An electrochemical glycan biosensor based on a thionine-bridged multiwalled carbon nanotube/gold nanoparticle composite-modified electrode
Xinai Zhang*a,
Chenyong Huanga,
Yuxiang Jianga,
Jianzhong Shena,
Ping Gengb,
Wen Zhang*b and
Qilin Huangc
aSchool of Food and Biological Engineering, Jiangsu University, Zhenjiang, 212013, Jiangsu, China. E-mail: zhangxinai@mail.ujs.edu.cn
bDepartment of Chemistry, East China Normal University, Shanghai, 200062, China
cChemical Department, YuXi Normal University, Yuxi 653100, Yunnan, China
First published on 25th November 2016
Abstract
Glycans that attached to glycoproteins on cell surfaces are known to be functionally important and they have potential diagnostic values. The development of effective methods to analyze glycan expression on living cell surface is desirable for understanding their functions in disease progression and providing a powerful tool for early diagnosis. In this paper, thionine-bridged multiwalled carbon nanotube/gold nanoparticle (MWCNT/Th/AuNP) as a mediator/nanomaterial composite was synthesized to develop an electrochemical biosensor for glycan assay on living cancer cells. The MWCNT/Th/AuNP composite was fabricated by binding AuNPs to the surface of Th-coated MWCNTs, in which thionine acted as a linker to enable the negatively charged AuNPs to bind to the anionic MWCNT surface. When compared with conventional nanomaterial-based electrochemical biosensors, the MWCNT/Th/AuNP-modified electrode contained thionine as an electron mediator and simplified the electrochemical process based on enzymatic catalysis for signal amplification. Under a sandwich-type assay format, the MWCNT/Th/AuNP-based biosensor facilitated highly sensitive and specific detection of mannose on human liver and prostate cancer cells and offered great promise for the analysis of other glycans on living cells by utilizing a greater variety of lectins.
Introduction
Glycans that exist on glycoproteins play important roles in various biological processes such as cell growth and differentiation, cell–cell interactions, immune response modulation, and protein folding.1–5 Importantly, alterations of glycan expression levels have often been associated with development, progression and metastasis of cancer. Therefore, glycans on cell surfaces are considered to have potential as therapeutic targets or clinical biomarkers for diagnosis of various malignant diseases.6–8 Approved conventional methods for glycan detection include mass spectrometry, nuclear magnetic resonance and the detectors used in conjunction with chromatography.9,10 Although these methods can reveal molecular details, they are not amenable to evaluate glycans on living cells because of the associated destructive sample preparation. Recently, lectin-based electrochemical biosensors, taking the advantage of electrochemical detector and specific recognition between lectins and glycans, offer an alternative nondestructive tool to analyze glycans on living cell surfaces.11–13 However, it remains a great challenge to electrochemically assay cell surface glycans due to the lack of electroactive groups in glycans.With the aim of developing a successful electrochemical biosensor, considerable efforts have been made to improve the biosensor performance based on labeling technologies.14–16 The enzyme-based electrochemical biosensors are among the most popular strategies for easy labeling and signal amplification capability of enzymes.17,18 In the case of the biosensor development, a key factor is to provide a good pathway of electron transfer between the redox center of enzyme and electrode surface.19 Recently, the application of redox mediators (e.g., Prussian blue, thionine and ferrocene) to electrode modification has attracted increasing attention due to their special structural and electrochemical properties. For example, an electrochemical immunosensor was prepared to detect tumor markers using the immobilized Prussian blue as a mediator to catalyze the reduction of H2O2 produced in the enzymatic cycle.20 Similarly, a three-dimensional architecture was fabricated to design an electrochemical cytosensor based on the enzymatic catalytic reaction of horseradish peroxidase (HRP) toward the oxidation of thionine by H2O2.21 A 6-ferrocenylhexanethiol self-assembled multiwalled carbon nanotube–Au nanoparticle composite was synthesized to modify an electrode for the sensitive and selective assay of ascorbic acid.22 Compared with a mediator-modified electrode, the mediator/nanomaterial composite-modified electrode demonstrated an increased electrochemical stability and provided high sensitivity due to the excellent electrocatalytic effects of nanomaterials.23–26 Therefore, the mediator/nanomaterial composite was found to be suitable for constructing a biosensor based on the mediator-mediated electron transfer process.
