The preparation of high-quality water-soluble silicon quantum dots and their application in the detection of formaldehyde†
Xiaoling Xu,
Shiyao Ma,
Xincai Xiao,
Yan Hu and
Dan Zhao*
School of Pharmaceutical Sciences, South-Central University for Nationalities, Wuhan 430074, P. R. China. E-mail: wqzhdpai@163.com
First published on 11th October 2016
Abstract
This paper reports the synthesis of water-soluble fluorescence silicon quantum dots (Si QDs) through a hydrothermal route with urea propyl triethoxysilane (UPTES) as the source of silicon and sodium citrate as the deoxidizer. The UPTES was for the first time reported as the source of silicon. The preparation process is simple and green, and the prepared Si QDs exhibit high quantum yields, which would be ideal as a low-toxic material in biochemical applications. In addition, the prepared QDs emit excellent and stable fluorescence in a wide range of pH values (2–14), and show strong tolerance to salt and several common organic reagents, overcoming the weak anti-interference ability of traditional QDs and thus ensuring them as satisfactory candidates for biochemical detection. Using the prepared QDs as fluorescence probes, formaldehyde has been successfully detected in aqueous phase and acetonitrile through an electron transfer mechanism. The detection method is simple, sensitive and strongly anti-interfering, providing a new way for detecting formaldehyde in different solvents and expanding the potential applications of water-soluble low-toxic QDs in biochemical detection.
1. Introduction
The great development of water-soluble quantum dots (QDs) has shown their immense potential in biological imaging,1,2 drug delivery systems3 and detection.4,5 Apparently, their biological safety is still a hot topic.6 Si QDs have attracted more attention due to their good biocompatibility, environmental-friendliness, inexpensive raw materials and richness in their synthesis methods.The common methods of preparing Si QDs can be classified into two types: two-step synthesis and one-step synthesis. The two-step synthesis procedures include liquid reduction methods,7 photochemical etching methods,8 high temperature pyrolysis methods9 and plasma synthesis.10 The universally-used silicon sources in two-step synthesis methods are silica powder, silicon halides, silicon Zintl salt (i.e. ASix, A = Na, K, Mg etc.), or silicon oxides,11 leading to poor water-solubility of the prepared Si QDs and inferior stability caused by easy oxidation tendency. Therefore, further modification is used to solve these problems,12,13 which would cause attendant problems like decreased fluorescence intensity of prepared QDs,14 aggregation of QDs in alkaline conditions15 and the time consuming synthesis process.16,17
On the other hand, the one-step synthesis methods contains microwave method,19 ultraviolet radiation method2 and hydrothermal route.4,5,20–22 Hydrothermal route requires no complicated and plentiful equipment, and is environmental friendly and convenient in operation, making it an appropriate method for Si QDs preparation. However, most of the fluorescence emissions of Si QDs prepared by hydrothermal route are blue, which can be easily interfered by tissue scatter and auto-fluorescence. Some team has already overcome the problem by coupling dye on the surface of Si QDs to make QDs suitable for biological imaging.18
The characteristics of the silicon source would greatly affect the synthesis factors of QDs. Recently, silicon sources for hydrothermal route are quite limited and the proposed synthesis process often take too long time (160–180 °C, 16–20 h).4,5 Filtering proper silicon sources would be significant to the shortening of interaction time, the simplification and efficiency of synthesis process. In addition, it has been proposed that silicon quantum dots synthesised by the silicon source containing two nitrogen atoms (such as N-[3-(trimethoxysilyl)propyl]ethylenediamine) possess higher quantum yield35 when compared with those prepared by APTMS, indicating that the proper nitrogen content of silicon sources will enhance the quality of prepared Si QDs. Therefore, it is necessary to develop a new silicon source containing two nitrogen atoms optimally. In addition, the diversity of silicon sources can also enriches the surface groups of Si QDs and thus extends their applications in the field of detection and analysis.
