM71381) for a research fellowship.References
- (a) R. H. Martin, Angew. Chem., Int. Ed., 1974, 13, 649–660 CrossRef; (b) A. Urbano, Angew. Chem., Int. Ed., 2003, 42, 3986–3989 CrossRef CAS PubMed; (c) Y. Shen and C.-F. Chen, Chem. Rev., 2011, 112, 1463–1535 CrossRef PubMed; (d) M. Gingras, Chem. Soc. Rev., 2013, 42, 968–1006 RSC.
- (a) K. E. S. Phillips, T. J. Katz, S. Jockusch, A. J. Lovinger and N. J. Turro, J. Am. Chem. Soc., 2001, 123, 11899–11907 CrossRef CAS PubMed; (b) J. E. Field, G. Muller, J. P. Riehl and D. Venkataraman, J. Am. Chem. Soc., 2003, 125, 11808–11809 CrossRef CAS PubMed; (c) R. Hassey, E. J. Swain, N. I. Hammer, D. Venkataraman and M. D. Barnes, Science, 2006, 314, 1437–1439 CrossRef CAS PubMed; (d) T. Kaseyama, S. Furumi, X. Zhang, K. Tanaka and M. Takeuchi, Angew. Chem., Int. Ed., 2011, 50, 3684–3687 CrossRef CAS PubMed; (e) K. Nakamura, S. Furumi, M. Takeuchi, T. Shibuya and K. Tanaka, J. Am. Chem. Soc., 2014, 136, 5555–5558 CrossRef CAS PubMed.
- (a) D. J. Morrison, T. K. Trefz, W. E. Piers, R. McDonald and M. Parvez, J. Org. Chem., 2005, 70, 5309–5312 CrossRef CAS PubMed; (b) H. Oyama, K. Nakano, T. Harada, R. Kuroda, M. Naito, K. Nobusawa and K. Nozaki, Org. Lett., 2013, 15, 2104–2107 CrossRef CAS PubMed; (c) L. Shi, Z. Liu, G. Dong, L. Duan, Y. Qiu, J. Jia, W. Guo, D. Zhao, D. Cui and X. Tao, Chem.–Eur. J., 2012, 18, 8092–8099 CrossRef CAS PubMed; (d) W. Hua, Z. Liu, L. Duan, G. Dong, Y. Qiu, B. Zhang, D. Cui, X. Tao, N. Cheng and Y. Liu, RSC Adv., 2015, 5, 75–84 RSC.
- (a) L. Latterini, E. Galletti, R. Passeri, A. Barbafina, L. Urbanelli, C. Emiliani, F. Elisei, F. Fontana, A. Mele and T. Caronna, J. Photochem. Photobiol., A, 2011, 222, 307–313 CrossRef CAS; (b) M. Li, L.-H. Feng, H.-Y. Lu, S. Wang and C.-F. Chen, Adv. Funct. Mater., 2014, 24, 4405–4412 CrossRef CAS.
- (a) G. Qian and Z. Y. Wang, Chem.–Asian J., 2010, 5, 1006–1029 CrossRef CAS PubMed; (b) Z. Guo, S. Park, J. Yoon and I. Shin, Chem. Soc. Rev., 2014, 43, 16–29 RSC.
- (a) S. Arai, T. Yafune, M. Ōkubo and M. Hida, Tetrahedron Lett., 1989, 30, 7217–7218 CrossRef CAS; (b) F. Torricelli, J. Bosson, C. Besnard, M. Chekini, T. Bürgi and J. Lacour, Angew. Chem., Int. Ed., 2013, 52, 1796–1800 CrossRef CAS PubMed; (c) L. Pospíšil, L. Bednárová, P. Štěpánek, P. Slavíček, J. Vávra, M. Hromadová, H. Dlouhá, J. Tarábek and F. Teplý, J. Am. Chem. Soc., 2014, 136, 10826–10829 CrossRef PubMed; (d) P. E. Reyes-Gutierrez, M. Jirasek, L. Severa, P. Novotna, D. Koval, P. Sazelova, J. Vavra, A. Meyer, I. Cisarova, D. Saman, R. Pohl, P. Stepanek, P. Slavicek, B. J. Coe, M. Hajek, V. Kasicka, M. Urbanova and F. Teply, Chem. Commun., 2015, 51, 1583–1586 RSC; (e) S. Menichetti, S. Cecchi, P. Procacci, M. Innocenti, L. Becucci, L. Franco and C. Viglianisi, Chem. Commun., 2015, 51, 11452–11454 RSC; (f) D. Sakamaki, D. Kumano, E. Yashima and S. Seki, Chem. Commun., 2015, 51, 17237–17240 RSC.