In this work, a thionine-bridged multiwalled carbon nanotube/gold nanoparticle (MWCNT/Th/AuNP) composite was designed for glycan assay on living cancer cells based on enzymatic catalysis for signal amplification. Here, mannose on cancer cells derived from human liver and prostate was used as a model glycan. Concanavalin A (Con A), a mannose-specific lectin with four binding sites for sugar epitopes,27 was used to construct the biosensor for gaining insight into cell surface mannose. In order to develop the lectin-based biosensor, MWCNT/Th/AuNP composite was initially fabricated by binding AuNPs to the surface of Th-coated MWCNTs, in which thionine acted as interlinker to enable the negatively charged AuNPs to bind to the anionic MWCNT surface. Owing to the good biocompatibility of AuNPs and the excellent conductivity of MWCNTs, a composite-modified glassy carbon electrode (GCE) not only provided an effective biosensing platform for loading Con A with high stability and bioactivity, but also played an important role in signal amplification. Under a sandwich-type format, analytical procedure involves the specific recognition between cell surface mannose and the biosensor surface-confined Con A, followed by the attachment of HRP labeled Con A (Con A-HRP). The electrochemical signals derived from the bound HRP toward the reduction of H2O2 using the immobilized thionine as electron mediator for further signal amplification. Under optimal conditions, the MWCNT/Th/AuNP-based biosensor exhibited advantageous performances for the quantification of cancer cells with broad dynamic ranges and low detection limits of 20 cells per mL for QGY-7701 (liver cancer cells) and 35 cells per mL for LNCaP (prostate cancer cells). The proposed method was also used to evaluate the average amount of mannose on each cell surface corresponding to 3.39 × 1010 molecules for QGY-7701 and 1.84 × 1010 molecules for LNCaP. Compared with the reported methods in the literatures,6,11,12 the lectin-based biosensor gave a useful protocol for glycan assay on cell surface with high sensitivity, acceptable accuracy and satisfying specificity, and may greatly facilitate the medical diagnosis and treatment in early process of cancer.
Experimental
Materials and reagents
Concanavalin A (Con A), HRP labeled Con A (Con A-HRP) and bovine serum albumin (BSA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). MWCNTs (φ = 10–30 nm) were purchased from Nanotech Port Co., Ltd. (Shenzhen, China). Mannose was from Aladdin Reagent Database Inc. (Shanghai, China). HAuCl4, thionine and other chemical reagents were purchased from Chemical Reagents Co., Ltd. (Shanghai, China). All solutions were prepared with MilliQ water (18 MΩ cm) from a Millipore system.Cell culture and treatment
Human liver cancer cells (QGY-7701) and prostate cancer cells (LNCaP) were obtained from Cell Bank of Chinese Academy of Sciences (Shanghai, China). QGY-7701 and LNCaP cells were grown in Roswell Park Memorial Institute-1640 medium with 10% fetal bovine serum at 37 °C in a humidified atmosphere of 5% CO2. All cell culture reagents were from Sigma-Aldrich (St. Louis, MO, U.S.A.). At the logarithmic growth phase, cells were trypsinized and washed twice with a phosphate buffered saline (PBS; 0.01 M, pH 7.4) by centrifugation at 1000 rpm for 10 min. The sediment was dispersed in a pH 7.4 tris–HCl buffer containing 1.0 mM Mn2+, 1.0 mM Ca2+ and 0.10 M Na+ to obtain a homogeneous cell suspension.Apparatus
Electrochemical measurements were performed with a CHI 660C electrochemical workstation (CH Instruments Inc., Austin, TX, USA). A conventional three-electrode configuration was used all through the experiment, which included a glassy carbon electrode (GCE) as a working electrode, a platinum wire auxiliary electrode, and a saturated calomel reference electrode (SCE). Cyclic voltammetry (CV) was carried out at a scan rate of 100 mV s−1, and differential pulse voltammetry (DPV) at a pulse amplitude of 50 mV and a pulse width of 50 ms. Transmission electron microscopy (TEM) was conducted on a JEM-2100 microscope (JEOL Ltd., Tokyo, Japan). Fourier transform infrared (FT-IR) spectra were recorded on a Nicolet Nexus 670 instrument (Thermo Nicolet Co., USA) in KBr pellet form. Zeta potentials were measured by dynamic light scattering (Zetasizer Nano ZS, Malvern Instruments, UK). Energy dispersive X-ray (EDX) analyses were carried out on a Hitachi S-4800 analyser (Hitachi Co. Ltd, Tokyo, Japan). Fluorescence images were collected by fluorescence microscope (Leica DM4000B, Leica Microsystems, Nussloch, Germany).Synthesis of AuNPs