As we all known, silicon quantum dots has become an important tool in the field of biochemical detection.19–26 Since formaldehyde is harmful to both environment and human health,27 a fast, simple and sensitive detection of formaldehyde in organic reagents is essential to the purity examination of organic reagents as well as human health and natural environment protection. Many methods have been explored to realize the detection of formaldehyde, such as ultraviolet spectrophotometry,28 electrochemical sensor,29 Raman spectroscopy,30 capillary electrophoresis,31 high performance liquid chromatography,32 and fluorescent spectrometry.27,33 Some of these methods are time consuming and possess some drawbacks in selectivity and sensitivity. Therefore, a simple detection method of formaldehyde is challenging. Fluorescent spectrometry is an important research method in medicine, chemistry, biology, pharmacy and physics fields.24–26 Hitherto, the detection of formaldehyde by fluorescent probes are rarely reported, because of the necessity for the synthesis of formaldehyde-responding compounds,33 the complicated synthesis process and poor selectivity.34 In addition, most of the reports on the detection of formaldehyde focus on the interactions in aqueous and gaseous phases, and the detection in organic solvents are rarely seen in reports. Therefore, the detection of formaldehyde in organic reagents is still challenging.
This paper reports the synthesis of water-soluble Si QDs through hydrothermal route with urea propyl triethoxysilane as a new source of silicon, which greatly shortens the reaction time and enhances QYs. The prepared QDs exhibit strong salt tolerance and stability in wide range of pH values. Furthermore, with the as-prepared Si QDs as fluorescence probe, the formaldehyde has been successfully detected in aqueous phase and acetonitrile, offering new outlook for the preparation of low-toxic QDs and their applications.
2. Experimental
2.1 Chemicals
Urea propyl triethoxysilane (UPTES), 3-aminopropyltrimethoxy-silane (APTMS) and 3-aminopropyltriethoxysilane (APTES) were obtained from Aladdin Chemistry Co., Ltd (China); HCHO, Na2HPO4·12(H2O), KH2PO4, NaOH and sodium citrate were purchased from Sinopharm Chemical Reagent Co., Ltd (China); chromatographic grade acetonitrile were purchased from Tedia Company Inc (USA). All solutions were prepared using Milli-Q water as the solvent. L-929 cell line was obtained from Wuhan Bioyeargene Biotechnology Co., Ltd (China). Dulbecco's modified Eagle's medium (DMEM) and fetal bovine serum (FBS) were purchased from Hyclone Laboratories, Inc (USA). 1-Methyltetrazole-5-thiol (MTT) were purchased from Sigma Corporation (USA).2.2 Synthesis of aqueous Si QDs
We used UPTES as the source of silicon, and trisodium citrate as the reduction reagent for the hydrothermal synthesis of the Si QDs. UPTES (1 mL) was added into sodium citrate solution (40 mM), followed by the removal of oxygen by purging with nitrogen gas for 20 min. The precursor solution was adjusted to specified pH value. Finally, the solution was transferred into a 50 mL Teflon-lined autoclave. It was loaded in an oven at 200 °C for different time and then cooled to room temperature naturally. The Si quantum dots synthetised by APTMS and APTES are performed according to the previous work.4,5In order to purify as-prepared Si quantum dots, quadruple volume acetonitrile (volume ratio) were added into the solution of quantum dots and precipitated through centrifugation at 8000 rpm for 10 min.