- For recent developments with metallo-organic helicenes, see (a) L. Norel, M. Rudolph, N. Vanthuyne, J. A. G. Williams, C. Lescop, C. Roussel, J. Autschbach, J. Crassous and R. Réau, Angew. Chem., Int. Ed., 2010, 49, 99–102 CrossRef CAS PubMed; (b) E. Anger, M. Srebro, N. Vanthuyne, L. Toupet, S. Rigaut, C. Roussel, J. Autschbach, J. Crassous and R. Réau, J. Am. Chem. Soc., 2012, 134, 15628–15631 CrossRef CAS PubMed; (c) E. Anger, M. Srebro, N. Vanthuyne, C. Roussel, L. Toupet, J. Autschbach, R. Reau and J. Crassous, Chem. Commun., 2014, 50, 2854–2856 RSC; (d) D. Schweinfurth, M. Zalibera, M. Kathan, C. Shen, M. Mazzolini, N. Trapp, J. Crassous, G. Gescheidt and F. Diederich, J. Am. Chem. Soc., 2014, 136, 13045–13052 CrossRef CAS PubMed; (e) C. Shen, E. Anger, M. Srebro, N. Vanthuyne, K. K. Deol, T. D. Jefferson, G. Muller, J. A. G. Williams, L. Toupet, C. Roussel, J. Autschbach, R. Reau and J. Crassous, Chem. Sci., 2014, 5, 1915–1927 RSC; (f) N. Saleh, B. Moore, M. Srebro, N. Vanthuyne, L. Toupet, J. A. G. Williams, C. Roussel, K. K. Deol, G. Muller, J. Autschbach and J. Crassous, Chem.–Eur. J., 2015, 21, 1673–1681 CrossRef CAS PubMed; (g) N. Saleh, M. Srebro, T. Reynaldo, N. Vanthuyne, L. Toupet, V. Y. Chang, G. Muller, J. A. G. Williams, C. Roussel, J. Autschbach and J. Crassous, Chem. Commun., 2015, 51, 3754–3757 RSC.
- pKR+ values are used to define the chemical stability of carbenium ions in water by the quantification of the equilibrium constant between the carbocationic and the corresponding carbinol forms. The higher and positive the pKR+ value, the higher the chemical stability of the carbenium ion. For the formula and calculations of pKR+ values, see ref. 10b.
- (a) B. W. Laursen and F. C. Krebs, Angew. Chem., Int. Ed., 2000, 39, 3432–3434 CrossRef CAS; (b) C. Herse, D. Bas, F. C. Krebs, T. Bürgi, J. Weber, T. Wesolowski, B. W. Laursen and J. Lacour, Angew. Chem., Int. Ed., 2003, 42, 3162–3166 CrossRef CAS PubMed; (c) J. Bosson, J. Gouin and J. Lacour, Chem. Soc. Rev., 2014, 43, 2824–2840 CAS; (d) T. J. Sørensen, M. F. Nielsen and B. W. Laursen, ChemPlusChem, 2014, 79, 1030–1035 CrossRef.
- (a) J. C. Martin and R. G. Smith, J. Am. Chem. Soc., 1964, 86, 2252–2256 CrossRef CAS; (b) B. W. Laursen and F. C. Krebs, Chem.–Eur. J., 2001, 7, 1773–1783 CrossRef CAS PubMed.
- J. Guin, C. Besnard, P. Pattison and J. Lacour, Chem. Sci., 2011, 2, 425–428 RSC.
- (a) G. Solladié, J. Hutt and A. Girardin, Synthesis, 1987, 173 CrossRef; (b) B. Laleu, P. Mobian, C. Herse, B. W. Laursen, G. Hopfgartner, G. Bernardinelli and J. Lacour, Angew. Chem., 2005, 117, 1913–1917 CrossRef; (c) B. Laleu, M. S. Machado and J. Lacour, Chem. Commun., 2006, 2786–2788 RSC.
- (a) P. Mobian, N. Banerji, G. Bernardinelli and J. Lacour, Org. Biomol. Chem., 2006, 4, 224–231 RSC; (b) O. Kel, A. Fürstenberg, N. Mehanna, C. Nicolas, B. Laleu, M. Hammarson, B. Albinsson, J. Lacour and E. Vauthey, Chem.–Eur. J., 2013, 19, 7173–7180 CrossRef CAS PubMed.