AuNPs were prepared by the reduction of HAuCl4 with trisodium citrate.6 Briefly, a volume of 250 mL of 0.01% HAuCl4 in pure water (Milli-Q water) was brought to a boil with vigorous stirring. To this solution was added 3.75 mL of 1% trisodium citrate, which resulted in a change of color from pale yellow to deep purple within 1–2 min. Boiling was pursued for 10 min, and then the solution was allowed to cool to room temperature.Preparation of MWCNT/Th/AuNP composite
Briefly, MWCNTs were first carboxyl-functionalized and shortened by sonication in H2SO4/HNO3 (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) at 70 °C for 5 h.28,29 The dispersion was filtered and washed repeatedly with water until the filtrate was neutral. Then the pH of the functionalized MWCNTs was adjusted to ∼8.0 to achieve net negatively charged carboxylate anions. The resulting MWCNTs were centrifuged at 14
1 v/v) at 70 °C for 5 h.28,29 The dispersion was filtered and washed repeatedly with water until the filtrate was neutral. Then the pH of the functionalized MWCNTs was adjusted to ∼8.0 to achieve net negatively charged carboxylate anions. The resulting MWCNTs were centrifuged at 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 15 min to remove the supernatant and dried under vacuum overnight. Subsequently, 1.5 mg MWCNTs were re-dispersed in 1.0 mL water with sonication. This dispersion was mixed with 1.0 mL of thionine solution (3.0 mg mL−1) and sonicated at room temperature for 12 h.25,30 The purple color of thionine solution disappeared and a black solution was obtained. Then, 0.20 mL Th-coated MWCNTs solution was added dropwise into 20 mL as-prepared gold colloid to prepare the MWCNT/Th/AuNP composite under vigorous stirring. Immediately, the pink color faded and then changed slowly into gray. After stirring for an additional 3 h, the MWCNT/Th/AuNP composite was isolated by centrifugation at 10
000 rpm for 15 min to remove the supernatant and dried under vacuum overnight. Subsequently, 1.5 mg MWCNTs were re-dispersed in 1.0 mL water with sonication. This dispersion was mixed with 1.0 mL of thionine solution (3.0 mg mL−1) and sonicated at room temperature for 12 h.25,30 The purple color of thionine solution disappeared and a black solution was obtained. Then, 0.20 mL Th-coated MWCNTs solution was added dropwise into 20 mL as-prepared gold colloid to prepare the MWCNT/Th/AuNP composite under vigorous stirring. Immediately, the pink color faded and then changed slowly into gray. After stirring for an additional 3 h, the MWCNT/Th/AuNP composite was isolated by centrifugation at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min and washed twice with water. After that, the obtained MWCNT/Th/AuNP was redispersed in 4.0 mL PBS.
000 rpm for 5 min and washed twice with water. After that, the obtained MWCNT/Th/AuNP was redispersed in 4.0 mL PBS.
Preparation of the Con A-based biosensor
GCE (Φ = 3 mm) was used to prepare the Con A-based biosensor. Prior to surface modification, the electrode was polished to a mirror using 0.05 μm alumina on a polishing micro-cloth followed by rinsing with water. After successive sonication in acetone, HNO3 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v), NaOH (50%, w/w) and water, the cleaned GCE surface was modified by casting 10 μL the MWCNT/Th/AuNP composite in PBS and allowed to dry at room temperature. Subsequently, 10 μL Con A (0.10 mg mL−1) was introduced on the MWCNT/Th/AuNP-modified GCE for 60 min to prepare the Con A-based biosensor. The biosensor was treated with 1.0% BSA/PBS for 30 min to block any nonspecific binding.