2.3 Characterization
UV-visible absorption spectra were acquired with a Lambda-35 UV/visible spectrophotometer (PerkinElmer Company, USA) to determine the bandgap absorption of Si QDs. Fluorescence emission spectra were measured using LS55 spectrofluorometer (PerkinElmer Company, USA). X-ray photoelectron spectroscope (XPS) spectra were investigated at a VG Multilab 2000 X-ray photoelectron spectrometer (Thermo Electron Corporation, USA). Fourier transform infrared (FTIR) spectroscopy experiments were carried out for freeze-dried Si QDs. TEM image was obtained at 310 K magnification with an FEI Tecnai G2 20 s-twin transmission electron microscope (USA). SEM-EDS spectra were performed by the SEM-associated energy dispersive spectroscopy (EDAX GENSIS, AMETEK, USA). The relative PLQYs of as-prepared Si QDs were measured according to literatures with quinine sulfate in 0.1 M H2SO4 (QY = 58%) as a reference standard. All optical measurements were performed at room temperature under ambient conditions.2.4 MTT
L-929 cells were trypsinized and resuspended in DMEM with 10% of FBS and 1% of penicillin/streptomycin. The cells were seeded in a 96 well microplate at a density of 1 × 104 cells in 100 μL of complete DMEM culture medium, and incubated overnight at 37 °C under 5% CO2 atmosphere. The QDs were loaded in each well with a final concentration of 50, 100, 150, 200, 300, 400, 500 μg mL−1. After incubation for 24 h, 100 μL of MTT (0.5 mg mL−1) was added to each well. The medium was discarded by aspiration, and the intracellular purple formazan crystals were dissolved in 150 μL of dimethyl sulfoxide (DMSO). The plates were read for absorbance at 490 nm. Three duplicates were obtained for each concentration; the cell viability was calculated by normalizing with the results obtained with no QD-loading.2.5 Detection of formaldehyde in aqueous solution and acetonitrile
Samples containing buffer solution (Tris–HCl, 20 mM, pH = 6.0), different amounts of formaldehyde and Si QDs (1.55 × 10−5 g mL−1) were made up to 1 mL. The emission spectrum of the solution was then measured 10 min later. All optical measurements were performed at room temperature under ambient conditions, and the excitation wavelength (λex) was 365 nm, slit width was set at 10–15.Samples containing chromatographic grade acetonitrile, different amounts of formaldehyde and Si QDs (1.55 × 10−5 g mL−1) were made up to 1 mL. The fluorescence spectra were recorded 10 minutes later (λex = 365 nm).
3. Result and discussion
3.1 The synthesis of fluorescent Si QDs
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.55 (UPTES/sodium citrate), reaction hour at 2 h and pH value at 10.0. Due to the extremely low synthesis efficiency under low temperature, the investigation mainly focused on the impact of higher temperatures (>180 °C). As shown in Fig. 1, the fluorescence intensity of prepared Si QDs enhances more than 100% when the reaction temperature rises from 180 °C to 185 °C, because of the fast core growth and the reduction of surface defects. The fluorescence intensity continues enhancing at slower pace when temperature raises from 185 °C to 200 °C. However, when reaction temperature keeps rising to above 200 °C, the fluorescence intensity greatly decreases. That is because of the existence of self-aggregation of silane37 during the process of QDs growth and the speed-up of the side-reaction caused by high temperature and the consequent deterioration of the fluorescence properties of prepared QDs. Therefore, the optimal reaction temperature is set at 200 °C.
0.55 (UPTES/sodium citrate), reaction hour at 2 h and pH value at 10.0. Due to the extremely low synthesis efficiency under low temperature, the investigation mainly focused on the impact of higher temperatures (>180 °C). As shown in Fig. 1, the fluorescence intensity of prepared Si QDs enhances more than 100% when the reaction temperature rises from 180 °C to 185 °C, because of the fast core growth and the reduction of surface defects. The fluorescence intensity continues enhancing at slower pace when temperature raises from 185 °C to 200 °C. However, when reaction temperature keeps rising to above 200 °C, the fluorescence intensity greatly decreases. That is because of the existence of self-aggregation of silane37 during the process of QDs growth and the speed-up of the side-reaction caused by high temperature and the consequent deterioration of the fluorescence properties of prepared QDs. Therefore, the optimal reaction temperature is set at 200 °C.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.55, the pH value of the precursor was 8.17, and the prepared Si QDs exhibit excellent optical property with some flocculent precipitate. Sodium hydroxide or hydrochloric acid was used to adjust the pH value of the precursor (6.5–11.4). As shown in Fig. 2, the fluorescence intensities of prepared Si QDs exhibit no obvious change when the pH value is in the range of 6.5 to 10.0, but the precipitate of the reaction reduced with the increase of pH value. The clear and transparent solution of QDs can be acquired with pH value at 10.0 (inset of Fig. 2). The flocculent precipitate might be the result of condensation reaction of ethoxysilane in the process of hydrolysis37 when pH value of the precursor is below 10.0. However, when further increases pH value, the fluorescence intensity exhibits obvious decrease. The intensity drops 87.6% when pH increases from 10.8 to 11.4. Therefore, the optimal pH value was set at 10.0.