- (a) O. Kel, P. Sherin, N. Mehanna, B. Laleu, J. Lacour and E. Vauthey, Photochem. Photobiol. Sci., 2012, 11, 623–631 RSC; (b) A. Wallabregue, P. Sherin, J. Guin, C. Besnard, E. Vauthey and J. Lacour, Eur. J. Org. Chem., 2014, 2014, 6431–6438 CrossRef CAS.
- (a) B. W. Laursen, F. C. Krebs, M. F. Nielsen, K. Bechgaard, J. B. Christensen and N. Harrit, J. Am. Chem. Soc., 1998, 120, 12255–12263 CrossRef CAS; (b) F. Westerlund, C. B. Hildebrandt, T. J. Sørensen and B. W. Laursen, Chem.–Eur. J., 2010, 16, 2992–2996 CrossRef CAS PubMed; (c) T. J. Sørensen and B. W. Laursen, J. Org. Chem., 2010, 75, 6182–6190 CrossRef PubMed.
- Polyhalogenation had been only observed on neutral trioxatricornan and cationic triazatriangulenium analogues, see: (a) M. Lofthagen, R. Vernon Clark, K. K. Baldridge and J. S. Siegel, J. Am. Chem. Soc., 1991, 57, 61–69 Search PubMed; (b) X.-M. Hu, Q. Chen, Z.-Y. Sui, Z.-Q. Zhao, N. Bovet, B. W. Laursen and B.-H. Han, RSC Adv., 2015, 5, 90135–90143 RSC.
- K. Barral, A. D. Moorhouse and J. E. Moses, Org. Lett., 2007, 9, 1809–1811 CrossRef CAS PubMed.
- B. V. Rokade and K. R. Prabhu, J. Org. Chem., 2012, 77, 5364–5370 CrossRef CAS PubMed.
- A formanilide byproduct (Y = NHCHO) can be observed and it presents a characteristic green color.
- Hydrogenation over Pd/C of 13b afforded the reduction of the peripheral double bond instead.
- E. T. Da Silva, C. A. Câmara, O. A. C. Antunes, E. J. Barreiro and C. A. M. Fraga, Synth. Commun., 2008, 38, 784–788 CrossRef.
- E. Dalcanale and F. Montanari, J. Org. Chem., 1986, 51, 567–569 CrossRef CAS.
- Treatment of solutions of 16 in dichloromethane with aqueous NaBF4 solutions did not reveal, after phase separation, any presence of tetrafluoroborate counterion in the organic layer (19F NMR spectroscopy).
- For a study of the conformation of substituted benzaldehyde derivatives, see: S. Pache, P. Romanens and E. P. Kündig, Organometallics, 2003, 22, 377–380 CrossRef CAS.
- For the single 7 derivative, probably due to a strong charge transfer from the donor amino group to the central cationic core, the top cycle displays a weak double bond character (C(7)–C(8) = 1.372 Å, Fig. S1†).
- D. Conreaux, N. Mehanna, C. Herse and J. Lacour, J. Org. Chem., 2011, 76, 2716–2722 CrossRef CAS PubMed.
- In Table 1, compounds are ranked by the increasing first oxidation potentials.
- C. Reichardt, Solvents and Solvent Effects in Organic Chemistry, 2004, Wiley-VCH Verlag GmbH & Co. KGaA Search PubMed.
- Emission bands of 1 and 5 display moderate variations in the same solvents (THF to DMSO, Δ
![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif) ∼ 480 cm−1, Fig. S5†).
∼ 480 cm−1, Fig. S5†). - (a) R. Englman and J. Jortner, Mol. Phys., 1970, 18, 145–164 CrossRef CAS; (b) J. R. Lakowicz, Principles of Fluorescence Spectroscopy, 2007, Springer Search PubMed.
- J. Elm, J. Lykkebo, T. J. Sørensen, B. W. Laursen and K. V. Mikkelsen, J. Phys. Chem. A, 2012, 116, 8744–8752 CrossRef CAS PubMed.
- T. Biet, A. Fihey, T. Cauchy, N. Vanthuyne, C. Roussel, J. Crassous and N. Avarvari, Chem.–Eur. J., 2013, 19, 13160–13167 CrossRef CAS PubMed.
- (a) E. M. Sánchez-Carnerero, F. Moreno, B. L. Maroto, A. R. Agarrabeitia, M. J. Ortiz, B. G. Vo, G. Muller and S. de la Moya, J. Am. Chem. Soc., 2014, 136, 3346–3349 CrossRef PubMed; (b) S. Zhang, Y. Wang, F. Meng, C. Dai, Y. Cheng and C. Zhu, Chem. Commun., 2015, 51, 9014–9017 RSC; (c) E. M. Sánchez-Carnerero, A. R. Agarrabeitia, F. Moreno, B. L. Maroto, G. Muller, M. J. Ortiz and S. de la Moya, Chem.–Eur. J., 2015, 21, 13488–13500 CrossRef PubMed.