1, v/v), NaOH (50%, w/w) and water, the cleaned GCE surface was modified by casting 10 μL the MWCNT/Th/AuNP composite in PBS and allowed to dry at room temperature. Subsequently, 10 μL Con A (0.10 mg mL−1) was introduced on the MWCNT/Th/AuNP-modified GCE for 60 min to prepare the Con A-based biosensor. The biosensor was treated with 1.0% BSA/PBS for 30 min to block any nonspecific binding.
Electrochemical detection
After being rinsed and dried with nitrogen, the Con A-based biosensor was incubated with cell suspension or mannose standards at a certain concentration for 60 min at room temperature, and then washed with tris–HCl buffer. Subsequently, 10 μL Con A-HRP (0.10 mg mL−1) was applied on the cell-captured electrode and incubated for 60 min. The resulting electrode was rinsed with thoroughly with PBS, and then immersed in pH 7.0 HAc–NaAc buffer containing 1.0 mM H2O2 to perform DPV measurements from 0 to −0.60 V.Results and discussion
Preparation and characterization of MWCNT/Th/AuNP composite
The MWCNT/Th/AuNP composite was designed by utilizing thionine as interlinkers, in which thionine enabled the negatively charged AuNPs to bind to the anionic MWCNT surface. The TEM image of the MWCNT/Th/AuNP composite is shown in Fig. 1A and the inset.As can be seen, AuNPs (dark spots) appeared as small clusters on MWCNTs, but these clusters were well separated. Importantly, no free AuNPs were observed in the background of the TEM image, indicating strong interactions between MWCNTs and AuNPs. Fig. 1B shows the TEM image of MWCNT/AuNP without adsorbed thionine interlinker. As can be seen, the density of adsorbed AuNPs (dark spots) along MWCNTs was lower than that on Th-coated MWCNTs. Additionally, there were free AuNPs distributed on the background of the TEM image. The control result supports the proposed mechanism of thionine-mediated adsorption of AuNPs on MWCNT surface.
FT-IR spectroscopy was used to study the interaction between MWCNTs and thionine. As shown in Fig. 1C, there was no obvious difference between FT-IR spectra of thionine (curve a) and that of MWCNT/Th (curve b). The absence of amide C–N stretching vibrations in the 1000–1500 cm−1 range indicates that no covalent bonds were formed between MWCNTs and thionine,26 thus indicating that strong interactions arose from π–π stacking between these conjugated frames.
Moreover, EDX analysis was also used to characterize the MWCNT/Th/AuNP composite by illuminating electron beams on the composite (Fig. 1D). The EDX spectrum revealed the existence of C, N, O, S and Au elements. Fig. 1E shows the experimental values of zeta potential measured from aqueous solution of the various materials. The anionic MWCNTs exhibited a zeta potential of near −13 mV, while the zeta potential of MWCNT/Th increased to +5.5 mV due to the binding of positively charged thionine. Upon adsorption of negatively charged AuNPs, the zeta potential of MWCNT/Th/AuNP composite decreased to −32 mV. The results further confirmed the formation of thionine and AuNPs on the MWCNTs.
Cyclic voltammetric behavior of the modified electrode
Cyclic voltammetry (CV) of 5 mM [Fe(CN)6]3− and 5 mM [Fe(CN)6]4− on different modified electrodes (GCE/MWCNT, GCE/MWCNT/Th and GCE/MWCNT/Th/AuNP) was conducted in 10 mM PBS (pH 7.4) (Fig. 2). Curve a shows the reduction of [Fe(CN)6]3− at 0.411 V and the oxidation of [Fe(CN)6]4− at 0.529 V on GCE/MWCNT. When compared to those obtained from GCE/MWCNT, the oxidation peak of [Fe(CN)6]4− on GCE/MWCNT/Th has increased by 17.27% and the reduction peak of [Fe(CN)6]3− has increased by 15.18% (curve b). In addition, a new oxidation peak at 0.29 V and a new reduction peak at −0.15 V appeared, which were attributed to the redox behavior of thionine. When compared to those shown in curve b, the reduction peak of [Fe(CN)6]3− has further increased by 22.14% on GCE/MWCNT/Th/AuNP and the oxidation peak of [Fe(CN)6]4− has increased by 24.03% (curve c). Similarly, the reduction peak of thionine and the oxidation peak of thionine have also increased by 40.43% and 66.72%, respectively. Furthermore, the peak potential on GCE/MWCNT/Th/AuNP moved towards a negative direction, indicating an increased ease in electron transfer. | ||