0.55, the pH value of the precursor was 8.17, and the prepared Si QDs exhibit excellent optical property with some flocculent precipitate. Sodium hydroxide or hydrochloric acid was used to adjust the pH value of the precursor (6.5–11.4). As shown in Fig. 2, the fluorescence intensities of prepared Si QDs exhibit no obvious change when the pH value is in the range of 6.5 to 10.0, but the precipitate of the reaction reduced with the increase of pH value. The clear and transparent solution of QDs can be acquired with pH value at 10.0 (inset of Fig. 2). The flocculent precipitate might be the result of condensation reaction of ethoxysilane in the process of hydrolysis37 when pH value of the precursor is below 10.0. However, when further increases pH value, the fluorescence intensity exhibits obvious decrease. The intensity drops 87.6% when pH increases from 10.8 to 11.4. Therefore, the optimal pH value was set at 10.0.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.55. That is because in a certain range of ratio, the increased presence of deoxidizer could promote the formation of QDs core and be beneficial to QDs growth. The abundant combination of deoxidizer and the silicon source could introduces new gap states in Si QDs so that more electrons transits from the lowest unoccupied molecular orbits to new energy gap states or the highest occupied molecular orbits, leading to the enhanced fluorescence intensity.2 The excess amount of deoxidizer does not produce negative effect to the synthesis system.
0.55. That is because in a certain range of ratio, the increased presence of deoxidizer could promote the formation of QDs core and be beneficial to QDs growth. The abundant combination of deoxidizer and the silicon source could introduces new gap states in Si QDs so that more electrons transits from the lowest unoccupied molecular orbits to new energy gap states or the highest occupied molecular orbits, leading to the enhanced fluorescence intensity.2 The excess amount of deoxidizer does not produce negative effect to the synthesis system.
3.2 The characterization of fluorescent Si QDs
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.55, pH value at 10.0, reaction temperature at 200 °C, and reaction time at 2 h. As shown in fluorescence emission spectra and UV-Vis absorption spectra of the as-prepared Si QDs in Fig. 4, the symmetrical fluorescence peak and the obvious UV absorption peak illustrate excellent fluorescence properties of prepared QDs. The fluorescence peak does not shift with the change of excitation wavelength, due to the homogeneous surface state formed under the high temperature and pressure in the process of hydrothermal route and the single transition mode with a certain energy rendering the emission independent from excitation wavelength.35 With quinoline sulfate as the reference (QY = 58%), the QY of prepared Si QDs reaches 27.0%.
0.55, pH value at 10.0, reaction temperature at 200 °C, and reaction time at 2 h. As shown in fluorescence emission spectra and UV-Vis absorption spectra of the as-prepared Si QDs in Fig. 4, the symmetrical fluorescence peak and the obvious UV absorption peak illustrate excellent fluorescence properties of prepared QDs. The fluorescence peak does not shift with the change of excitation wavelength, due to the homogeneous surface state formed under the high temperature and pressure in the process of hydrothermal route and the single transition mode with a certain energy rendering the emission independent from excitation wavelength.35 With quinoline sulfate as the reference (QY = 58%), the QY of prepared Si QDs reaches 27.0%.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds mainly come from the silicon source urea propyl triethoxysilane. The results acquired from XPS spectra are identical to those from IR spectra.
O bonds mainly come from the silicon source urea propyl triethoxysilane. The results acquired from XPS spectra are identical to those from IR spectra.