- (a) C. Shen, E. Anger, M. Srebro, N. Vanthuyne, K. K. Deol, T. D. Jefferson, G. Muller, J. A. G. Williams, L. Toupet, C. Roussel, J. Autschbach, R. Reau and J. Crassous, Chem. Sci., 2014, 5, 1915–1927 RSC; (b) N. Saleh, B. Moore, M. Srebro, N. Vanthuyne, L. Toupet, J. A. G. Williams, C. Roussel, K. K. Deol, G. Muller, J. Autschbach and J. Crassous, Chem.–Eur. J., 2015, 21, 1673–1681 CrossRef CAS PubMed; (c) N. Saleh, M. Srebro, T. Reynaldo, N. Vanthuyne, L. Toupet, V. Y. Chang, G. Muller, J. A. G. Williams, C. Roussel, J. Autschbach and J. Crassous, Chem. Commun., 2015, 51, 3754–3757 RSC; (d) H. Isla and J. Crassous, C. R. Chim., 2016, 19, 39–49 CrossRef CAS.
- (a) S. Honzawa, H. Okubo, S. Anzai, M. Yamaguchi, K. Tsumoto and I. Kumagai, Bioorg. Med. Chem., 2002, 10, 3213–3218 CrossRef CAS PubMed; (b) R. Amemiya and M. Yamaguchi, Org. Biomol. Chem., 2008, 6, 26–35 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Synthetic protocols, 1H/13C/19F NMR and HR mass spectra, additional electrochemical and optical data. CCDC 1443632, 1443633, 1443635–1443641 and 1470066. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6sc00614k |
| This journal is © The Royal Society of Chemistry 2016 |

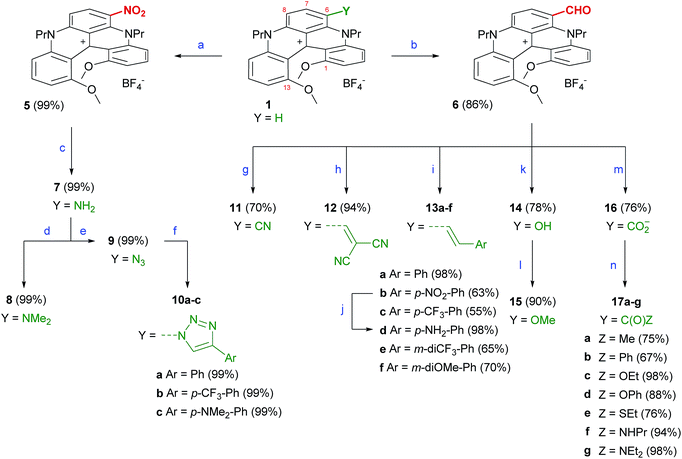
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH, CuSO4·5H2O (0.2 equiv.), ascorbic acid (0.3 equiv.), NaHCO3 (0.3 equiv.), MeCN/H2O, 25 °C, 12 h. (g) NaN3 (1.5 equiv.), TfOH (3 equiv.), MeCN, 25 °C, 10 min. (h) NCCH2CN (3 equiv.), Ph3P (20 mol%), MeCN, 130 °C (MW), 25 °C, 1 h. (i) (i) ArCH2X (1.2 equiv.), Ph3P (1.3 equiv.), CH2Cl2; (ii) NaH (2 equiv.), DCM, 25 °C, 30 min; (iii) 1 M aq. HBF4. (j) AcOH (15 equiv.), Zn (5 equiv.), CH2Cl2, 25 °C, 3 h. (k)
CH, CuSO4·5H2O (0.2 equiv.), ascorbic acid (0.3 equiv.), NaHCO3 (0.3 equiv.), MeCN/H2O, 25 °C, 12 h. (g) NaN3 (1.5 equiv.), TfOH (3 equiv.), MeCN, 25 °C, 10 min. (h) NCCH2CN (3 equiv.), Ph3P (20 mol%), MeCN, 130 °C (MW), 25 °C, 1 h. (i) (i) ArCH2X (1.2 equiv.), Ph3P (1.3 equiv.), CH2Cl2; (ii) NaH (2 equiv.), DCM, 25 °C, 30 min; (iii) 1 M aq. HBF4. (j) AcOH (15 equiv.), Zn (5 equiv.), CH2Cl2, 25 °C, 3 h. (k)