| Fig. 2 Cyclic voltammograms (CVs) on (a) GCE/MWCNT, (b) GCE/MWCNT/Th, and (c) GCE/MWCNT/Th/AuNP of 5 mM [Fe(CN)6]3−/4− in 10 mM PBS (pH 7.4). | ||
Evaluation of the analytical performance of the electrochemical biosensor
To evaluate the analytical performance of the biosensor, a comparative study of the electrocatalytic peak current was carried out by exploiting GCE/AuNP, GCE/MWCNT/AuNP and GCE/MWCNT/Th/AuNP as biosensing platform, respectively. Fig. 3 shows the peak current measured at different biosensors for electrochemical detection of QGY-7701 and LNCaP cells (5.0 × 106 cells per mL). Compared with GCE/AuNP and GCE/MWCNT/AuNP, the MWCNT/Th/AuNP-modified GCE exhibited the highest response. Some possible explanations may be contributed to these observations: firstly, the large surface area and excellent conductivity of MWCNT/Th/AuNP composite made them possible to enhance electrochemical detection capability; secondly, thionine in the MWCNT/Th/AuNP composite acted as a good mediator, which could effectively shuttle electrons from the electrode surface to the redox center of HRP. The results obviously suggested that the use of GCE/MWCNT/Th/AuNP for biosensor preparation is expected to improve the detection limits and reduce the risk of false negative determination at low cell concentrations.Effects of preparation conditions of MWCNT/Th/AuNP composite on current response
The MWCNT/Th/AuNP composite was used as a biosensing platform, and thus its preparation process may have an effect on the electrochemical signal generated. We investigated the dependence of the electrocatalytic peak current on different variables in the biosensor design for the detection of QGY-7701 cells (5.0 × 106 cells per mL), including the concentration of MWCNTs, the concentration of thionine, and the incubation time of AuNPs. With the increasing concentration of MWCNTs, the peak current (Δip) increased and tended to a steady value after 1.5 mg mL−1. Therefore, 1.5 mg mL−1 was used for the composite preparation (Fig. 4A). As shown in Fig. 4B, an obvious increase of the Δip was observed with the increasing thionine concentration from 0.50 to 3.0 mg mL−1, while the Δip decreased slightly at a greater concentration. Thus, the optimal concentration of thionine was 3.0 mg mL−1. The amount of AuNPs bound to MWCNT/Th was an important parameter for improving the conductivity of the sensing interface and the immobilization of Con A. Fig. 4C displays the influence of the incubation time for the attachment of AuNPs to MWCNT/Th on the Δip. With the increasing incubation time, the Δip increased and then leveled off after 180 min. A longer incubation time did not enhance the Δip. Therefore, 180 min was chosen for the incubation of AuNPs. Under these conditions, the MWCNT/Th/AuNP-modified GCE could provide a suitable interface with good conductivity and biocompatibility, thus amplifying the electrochemical signal.Effects of experimental conditions on current response
The electrochemical signal derived from the catalytic reaction of HRP in the bound Con A-HRP on the biosensor, and thus the incubation conditions greatly influenced the sensitivity of the proposed strategy. Fig. 4D shows the dependence of the Δip upon the Con A-HRP concentration in the second incubation step. After the Con A-based biosensor was incubated with QGY-7701 cells at the concentration of 5.0 × 106 cells per mL, the Δip at the GCE/MWCNT/Th/AuNP/Con A/cell/Con A-HRP increased with the increasing concentration of Con A-HRP and then tended to level off after 0.10 mg mL−1. Moreover, the effect of incubation time was also investigated in the presence of QGY-7701 cells (5.0 × 106 cells per mL). As shown in Fig. 4E, with increasing incubation time, the Δip increased and then reached a constant value when the incubation time was longer than 60 min. Therefore, 0.10 mg mL−1 Con A-HRP and 60 min incubation time were selected as the optimal conditions.The electrochemical performance of the enzyme-catalyzed analysis was related to the concentration of H2O2 in the measuring Δip. As can be seen, the Δip of the GCE/MWCNT/Th/AuNP/Con A/cell/Con A-HRP increased when the H2O2 concentration was in the range from 0.20 to 1.0 mM, and then it tended to a steady value. Thus, 1.0 mM H2O2 was selected for the enzyme-amplified detection.