3.3 The comparison of Si QDs synthesized through different silicon sources
| Silicon sources | Water-solubility | Structure | Synthesis conditions | QYs | Ref. |
|---|---|---|---|---|---|
| SiO2 | Not good |  |
— | 0 | 5 |
| Si powder | Excellent | Si | 200 °C, 2–8 h | 12% | 21 |
| TEOS | Excellent | 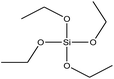 |
— | 0 | 5 |
| APTMS | Excellent |  |
160 °C, 12 h | 22% | 4 |
| 180 °C, 20 h | 20.5% | 5 | |||
| 200 °C, 24 h | 21% | 20 | |||
| APTES | Excellent | 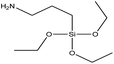 |
180 °C, 20 h | 17.8% | 5 |
| 200 °C, 3 h | 15% | 22 | |||
| UPTES | Excellent | 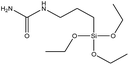 |
200 °C, 2 h | 27% | This paper |
As an element with abundant reserve on earth, silicon exists mainly in the form of silicon dioxide, which cannot directly work as silicon source. The water-solubility of silicon sources determines their degree of participation in the reaction.5 In addition, one research group also used special silicon wafer (nitrogen-doped (n-type), 0.01–0.02 sensitivity), after being ground for one hour, to prepare Si QDs through hydrothermal route with QYs at 12%.21 Through comparing tetraethoxysilane (TEOS) and some silane coupling agents, it can be discovered that when rest of the structures are similar, the nitrogen content of silicon source would greatly influence the QYs of prepared Si QDs. Without nitrogen, TEOS could not be synthesized into fluorescent Si QDs. Since the UPTES used in our experiments contains two nitrogen atoms, the prepared Si QDs thus exhibit higher QYs than those prepared by APTMS and APTES, which contain only one nitrogen atom in their structures. Similarly, the Si QDs prepared by N-[3-(trimethoxysilyl)propyl]ethylenediamine (which also contains two nitrogen atoms) through microwave route exhibit high QYs.35 Therefore, the proper nitrogen content and space structure of the silicon source are beneficial to the formation of core and surface deactivation, leading to the enhancement of QYs of prepared Si QDs.
3.4 The detection of formaldehyde
| F0/F = 1 + Ksv[c] |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times higher than that of formaldehyde (1.5 M), their influences onto fluorescence are still negligible. For acetone with similar structure as formaldehyde, its impact is slight at the concentration 100 times higher than that of formaldehyde (4 × 10−2 M). Although acetaldehyde has quite similar structure as formaldehyde, it exhibits no obvious impact on the fluorescence when its concentration is five times higher than that of formaldehyde (5.5 × 10−4 M). The results show that the prepared Si QDs reported in this paper exhibit excellent selectivity to formaldehyde.
000 times higher than that of formaldehyde (1.5 M), their influences onto fluorescence are still negligible. For acetone with similar structure as formaldehyde, its impact is slight at the concentration 100 times higher than that of formaldehyde (4 × 10−2 M). Although acetaldehyde has quite similar structure as formaldehyde, it exhibits no obvious impact on the fluorescence when its concentration is five times higher than that of formaldehyde (5.5 × 10−4 M). The results show that the prepared Si QDs reported in this paper exhibit excellent selectivity to formaldehyde.
| Detecting medium | Methods | Sensing range | Analysis time | Ref. |
|---|---|---|---|---|
| Gas and aqueous solution | Raman spectroscopy | 8.32 × 10−8 to 9.975 × 10−7 mol L−1 | Incubate 12 h | 30 |
| Solid matrice and aqueous solution | Fluorescence spectrometry | 900 nM to 1.3 × 10−4 M | Unknown | 27 |
| Food | HPLC | About 1.0–100 mg L−1 | 12 min | 39 |
| Gas | Physical method | Unknown | 1 min | 40 |
| Gas | UV-spectrophotometer | 0–6.4 ppm | 5 min | 28 |
| Aqueous solution | Electrochemical method | 0.99–9.09 mmol L−1 | Unknown | 29 |
| Aqueous solution and acetonitrile | Fluorescence spectrometry | 7.8–260 × 10−6 mol L−1 | 10 min | This work |
| 3.9–100 × 10−7 mol L−1 |
4. Conclusion
With UPTES as the novel silicon source, this paper reports the preparation of amido-modified Si QDs. The preparation greatly shortens reaction time, improves QYs and productivity of prepared QDs. It is also simple, environment-friendly without complex deactivation and modification processes. The as-prepared Si QDs exhibit excellent tolerance to pH, salt and some organic reagents, which is beneficial to their applications in biochemical detections. Since formaldehyde could effectively quench the fluorescence of prepared Si QDs through electron transfer between the carbonyl group of formaldehyde and the amino group on the surface of QDs, the sensitive detection of formaldehyde has been realized. The interfering experiments show that this system possesses excellent selectivity to formaldehyde in both aqueous phase and acetonitrile.Acknowledgements
This research was supported by Natural Science Foundation of Hubei Province (2016CFB615), the National Science Foundation of China (21105130), and the Fundamental Research Funds for the “Central Universities”, South Central University for Nationalities (CZY15020, CZW15017).Notes and references
- S. J. Zhu, N. Zhou, Z. Y. Hao, S. Maharjan, X. H. Zhao, Y. B. Song, B. Sun, K. Zhang, J. H. Zhang, H. C. Sun, L. J. Lu and B. Yang, RSC Adv., 2015, 5, 39399–39403 RSC.