Electrochemical detection of cancer cells with the MWCNT/Th/AuNP-based biosensor
Considering the critical need for the sensitive detection of cancer cells,31,32 the developed biosensor was used for the assay of QGY-7701 and LNCaP cells. The enzymatic catalytic signal was related to the cells attached to the electrode surface and the obtained results are shown in Fig. 5. The respective DPV peak current for the analysis of QGY-7701 (Fig. 5A) and LNCaP cells (Fig. 5C) were obtained by using the MWCNT/Th/AuNP-based biosensor under optimal conditions. A linear relationship between the Δip and the logarithm of cell concentration was found in the range from 50 to 5.0 × 106 cells per mL for QGY-7701 cells with a detection limit of 20 cells per mL (Fig. 5B) and 1.0 × 102 to 1.0 × 107 cells per mL for LNCaP cells with a detection limit of 35 cells per mL (Fig. 5D), respectively.The reproducibility of the electrochemical biosensor was evaluated from the peak current of the MWCNT/Th/AuNP/Con A-modified electrode. Six electrodes modified identically were explored in the solution containing the same concentration of cancer cells. The relative standard deviation (RSD) was 5.2%, indicating that the reproducibility of the proposed biosensor was acceptable.
When the GCE/MWCNT/Th/AuNP/Con A-modified biosensor was stored in the refrigerator at 4 °C, the peak current was still retained at 90.3% value of the initial response after a storage period of two weeks. The long-term stability might be attributed to the strong interactions between the MWCNT/Th/AuNP and Con A.
The regeneration of the Con A-based biosensor was performed by rinsing with 0.10 M glycine-hydrochloric acid (Gly-HCl, pH 2.8) to dissociate the cell–Con A complex. After each sandwich assay, the electrode was treated with Gly-HCl for 10 min to remove cancer cells and Con A-HRP. The as-renewed biosensor could restore 87.2% of the initial value after six assay runs, indicating acceptable reusability.
Moreover, the performance of the electrochemical biosensor has been compared with those cell sensors reported by Zhang et al., 2010,6 2015,11 2011 (ref. 12) and Gu et al., 2009.33 Characteristics such as the linear range and detection limit are summarized for all of them in Table 1. As can be observed, the proposed biosensor exhibited good performance for the detection of cancer cells with broad linear ranges and low detection limits.
| Sensor platform | Cells | Linear range [cells per mL] | LOD [cells per mL] | Ref. |
|---|---|---|---|---|
| AuNP/MWNT/GCE | A549 | 3.0 × 104 to 3.0 × 107 | 7000 | 6 |
| AuNP/MWCNT/GCE | QGY-7703 | 1.0 × 102 to 1.0 × 106 | 40 | 11 |
| Au-GSH/PDCNT/GCE | 95-D | 1.7 × 103 to 1.5 × 108 | 580 | 12 |
| PS/PANI/AuNPs/GCE | HL-60 | 1.6 × 103 to 1.6 × 108 | 730 | 33 |
| MWCNT/Th/AuNP/GCE | QGY-7701 | 50–5.0 × 106 | 20 | This report |
| MWCNT/Th/AuNP/GCE | LNCaP | 1.0 × 102 to 1.0 × 107 | 35 | This report |
Evaluation of mannose on cancer cell surface
Cell surface mannose is closely related to the biological processes such as tumor growth and metastasis, and the analysis of mannose expression was critical for understanding its role in cancer development. In view of the above challenges, a series of mannose standard samples was detected by the electrochemical biosensor and the results obtained were used to construct a calibration plot. As shown in Fig. 6A, the Δip exhibited a linear relationship with mannose concentration (cmannose, μM) in the range from 0.015–0.045 μM (R2 = 0.994).The linear regression equation is
| Δip(1 × 10−5 A) = 4.73cmannose + 0.71 | (1) |
Under the same experimental conditions, the Con A-based biosensor was used for the detection of QGY-7701 cells at different concentrations. The peak current (Δi′p) versus QGY-7701 concentration (cQGY-7701, cells per mL) showed linear relation in the ranges of 4 × 102 to 8 × 102 cells per mL (R2 = 0.992) (Fig. 6B), and the linear regression equation is:
| Δi′p(1 × 10−5 A) = 2.29 × 10−4cQGY-7701 + 0.73 | (2) |
When the Δip (resulted from mannose standard detection) was equal to Δi′p (resulted from QGY-7701 detection), the mannose concentration (cmannose) in the cell extract could be calculated according to the following equation:
 | (3) |
Meanwhile, with the use of eqn (1), the mannose number (nmannose) on the surface of one QGY-7701 cell could be calculated according to the following equation:
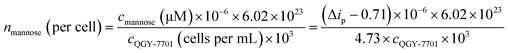 | (4) |
Five parallel measurements gave the average amount of mannose on single cell surface to be 3.39 × 1010 molecules for QGY-7701. Using the same method, the amount of mannose on each LNCaP cell was also evaluated to be 1.84 × 1010 molecules (Fig. 6C). Fluorescent analysis was also used to detect mannose expression on QGY-7701 and LNCaP cells by staining with fluorescein Con A for the bioimaging experiment. The results indicated that the mannose expression on QGY-7701 cells (the inset in Fig. 6B) was more abundant than that on LNCaP cells (the inset in Fig. 6C), which were in agreement with those obtained from the proposed methodology.