- Y. L. Zhong, X. T. Sun, S. Y. Wang, F. Peng, F. Bao, Y. Y. Su, Y. Y. Li, S.-T. Lee and Y. He, ACS Nano, 2015, 9(6), 5958–5967 CrossRef CAS PubMed.
- A. Paul, A. Jana, S. Karthik, M. Bera, Y. Zhao and N. D. Pradeep Singh, J. Mater. Chem. B, 2016, 4, 521–528 RSC.
- R. Ban, F. Zheng and J. Zhang, Anal. Methods, 2015, 7(5), 1732–1737 RSC.
- J. Lin and Q. Wang, RSC Adv., 2015, 5(35), 27458–27463 RSC.
- N. Chen, Y. He, Y. Su, X. Li, Q. Huang, H. Wang, X. Zhang, R. Tai and C. Fan, Biomaterials, 2012, 33(5), 1238–1244 CrossRef CAS PubMed.
- A. Shiohara, S. Hanada, S. Prabakar, K. Fujioka, T. H. Lim, K. Yamamoto, P. T. Northcote and R. D. Tilley, J. Am. Chem. Soc., 2010, 132(1), 248–253 CrossRef CAS PubMed.
- T. Matsumoto, M. Maeda, J. Furukawa, W. B. Kim and H. Kobayashi, J. Nanopart. Res., 2014, 16(3), 1–7 CrossRef.
- J. Ji, G. Wang, X. You and X. Xu, Nanoscale Res. Lett., 2014, 9(1), 1–7 CrossRef PubMed.
- D. Jurbergs, E. Rogojina, L. Mangolini and U. Kortshagen, Appl. Phys. Lett., 2006, 88(23), 233116 CrossRef.
- B. F. P. Mcvey and R. D. Tilley, Acc. Chem. Res., 2014, 47(10), 3045–3051 CrossRef CAS PubMed.
- Y. He, Y. Y. Su, X. B. Yang, Z. H. Kang, T. T. Xu, R. Q. Zhang, C. H. Fan and S. T. Lee, J. Am. Chem. Soc., 2009, 131, 4434–4438 CrossRef CAS PubMed.
- Y. L. Zhong, P. Fei, X. P. Wei, Y. F. Zhou, J. Wang, X. X. Jiang, Y. Y. Su, S. Su, S.-T. Lee and Y. He, Angew. Chem., 2012, 51(34), 8485–8489 CrossRef CAS PubMed.
- F. Erogbogbo, K. T. Yong, I. Roy, R. Hu, W. C. Law, W. W. Zhao, H. Ding, F. Wu, R. Kumar, M. Swihart and P. N. Prasad, ACS Nano, 2011, 5, 413–423 CrossRef CAS PubMed.
- Q. Li, Y. He, J. Chang, L. Wang, H. Z. Chen, Y. W. Tan, H. Y. Wang and Z. Z. Shao, J. Am. Chem. Soc., 2013, 135(40), 14924–14927 CrossRef CAS PubMed.
- J. A. Kelly and J. G. Veinot, ACS Nano, 2010, 4(8), 4645–4656 CrossRef CAS PubMed.
- M. Dasog, Z. Y. Yang, S. Regli, T. M. Atkins, A. Faramus, M. P. Singh, E. Muthuswamy, S. M. Kauzlarich, R. D. Tilley and J. G. C. Veinot, ACS Nano, 2013, 7(3), 2676–2685 CrossRef CAS PubMed.
- X. Cheng, E. Hinde, D. M. Owen, S. B. Lowe, P. J. Reece, K. Gaus and J. J. Gooding, Adv. Mater., 2015, 27(40), 6144–6150 CrossRef CAS PubMed.
- T. M. Atkins, T. Arthur, D. S. Larsen, S. Dey, N. D. Browning and S. M. Kauzlarich, J. Am. Chem. Soc., 2011, 133(51), 20664–20667 CrossRef CAS PubMed.
- J. Wu, J. Dai, Y. Shao and Y. Sun, RSC Adv., 2015, 5(102), 83581–83587 RSC.
- B. Liao, W. Wang, X. Deng, B. He, W. Zeng, Z. Tang and Q. Liu, RSC Adv., 2016, 6, 14465–14467 RSC.
- Y. Feng, Y. Liu, C. Su, X. Ji and Z. He, Sens. Actuators, B, 2014, 203(21), 795–801 CrossRef CAS.
- J. Zhang and S. H. Yu, Nanoscale, 2014, 6(8), 4096–4101 RSC.
- Y. Guo, L. Zhang, F. Cao, L. Mang, X. Lei, S. Cheng and J. Song, Anal. Methods, 2016, 8, 2723–2728 RSC.
- B. B. Campos, M. Algarra, B. Alonso, C. M. Casado, J. Jiménez-Jiménez, E. Rodríguez-Castellón and J. C. G. Esteves da Silva, Talanta, 2015, 144, 862–867 CrossRef CAS PubMed.
- J. Lin and Q. Wang, RSC Adv., 2015, 5(35), 27458–27463 RSC.
- C. Liu, C. Shi, H. Li, W. Du, Z. Li, L. Wei and M. Yu, Sens. Actuators, B, 2015, 219, 185–191 CrossRef CAS.
- A. Afkhami and H. Bagheri, Microchim. Acta, 2012, 176(1–2), 217–227 CrossRef CAS.
- T. D. Román, M. A. Alonso-Lomillo and O. Domínguez-Renedo, Talanta, 2011, 86, 324–328 CrossRef PubMed.
- Z. Y. Lv, L. P. Mei and W. Y. Chen, Sens. Actuators, B, 2014, 201(201), 92–99 CrossRef CAS.
- B. Deng, L. Yang, H. Yin, X. Ning and H. Lu, Talanta, 2012, 91(6), 128–133 CrossRef CAS PubMed.
- H. Wang, J. Ding, X. Du, X. Sun and L. Chen, Food Chem., 2012, 131(1), 380–385 CrossRef CAS.
- B. Dong, X. Song, Y. Tang and W. Lin, Sens. Actuators, B, 2016, 222, 325–330 CrossRef CAS.
- Q. Ma, H. Cui and X. Su, Biosens. Bioelectron., 2009, 25(4), 839–844 CrossRef CAS PubMed.
- F. G. Wu, X. Zhang, S. Kai, M. Zhang, H. Y. Wang, J. N. Myers, Y. Weng, P. Liu, N. Gu and Z. Chen, Adv. Mater. Interfaces, 2015, 2(16), 1–11 Search PubMed.
- S. B. Rizvi, L. Yildirimer, S. Ghaderi, B. Ramesh, A. M. Seifalian and M. Keshtgar, Int. J. Nanomed., 2012, 7, 3915–3927 Search PubMed.
- J. K. Premachandra, W. J. Van Ooij and J. E. Mark, Reaction kinetics of y – ureidopropyltrimethoxysilane in the water–methanol system studied by FTIR spectroscopy, J. Adhes. Sci. Technol., 1998, 12(12), 1361–1376 CrossRef CAS.
- Y. Zhong, F. Peng, F. Bao, S. Wang, X. Ji, L. Yang, Y. Su, S. Lee and Y. He, Large-scale aqueous synthesis of fluorescent and biocompatible silicon nanoparticles and their use as highly photostable biological probes, J. Am. Chem. Soc., 2013, 135(22), 8350–8356 CrossRef CAS PubMed.
- P. Wahed, M. A. Razzaq and S. Dharmapuri, Food Chem., 2016, 202, 476–483 CrossRef CAS PubMed.
- S. Antwi-Boampong and J. J. Belbruno, Sens. Actuators, B, 2013, 182(6), 300–306 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra24654k |
| This journal is © The Royal Society of Chemistry 2016 |