In order to investigate the specific binding affinities between mannose and Con A, the biosensor was used to detect QGY-7701 cells (at a concentration of 6 × 102 cells per mL, containing 0.03328 μM mannose expression) and a mannose sample (0.03328 μM mannose) under the same experimental condition (Fig. 7). The results demonstrated that the peak current obtained from cell detection was in accordance with that obtained from mannose standard detection, indicating that the other biological molecules such as amino acids and proteins on living cells have no interference with the assay of cell surface mannose.
Conclusions
MWCNT/Th/AuNP composite was designed to construct the electrochemical biosensor for mannose assay on living cancer cells. The MWCNT/Th/AuNP composite not only provided an effective biosensing platform for Con A loading, but also played an important role in sensitivity enhancement. With the dual signal amplification of the MWCNT/Th/AuNP composite and enzymatic catalysis, the developed biosensor was successfully used for analysis of cancer cells and also for evaluation of the average amount of mannose on single cell surface. When compared with the conventional nanomaterial-based electrochemical biosensors, the composite-modified electrode contained thionine as electron mediator and simplified the electrochemical process. Overall, the MWCNT/Th/AuNP-based biosensor provides a useful tool for mannose assay and possesses promising application for the assay of other glycans on living cells with the selection of more lectins.Acknowledgements
This work was supported by the National Natural Science Foundation of China (21205051, 21375040), Key projects of Yunnan Provincial Department of Education (2013Z073), General project of Yunnan Provincial Department of Education (2015Y358), and Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (1033000006).References
- A. Varki, Nature, 2007, 446, 1023 CrossRef CAS PubMed.
- W. Cheng, L. Ding, S. Ding, Y. Yin and H. Ju, Angew. Chem., Int. Ed., 2009, 48, 6465 CrossRef CAS PubMed.
- K. Ohtsubo and J. D. Marth, Cell, 2006, 126, 855 CrossRef CAS PubMed.
- A. Matsumoto, N. Sato, K. Kataoka and Y. Miyahara, J. Am. Chem. Soc., 2009, 131, 12022 CrossRef CAS PubMed.
- Y. Tian, Y. Zhou, S. Elott, R. Aebersold and H. Zhang, Nat. Protoc., 2007, 2, 334 CrossRef CAS PubMed.
- X. Zhang, Y. Teng, Y. Fu, L. Xu, S. Zhang, B. He, C. Wang and W. Zhang, Anal. Chem., 2010, 82, 9455 CrossRef CAS PubMed.
- L. I. Ang, T. C. W. Poon, P. B. Lai, A. T. Chan, S.-M. Ngai, A. Y. Hui, P. J. Johnson and J. J. Sung, J. Proteome Res., 2006, 5, 2691 CrossRef PubMed.
- Y. Kaneko, F. Nimmerjahn and J. V. Ravetch, Science, 2006, 313, 670 CrossRef CAS PubMed.
- K. Kubota, Y. Sato, Y. Suzuki, N. Goto-Inoue, T. Toda, M. Suzuki, S. Hisanaga, A. Suzuki and T. Endo, Anal. Chem., 2008, 80, 3693 CrossRef CAS PubMed.
- K. T. Pilobello and L. K. Mahal, Curr. Opin. Chem. Biol., 2007, 11, 300 CrossRef CAS PubMed.
- X. Zhang, W. Lu, J. Shen, Y. Jiang, E. Han, X. Dong and J. Huang, Biosens. Bioelectron., 2015, 74, 291 CrossRef CAS PubMed.
- X. Zhang, Y. Teng, Y. Fu, S. Zhang, T. Wang, C. Wang, L. Jin and W. Zhang, Chem. Sci., 2011, 2, 2353 RSC.
- Y. Hu, P. Zuo and B.-C. Ye, Biosens. Bioelectron., 2013, 43, 79 CrossRef CAS PubMed.
- X. Zhang, F. Zhang, H. Zhang, J. Shen, E. Han and X. Dong, Talanta, 2015, 132, 600 CrossRef CAS PubMed.
- J. Narayanan, M. K. Sharma, S. Ponmariappan, Sarita, M. Shaik and S. Upadhyay, Biosens. Bioelectron., 2015, 69, 249 CrossRef CAS PubMed.
- A. Singh, S. Park and H. Yang, Anal. Chem., 2013, 85, 4863 CrossRef CAS PubMed.
- X. Zhang, J. Shen, H. Ma, Y. Jiang, C. Huang, E. Han, B. Yao and Y. He, Biosens. Bioelectron., 2016, 80, 666 CrossRef CAS PubMed.
- Y. Zhao, L. Liu, D. Kong, H. Kuang, L. Wang and C. Xu, ACS Appl. Mater. Interfaces, 2014, 6, 21178 CAS.
- D. Tang, B. Su, J. Tang, J. Ren and G. Chen, Anal. Chem., 2010, 82, 1527 CrossRef CAS PubMed.
- G. Lai, F. Yan and H. Ju, Anal. Chem., 2009, 81, 9730 CrossRef CAS PubMed.
- J.-J. Zhang, F.-F. Cheng, T.-T. Zheng and J.-J. Zhu, Anal. Chem., 2010, 82, 3547 CrossRef CAS PubMed.
- J.-D. Qiu, R. Wang, R.-P. Liang and M. Xiong, Electroanalysis, 2008, 20, 1819 CrossRef CAS.
- E. Nossol and A. J. G. Zarbin, Adv. Funct. Mater., 2009, 19, 3980 CrossRef CAS.
- L. Deng, Y. Wang, L. Shang, D. Wen, F. Wang and S. Dong, Biosens. Bioelectron., 2008, 24, 951 CrossRef CAS PubMed.
- Z. Wang, M. Li, P. Su, Y. Zhang, Y. Shen, D. Han, A. Ivaska and L. Niu, Electrochem. Commun., 2008, 10, 306 CrossRef CAS.
- Z. Wang, M. Li, Y. Zhang, J. Yuan, Y. Shen, L. Niu and A. Ivaska, Carbon, 2007, 45, 2111 CrossRef CAS.
- J. K. Bhattarai, A. Sharma, K. Fujikawa, A. V. Demchenko and K. J. Stine, Carbohydr. Res., 2015, 405, 55 CrossRef CAS PubMed.
- R. Villalonga, P. Díez, M. Eguílaz, P. Martínez and J. M. Pingarrón, ACS Appl. Mater. Interfaces, 2012, 4, 4312 CAS.
- X. Xin, S. Sun, H. Li, M. Wang and R. Jia, Sens. Actuators, B, 2015, 209, 275 CrossRef CAS.
- H.-P. Peng, Y. Hu, A.-L. liu, W. Chen, X.-H. Lin and X.-B. Yu, J. Electroanal. Chem., 2014, 712, 89 CrossRef CAS.
- C. Ding, Y. Ge and S. Zhang, Chem.–Eur. J., 2010, 16, 10707 CrossRef CAS PubMed.
- M. J. Rutherford, S. R. Hinchffe, G. A. Abel, G. Lyratzopoulos, P. C. Lambert and D. C. Greenberg, Int. J. Cancer, 2013, 133, 2192 CrossRef CAS PubMed.
- M. Gu, J. Zhang, Y. Li, L. Jiang and J.-J. Zhu, Talanta, 2009, 80, 246 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |






