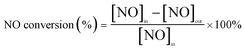Nonprecious mixed oxide catalysts Co3AlO and Co2NiAlO derived from nanoflowerlike cobalt-based hydrotalcites for highly efficient oxidation of nitric oxide†
Ting Fan,
Liguang Dou and
Hui Zhang*
State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, P. O. Box 98, Beijing 100029, China. E-mail: huizhang67@gst21.com; Fax: +86-10-6442-5385; Tel: +86-10-6442-5872
First published on 14th November 2016
Abstract
Nonprecious mixed oxide catalysts Co3AlO-T and Co2NiAlO-T (T = 500, 800) were obtained from thermal decomposition of nanoflowerlike Co3Al-HT and Co2NiAl-HT hydrotalcite-like precursors at 500 and 800 °C, which were synthesized by a facile co-precipitation method, and systematically characterized by combinational techniques. The mixed oxide catalysts mainly consisted of homogeneous and stable non-stoichiometric cobalt-based spinel phases Co(Co,Al)2O4 and Ni(Co,Al)2O4. The 500 °C calcined catalysts showed nanoflowerlike morphology, while the 800 °C calcined catalysts presented disorderly stacked states consisting of severely agglomerated irregular nanoparticles. The catalysts Co3AlO-500 and Co2NiAlO-500 presented excellent catalytic NO oxidation performance, with the maximum conversion efficiency ca. 88.8% and 87.6% (285 °C), respectively, which is much higher than those of Co3AlO-800 and Co2NiAlO-800 (55.6% and 66.6% at 350 °C). The excellent catalytic NO oxidation performance of Co3AlO-500 and Co2NiAlO-500 could be attributed to the much larger amount of surface active sites endowed by these catalysts with smaller crystallite sizes of cobalt-based spinel phases, higher specific surface areas, and mesoporous structure. The active sites and NO oxidation pathways of the present catalysts were tentatively proposed. Although Co2NiAlO-500 possessed a slightly smaller amount of active sites (Co3+ and Ni3+ associated with surface adsorbed oxygen), compared to Co3AlO-500 with active sites (Co3+ associated with surface adsorbed oxygen), catalyst Co2NiAlO-500 possessed higher reducibility, leading to their similar desorption amount of NO2, thus similar NO oxidation performance. Moreover, the lower cost of catalyst Co2NiAlO-500 gives it a greater potential for practical applications.
1. Introduction
Diesel and lean-burn gasoline engines with high air/fuel ratios have drawn much attention because of their higher fuel efficiency and lower CO2 emission than conventional gasoline engines.1–4 However, the nitrogen oxide (NOx) pollutants emitted from the lean-burn engines cannot be effectively removed by the traditional three-way catalysts due to the high oxygen content in the lean-burn exhaust, which can cause severe environmental problems such as photochemical smog, acid rain, and ozone layer depletion.1,5,6 In order to abate the NOx pollutants and meet the increasingly stringent NOx emission regulations, various techniques have been developed, among which the NOx storage and reduction (NSR),7–9 selective catalytic reduction (SCR),10–12 and continuously regenerating trap (CRT) for simultaneous NOx and soot removal2,13,14 are the main solutions. It is known that the NOx pollutants emitted from the lean-burn engines mainly consist of NO (∼90%), while NO2 is considered as an important intermediate species for NOx conversion in all these techniques.1,2 The oxidation of NO to NO2 is the first step in the NSR technology and NO2 is much easier to be absorbed by the storage materials than NO.7–9 The reaction rate of NH3-SCR can be greatly improved at low temperatures (200–300 °C) with equal amount of NO and NO2 in the exhaust during the so-called fast-SCR process, which is achieved by setting an oxidation catalyst at the upstream of the SCR unit.10–12 Furthermore, in the CRT technology, NO2 shows stronger oxidizability than O2, and can oxidize soot particles collected on a particulate filter for the diesel vehicles at lower temperature.2,13,14 Therefore, it is of great importance to develop the effective catalysts for oxidizing NO to NO2.Among the previously reported catalysts for NO oxidation, platinum based noble metal catalysts have been widely used due to their good performance but are limited by their high cost and poor thermal stability under lean-burn conditions.15,16 Transition metal oxide catalysts such as Co-related oxides are favorable alternatives to noble metal catalysts because of their low cost, high catalytic activity, and potential practicality.2,10,17–20 Ren et al.19 reported two Co3O4 nanowires-array catalysts, grown on ceramic cordierite honeycomb substrates using nitrate and acetate as cobalt sources followed 300 °C calcinations, showing the highest catalytic NO oxidation property with similar conversion efficiency as 80% at 275 °C. Wang et al.20 reported ordered mesoporous catalysts Co3O4, NiO and NiCo2O4, prepared by nanocasting method using mesoporous KIT-6 as a hard template. The catalyst NiCo2O4 showed the best low-temperature H2-SCR activity and the highest NO-to-NO2 conversion (∼60%, 350 °C), which was attributed to the higher surface adsorption oxygen concentration in NiCo2O4 than in Co3O4 and NiO, and the mesoporous structure of NiCo2O4 rendered it larger surface area and extra surface oxygen vacancies. However, the synthesis of the hierarchical Co3O4 nanowires-array catalysts needs the usage of ceramic cordierite honeycomb substrates and the hydrothermal condition. While the preparation of the ordered mesoporous catalyst NiCo2O4 uses the mesoporous KIT-6 template in the nanocasting method. Obviously, the preparation procedures of these Co-based oxide catalysts are complicated and time consuming, which may restrict the large scale synthesis of these catalysts in the practical applications.
Hydrotalcite-like compounds, also known as layered double hydroxides (LDHs) with the general formula [M1−x2+Mx3+(OH)2](Ax/nn−)·mH2O, are a class of 2D layered clay materials consisting of positively charged hydroxide layers with M2+ and M3+ metal ions arranged in a uniform and ordered manner and interlayer hydrated anions for charge balance.21–23 Hydrotalcite-like compounds can contain more than two types of highly dispersed metal cations and their morphology and particle sizes are tunable through different synthesis methods, and these unique structural characters can provide a good platform for the smart design of LDH-based NOx abatement catalysts.6,7,24–27 Yu et al.24,25 reported a series of Co-based oxide catalysts xCoMgAlO and xCaCoAlO obtained by thermal decomposition hydrotalcite precursors at 800 °C in air for the effective abatement of NOx in the NSR technology, while one demerit of these catalysts was the relatively low surface area (∼20 m2 g−1) due to the high calcination temperature. Li et al.14 reported a K-containing (4.5 wt%) CoMgAlO catalyst calcined at 600 °C, showing the best catalytic performance in soot combustion and simultaneous soot-NOx removal with the highest NOx reduction (by soot) percentage of 32% among a series of K-promoted CoMgAlO catalysts due to the high surface K/Co atomic ratio and the strong interaction between K and Co. Wang et al.26 reported a series of hydrotalcites derived oxide catalysts MgAlO, CoMgAlO, CuMgAlO, and CuCoMgAlO with large surface areas and highly dispersed redox components, exhibiting high performance in NOx storage and soot combustion, however, the NO oxidation activities of these catalysts were relatively low. Recently, our group27 reported a 3D oxide nanosheet array catalyst CoMgAlO-array with small-sized active Co3O4 species (5.7 nm) highly dispersed on a Mg/Al-oxide matrix and relatively larger surface area, showing much higher NOx storage capacity and catalytic soot combustion activity compared to traditional CoMgAlO catalyst. However, although the above hydrotalcite-like precursors derived oxide catalysts are reported for effective abatement of NOx in the NSR and simultaneous soot-NOx removal techniques, the NO oxidation activities of these catalysts are relatively low probably due to the containing of alkali metals (K) or alkaline-earth metals (Mg, Ca). Up to now, there are few reports on the specially designed hydrotalcite-like precursors derived cobalt-based mixed oxide catalysts for NO oxidation. Therefore, it is highly desired and urgent for the special design and fabrication of cobalt-based mixed oxide catalysts with high catalytic NO oxidation activity and low cost using hydrotalcite-like precursors with adjustable metal cations.
Herein, nonprecious mixed oxide catalysts Co3AlO-T and Co2NiAlO-T (T = 500, 800) have been synthesized via a facile and economical thermal decomposition of hydrotalcite precursor method and firstly used for catalytic oxidation of NO. The crystal structure, textural properties and redox property of the samples were systematically characterized by XRD, EXAFS, SEM/HRTEM, BET, XPS, H2-TPR and in situ FT-IR etc. techniques. Furthermore, the active sites and oxidation pathways of the NO oxidation reaction for these catalysts were also carefully studied and the relationship of the structure and activity for the catalysts was also well explained.
2. Experimental
2.1 Materials
All reagents were of analytical pure grade. Cobalt nitrate hexahydrate (Co(NO3)2·6H2O), nickel nitrate hexahydrate (Ni(NO3)2·6H2O) and aluminum nitrate nonahydrate (Al(NO3)3·9H2O) were purchased from Xi Long Chemical Corporation. Sodium hydroxide and sodium carbonate were purchased from Beijing Chemical Works.2.2 Preparation of the catalysts
Co3Al and Co2NiAl hydrotalcite-like compounds with M2+/Al3+ molar ratio fixed at 3.0 were prepared with a constant-pH coprecipitation method. Typically, a mixed salt solution (100 mL) and a mixed basic solution (200 mL) were simultaneously dropwise added into 100 mL distilled water for ∼1 h under vigorous mechanical stirring at room temperature, keeping a constant pH as 10 ± 0.2. The mixed salt solution with a total metal ion concentration of 0.2 M contains suitable amounts of Co(NO3)2·6H2O and Al(NO3)3·9H2O. The mixed basic solution contains NaOH and Na2CO3 with [OH−] = 1.6([M2+] + [M3+]) and [CO32−] = 2.0[M3+]. The resulting suspension was kept at 65 °C for 4 h under stirring in static air and the resulting precipitate was filtered, washed thoroughly with distilled water and dried at 60 °C for 12 h. The obtained hydrotalcite-like precursor was denoted as Co3Al-HT. Precursor Co2NiAl-HT was prepared using a mixed salt solution containing Co(NO3)2·6H2O, Ni(NO3)2·6H2O and Al(NO3)3·9H2O with molar ratio of 2/1/1 and other conditions unchanged. The precursor samples were ground to fine powders and then calcined at 500 °C and 800 °C for 4 h in static air, respectively, to get the mixed oxide catalysts Co3AlO-T and Co2NiAlO-T (T = 500, 800). The oxide catalysts were pressed to a disk, crushed and sieved to particles with 20–40 mesh size and kept in a desiccator.2.3 Catalytic activity tests
The tests of catalytic NO oxidation were conducted in a fixed-bed flow microreactor under atmospheric pressure. Typically, 0.3 g catalyst (sieve fraction of 20–40 mesh) was placed in a quartz reactor (i.d. = 6 mm, L = 540 mm), the reactant gas mixture (500 ppm NO, 8% O2 and N2 balance) was fed to the reactor with a total flow rate of 500 mL min−1, corresponding to a gas hourly space velocity (GHSV) of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mL g−1 h−1. Before the test, the catalyst was pre-treated in a gas flow of O2/N2 (8% O2) at 450 °C at a constant flow rate of 500 mL min−1 for 0.5 h and cooled down to the experiment temperature (150 °C), then the catalyst was pre-adsorbed the reaction gas mixture at 150 °C for 2 h to avoid errors caused by NO adsorption. The steady-state tests were conducted from 150 °C to 350 °C and the gas products (after 0.5 h reaction) were analyzed by a Chemiluminescence NO–NO2–NOx analyzer (model EC 9841, Ecotech Corporation). The NO conversion is defined as:
000 mL g−1 h−1. Before the test, the catalyst was pre-treated in a gas flow of O2/N2 (8% O2) at 450 °C at a constant flow rate of 500 mL min−1 for 0.5 h and cooled down to the experiment temperature (150 °C), then the catalyst was pre-adsorbed the reaction gas mixture at 150 °C for 2 h to avoid errors caused by NO adsorption. The steady-state tests were conducted from 150 °C to 350 °C and the gas products (after 0.5 h reaction) were analyzed by a Chemiluminescence NO–NO2–NOx analyzer (model EC 9841, Ecotech Corporation). The NO conversion is defined as:Experiments for testing the effect of the NO concentration on the NO conversion were made at NO concentration ranging from 300–700 ppm (300, 400, 500, 600, 700 ppm) in a feed containing 8% O2. Experiments for testing the effect of the O2 concentration on the NO conversion were made at O2 concentration ranging from 2–9% (2%, 4%, 6%, 8%, 9%) in a feed containing 500 ppm NO. All of these experiments were conducted at 300 °C with N2 as balance gas and GHSV of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mL g−1 h−1. Experiments for measuring the reaction orders of NO and O2 were made at the same conditions as those of the experiments for testing the effects of NO and O2 concentration on the NO conversion, except the reaction temperature was set at 150 °C.
000 mL g−1 h−1. Experiments for measuring the reaction orders of NO and O2 were made at the same conditions as those of the experiments for testing the effects of NO and O2 concentration on the NO conversion, except the reaction temperature was set at 150 °C.
2.4 Characterizations
Powder X-ray diffraction (PXRD) patterns were taken on a Shimadzu XRD-6000 diffractometer using Cu Kα radiation (λ = 1.5418 Å, 40 kV, 30 mA) in 2θ 3–70° with a scan speed of 10° min−1. Fourier transform infrared spectra (FT-IR) were recorded on a Bruker Vector-22 FT-IR spectrophotometer using the KBr pellet method (sample/KBr = 1/100) in the range of 4000–400 cm−1 with a resolution of 4 cm−1. Scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX) measurements were carried out on an Oxford Instrument INCAx-act EDX detector attached to a Zeiss Supra 55 field emission scanning electron microscope using a 20 kV electron beam and 60 s acquisition. The high resolution transmission electron microscopy (HRTEM) images were recorded on a JEM-2010 high resolution transmission electron microscope with an accelerating voltage of 200 kV. Raman spectra were recorded on a Jobin Yvon Horiba Raman spectrometer model HR800 using a 532 nm line of Ar+ ion laser as the excitation source at room temperature. The content of metal components Co, Ni and Al in the hydrotalcite precursors was measured by inductively coupled plasma (ICP) emission spectroscopy on a Shimadzu ICPS-7500 instrument. The sample (0.025 g) was solubilized using concentrated nitric acid (1 mL) and the solution was diluted to 25 ppm. Thermogravimetric (TG) analysis was performed on a Mettler Toledo TGA/DSC1 STARe system under static air atmosphere at a heating rate 10 °C min−1 from room temperature to 900 °C.Low temperature N2 adsorption–desorption measurement was conducted at 77 K using a Quantachrome Autosorb-1C-VP system. Before the measurement, the samples were outgassed under vacuum at 300 °C for 6 h. The specific surface area was determined by the Brunauer–Emmett–Teller (BET) method upon the adsorption branch and the pore size distribution by the Barrett–Joyner–Halenda (BJH) method upon the desorption branch of the isotherm.
X-ray photoelectron spectra (XPS) were obtained on a Thermo VG ESCALAB 250 X-ray photoelectron spectrometer at a base pressure of 2 × 10−9 Pa in the analysis chamber using Al Kα X-ray (1486.6 eV) as excitation source and the binding energies were calibrated by the C 1s line at 284.9 eV.
Temperature-programmed reduction (TPR) measurements of the samples were carried out on a Thermo Scientific TPDRO 1100 instrument equipped with a thermal conductivity detector (TCD). In each TPR run, 20 mg sample was placed in a quartz tube and sandwiched between two quartz wool plugs. Prior to each run, the sample was heated to 300 °C with Ar flushing (20 mL min−1) for 30 min, and then cooled down to room temperature. Afterward, the sample was heated to 1000 °C at a ramp of 10 °C min−1 and reduced in a reducing environment (10 vol% H2 in Ar at a flow rate of 30 mL min−1). The H2 consumption was monitored by the TCD during the heating.
Temperature-programmed desorption (TPD) tests were conducted on the fixed-bed flow microreactor. Prior to the TPD test of NO adsorption alone at 50 °C, 0.3 g sample was placed in the quartz tube and pre-treated at 450 °C for 0.5 h under 8% O2/N2 gas flow (500 mL min−1) and then cooled down to 50 °C. After the sample was exposed in 500 ppm NO/N2 (500 mL min−1) at 50 °C for 2 h and flushed by N2 (500 mL min−1) for 1 h, the desorption profiles of NO and NO2 were obtained by heating the sample to 400 °C at a heating rate of 5 °C min−1 in a N2 flow (500 mL min−1). Prior to the TPD test of NO and O2 co-adsorption at 300 °C, 0.3 g sample was placed in the quartz tube and pre-treated at 450 °C for 0.5 h under 8% O2/N2 gas flow (500 mL min−1) and then cooled down to 300 °C. Subsequently, the sample was exposed in 500 ppm NO/8% O2/N2 (500 mL min−1) at 300 °C for 2 h and cooled down to 50 °C, then flushed by N2 (500 mL min−1) for 1 h, and the desorption profiles of NO and NO2 were obtained by heating the sample to 400 °C at a heating rate of 5 °C min−1 in a N2 flow (500 mL min−1). The NO and NO2 desorption signals were continuously recorded as a function of increasing temperature using a on-line Chemiluminescence NO–NO2–NOx analyzer (model EC 9841, Ecotech Corporation).
Extended X-ray absorption fine structure (EXAFS) measurements were carried out at 1W2B beamline of the Beijing Synchrotron Radiation Facility (BSRF) operating at about 2.5 GeV and 200 mA and using a Si (111) double-crystal monochromator. The absorption spectra of Co K-edge and Ni K-edge of the samples and the reference compounds Co3O4 and NiO were collected in the transmission mode at room temperature. Data analysis was performed using IFEFFIT program packages, the back-subtracted EXAFS function was converted into k space and weighted by k3, then the Fourier transforming of the k3-weighted EXAFS data was performed in the range of k = 3–14 Å−1 with a Hanning function window and the radial structure function was obtained.
In situ FT-IR spectra were obtained on a Nicolet 380 FT-IR spectrometer in the range of 650–4000 cm−1 after 64 scans at a resolution of 4 cm−1. Prior to the recording of IR spectra, the sample (10 mg, pellets with diameter of 13 mm) was pre-treated in pure N2 at 300 °C for 1 h with a flow rate of 30 mL min−1 to eliminate the impure species on the surface of the sample. After the background spectrum was recorded with the flowing of N2 and was subtracted, the sample was exposed to 500 ppm NO/N2 for 60 min and followed by exposed to 500 ppm NO and 8% O2 in N2 for 60 min and the time-dependent FT-IR spectra were recorded.
3. Results and discussion
3.1 Crystal structure and textural properties
The XRD patterns of the precursors Co3Al-HT, Co2NiAl-HT and the derived mixed oxide catalysts Co3AlO-500, Co2NiAlO-500, Co3AlO-800 and Co2NiAlO-800 are shown in Fig. 1. The crystal structure parameters of precursors and catalysts are summarized in Tables S1† and 1, respectively. All the precursors show the characteristic reflections of a pure hydrotalcite phase (JCPDS 22-0700),24,28,29 with sharp and symmetric reflections at 2θ ∼ 11.8°, 23.5°, 35° for the basal (003), (006) and (009) planes, and broad and asymmetric reflections at ∼35°, 39°, 47° for the non-basal (012), (015) and (018) planes. The broad signal between 32° and 38° may be resulted from the overlap of (009) and (012) reflections. Moreover, the reflections of (110) and (113) can be clearly seen at 2θ around 60–62°, implying a good dispersion of metal cations in the hydrotalcite layers.24 These phenomena suggest that the hydrotalcite phase possesses well-formed layered structure and is highly crystallized. The reflections appeared at d003 = 0.76 nm indicate that the interlayer anions are CO32−, which is confirmed by the sharp and strong FT-IR absorption peak at ca. 1371 cm−1 due to the ν3 (asymmetric stretching) vibrational mode of CO32− ions associated with a shoulder at 857 cm−1 (Fig. 3A).30,31 Compared to Co3Al-HT, the absence of other crystalline phase in Co2NiAl-HT suggests that Ni2+ ions isomorphically substitute partial Co2+ ions in the brucite-like layers due to the similar ionic radii of Co2+ ions (0.74 Å) and Ni2+ ions (0.69 Å),30 while the lower values of d110 and D110 in Table S1† for Co2NiAl-HT may be attributed to the slightly smaller ionic radii of Ni2+ ions than Co2+ ions and the synergetic effect between Co2+ ions and Ni2+ ions.30,32 | ||
| Fig. 1 XRD patterns of the hydrotalcite precursors: Co3Al-HT (a0) and Co2NiAl-HT (b0) and the oxide catalysts: Co3AlO-500 (a), Co2NiAlO-500 (b), Co3AlO-800 (a1) and Co2NiAlO-800 (b1). | ||
| Samples | Crystallite size of spinela (nm) | a (nm) | SSA (m2 g−1) | Total pore volume (cm3 g−1) | (Co + Ni)/Alb | Co/Nib |
|---|---|---|---|---|---|---|
a Calculated from the diffraction peaks at 2θ ∼ 59° (511) in the XRD (Fig. 1) upon Scherrer formula D = 0.89λ(β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ) (λ is the X-ray wavelength (0.1542 nm), θ is the diffraction angle and β is the full width at half maximum (in radian)).b Obtained from the ICP analysis on the as-prepared Co3Al-HT and Co2NiAl-HT precursors. θ) (λ is the X-ray wavelength (0.1542 nm), θ is the diffraction angle and β is the full width at half maximum (in radian)).b Obtained from the ICP analysis on the as-prepared Co3Al-HT and Co2NiAl-HT precursors. |
||||||
| Co3AlO-500 | 16.5 | 0.8063 | 96.2 | 0.69 | 2.83 | — |
| Co2NiAlO-500 | 11.0 | 0.8082 | 88.6 | 0.78 | 2.86 | 2.11 |
| Co3AlO-800 | 50.0 | 0.8090 | 19.3 | 0.07 | 2.83 | — |
| Co2NiAlO-800 | 40.8 | 0.8091 | 17.2 | 0.10 | 2.86 | 2.11 |
After calcination at 500 °C and 800 °C, the characteristic XRD reflections of the hydrotalcite-like compounds completely disappear, indicating that the hydrotalcite phase has changed to mixed metal oxide phase, supported by the TG results without obvious weight loss after 500 °C (Fig. S1†). While the strong diffraction peaks for all the catalysts at 2θ ∼ 19°, 31°, 37°, 39°, 45°, 55°, 59° and 65° can be indexed to the (111), (220), (311), (222), (400), (422), (511) and (440) planes of cobalt-based spinel phases (Co3O4, JCPDS 42-1467; Co2AlO4, JCPDS 38-0814; CoAl2O4, JCPDS 44-0160; NiCo2O4, JCPDS 73-1702),24,28 and the additional diffraction peaks appeared at 2θ ∼ 37°, 43° and 63° for Co2NiAlO-800 can be attributed to the (111), (200) and (220) planes of cubic NiO phase (JCPDS 73-1523). From the XRD analysis, it is impossible to distinguish the cobalt-containing spinel-type oxides (Co3O4, Co2AlO4, CoAl2O4 and NiCo2O4) due to the almost identical positions of the diffractions. Compared to Co3AlO-500 and Co2NiAlO-500, the obvious increase in the intensity and sharpness of the XRD diffraction peaks for Co3AlO-800 and Co2NiAlO-800 may be attributed to the growth of the spinel phases and sintering of the particles, implying that the increase in the calcination temperature can enhance the crystallinity of the spinel phases.33 The lattice parameter a values (Table 1) of the catalysts are lower than those of the pure cobalt-containing spinel-type oxides (Co3O4 0.8084 nm, Co2AlO4 0.8086 nm, CoAl2O4 0.8104 nm and NiCo2O4 0.8114 nm), indicating the formation of homogeneous and stable non-stoichiometric spinel-like oxides, probably in the form of anion vacancies and/or dissolution of Al3+ ions in the Co3O4 and/or NiCo2O4 spinel phases.28,33,34 And these solid solution phases are denoted as Co(Co,Al)2O4 and Ni(Co,Al)2O4.
Based on the strong and isolated diffraction peaks at 2θ ∼ 59°, the crystallite sizes of spinel phase for the catalysts were calculated by the Scherrer equation and listed in Table 1. The crystallite size of Co2NiAlO-500 is smaller than that of Co3AlO-500, suggesting that the incorporation of Ni in the cobalt-based spinel phase may inhibit the nanocrystal growth. Similar inhibiting effect has been reported by Tang et al. in the formation of Co–Mn solid solution.35 The crystallite sizes of Co3AlO-800 and Co2NiAlO-800 are much larger than those of the 500 °C calcined catalysts, confirming the further growth of the spinel phases due to the higher calcination temperature, while the formation of NiO in Co2NiAlO-800 may also inhibit the growth of the spinel phase to some extent.
EXAFS measurement was performed to confirm the mainly existed Co and Ni phases in the oxide catalysts. The radial structure functions (RSFs) of Co K-edge of the oxide catalysts Co3AlO-500 and Co2NiAlO-500, compared to Co3O4, are shown in Fig. 2A. It has been reported that the Co environment in Co3O4 corresponds to a spinel atomic arrangement with Co2+ and Co3+ ions located in tetrahedron (Td) and octahedron (Oh) coordination, giving an average environment around the Co atoms of 5.3 O at 1.91 Å (first shell), 4Co at 2.85 Å (second shell) and 8Co at 3.36 Å (third shell).36 Thus, the first three peaks in the RSF at 1.496 Å, 2.399 Å, 2.963 Å (not corrected by phase scattering shift) can be ascribed to the above mentioned three coordination shells, and the fourth peak at 4.633 Å corresponds to higher Co–Co coordination shell.14,37 It can be clearly seen that the RSF of Co K-edge of the oxide catalyst Co3AlO-500, very different from that of CoAl2O4,14 is similar to that of Co3O4, implying that Co3AlO-500 mainly existed as Co3O4-like spinel phase. While the RSF of Co K-edge of Co2NiAlO-500 (Fig. 2A) is resemble to those of Co3O4 and NiCo2O4 reported by J. F. Marco et al.38 and its RSF of Ni K-edge (Fig. 2B), different from that of NiO, is similar to that of NiCo2O4.38 These results suggest that the oxide catalyst Co2NiAlO-500 mainly existed as NiCo2O4-like spinel phase, in line with the above XRD results.
 | ||
| Fig. 2 (A) Co K-edge and (B) Ni K-edge radial structure functions of the catalysts: Co3AlO-500 (a), Co2NiAlO-500 (b) and the references Co3O4 and NiO. | ||
The obvious bands around 670 and 568 cm−1 of the oxide catalysts in FT-IR spectra (Fig. 3A), resemble the band position of Co3O4 (672 and 590 cm−1), correspond to the ν1 and ν2 vibrations of spinel phase.39 The obvious shift of ν2 band (characteristic of Co3+ in octahedral coordination) of the oxides may be ascribed to some dissolution of Al3+ in Co3O4 to form Co(Al)O solid solution and/or the formation of non-stoichiometric spinel phases,33,40,41 in line with the XRD results. Moreover, the Raman spectra (Fig. 3B) of the oxide catalysts exhibit the strongest bands at ∼676 cm−1 and very weak bands at ∼186 cm−1, which are different from those of the CoAl2O4 with additional bands at 753 cm−1 and 412 cm−1,42 but similar to those of the reference Co3O4 and the Co–Al mixed oxides derived from hydrotalcites.43 These results further confirm the dissolution of Al3+ in Co3O4 and NiCo2O4 spinel phases and the formation of non-stoichiometric cobalt-based spinel phases.
The (Co + Ni)/Al molar ratios (Table 1) of the oxide catalysts Co3AlO-T and Co2NiAlO-T (T = 500, 800) obtained from ICP data of the corresponding hydrotalcite precursors Co3Al-HT and Co2NiAl-HT are 2.83 and 2.86, respectively, similar to the feeding values (3.0) during the synthesis, indicating the complete coprecipitation of the metal cations in the hydrotalcite precursors. While the Co/Ni molar ratios of Co2NiAlO-T (T = 500, 800) (2.11) are nearly the same as the feeding ratios (2.0), further confirming the substitution of partial Co2+ ions by Ni2+ ions in the well-crystallized Co2NiAl-HT precursor compared to Co3Al-HT. Thus, the obtained mixed oxide catalysts mainly consist of homogeneous and stable non-stoichiometric cobalt-based spinel phases. In detail, both of Co3AlO-500 and Co3AlO-800 are composed of Co(Co,Al)2O4 phase, Co2NiAlO-500 consists of Ni(Co,Al)2O4 phase, while Co2NiAlO-800 consists of Co(Co,Al)2O4 and NiO phases.
The morphological properties of the hydrotalcite precursors and derived oxide catalysts are revealed by the SEM images (Fig. 4). For the precursors, the high-magnification SEM images (Fig. 4(a0 and b0)) show the typical “sand-rose” morphology, characteristic to the hydrotalcite-like materials.44,45 In detail, the SEM image of Co3Al-HT clearly show the flowerlike ensembles (∼830 nm) consisting of interlaced petal-like nanoplates with thickness of ca. 16 nm and width of ca. 200 nm. While for Co2NiAl-HT, much smaller flowerlike ensembles (∼420 nm) consisting of decreased interlaced petal-like nanoplates (thickness ca. 16 nm and width ca. 116 nm) can be clearly seen, in line with the smaller D110 values (Table S1†).
After calcination at 500 °C, the SEM images of Co3AlO-500 and Co2NiAlO-500 (Fig. 4(a and b)) also show the similar nanoflowerlike morphology with no observable size changes of the ensembles and nanoplates, well inheriting the morphology of the precursors. Compared to Co3AlO-500, the smaller size of nanoplates in Co2NiAlO-500 can be obviously seen, consistent with its smaller crystallite size of spinel phase revealed by XRD. Many stack holes (∼60–100 nm) are formed by the self assembly interlaced petal-like nanoplates in the nanoflowerlike oxide catalysts. However, after calcination at 800 °C, the SEM images of Co3AlO-800 and Co2NiAlO-800 (Fig. 4(a1 and b1)) depict the formation of irregular nanoparticles which are disorderly stacked and severely agglomerated, completely different from the nanoflowerlike morphology of the precursors. The EDX results of the precursors (inset in Fig. 4(a0 and b0)) reveal the existence of Co, Al, O, C and Co, Ni, Al, O, C elements in the hydrotalcite precursors Co3Al-HT and Co2NiAl-HT, respectively. The mapping analysis of the samples (Fig. S2†) shows that all the precursors and catalysts possess nearly uniformly distributed metal elements (Co, Ni and Al) due to the atomic-scale uniform distribution of metal cations in the hydrotalcite layers and topotactic transformation nature of the hydrotalcite thermal decomposition to mixed oxides.22,23 Thus, it can be deduced that the nanoflowerlike morphology of cobalt-based mixed oxide catalysts may avoid the severe aggregation of the particles and disperse the active redox components, and the presence of many stack holes in the nanoflowerlike oxide catalysts may lead to the relatively high surface areas and pore volumes.
The textural structures of the oxide catalysts are studied by low temperature N2 adsorption analysis. The N2 adsorption–desorption isotherms of Co3AlO-500 and Co2NiAlO-500 (Fig. 5) exhibit type IV isotherms with H3 type hysteresis loop upon the IUPAC classification,46 typical of mesoporous materials, implying the probable existence of slit-shaped pores.18,26,47,48 The BJH pore size distribution curves of Co3AlO-500 and Co2NiAlO-500 (inset in Fig. 5) show that the pore sizes at the maximum probability of the samples are 8.6 nm, suggesting that the samples possess relatively uniform mesoporous structure.37 The BET surface area and total pore volume of the oxide catalysts are summarized in Table 1. The catalysts Co3AlO-500 and Co2NiAlO-500 possess larger specific surface area of 96.2–88.6 m2 g−1 and total pore volume of 0.69–0.78 m3 g−1, compared to those of Co3AlO-800 (19.3 m2 g−1, 0.07 m3 g−1) and Co2NiAlO-800 (17.2 m2 g−1, 0.10 m3 g−1), in agreement with the smaller spinel crystallite sizes revealed by XRD and the nanoflowerlike morphology shown by SEM, which is benefit for the exposure of the active sites. The nanoflowerlike morphology and larger surface area can also facilitate the diffusion of gas reactants and products and the gas–solid interaction.
 | ||
| Fig. 5 N2 adsorption–desorption isotherms and pore size distribution curves of Co3AlO-500 (a) and Co2NiAlO-500 (b). | ||
The morphology and microstructure of the mixed oxide catalysts are further revealed by the HRTEM analysis. The HRTEM images of Co3AlO-500 and Co2NiAlO-500 (Fig. 6(a and b)) show relatively small spherical-like nanoparticles with sizes of 6–22 nm, close to the crystallite sizes of spinel phases (11.0–16.5 nm) revealed by XRD. One can also recognize the slit-like pores formed by these small nanoparticles, confirming the mesoporous characteristic of these catalysts. However, the HRTEM images for Co3AlO-800 and Co2NiAlO-800 (Fig. 6(c and d)) show typical hexagonal sheet-like nanoparticles with sizes of 50–80 nm, slightly bigger than the crystallite sizes of spinel phases (40.8–50 nm). Compared to Co3AlO-500 and Co2NiAlO-500, the greatly increased particles sizes and crystallinity, and the greatly decreased surface area and pore volume for Co3AlO-800 and Co2NiAlO-800 can be ascribed to the sintering effect that promotes small particles to merge and recrystallize into larger particles19 and also leads to the disappear of mesoporous structure.
The morphology and structure of these nanoparticles have also been analyzed by HRTEM shown in Fig. 6(a′–d′). The catalysts Co3AlO-500, Co2NiAlO-500 and Co2NiAlO-800 are revealed to be assembled by spherical or rod-like nano-crystallites with random orientation and high dispersion.37 While Co3AlO-800 only shows a well-defined large plate-like nano-crystallite with high crystallinity. The observed distinct lattice fringes in HRTEM images (Fig. 6(a′, b′ and d′)) of Co3AlO-500, Co2NiAlO-500 and Co2NiAlO-800 with interplanar distances of ca. 0.47 nm, ca. 0.28 nm and ca. 0.24 nm correspond to the crystallographic planes (111), (220) and (311), respectively, of the cubic cobalt-based spinel phases,27,43 and the corresponding fast Fourier transform (FFT) patterns (inset in Fig. 6(a′, b′ and d′)) also show diffraction spots due to these planes of the cubic cobalt-based spinel phases, in line with the XRD results. For Co3AlO-800, the special interplanar angles between the planes with interplanar distances of 0.473 nm, 0.283 nm and 0.241 nm indicate that the observed planes are (111), (2−20) and (3−11) planes.49 In addition, the lattice fringes in HRTEM image of Co2NiAlO-800 (Fig. S3†) with the interplanar distance of 0.212 nm correspond to the (200) plane of cubic NiO phase,50 in line with the XRD results. Thus, the nanoflowerlike mixed oxide catalysts Co3AlO-500 and Co2NiAlO-500 possess highly dispersed active cobalt-based spinel phases with smaller crystallite sizes, larger surface areas and mesoporous structure, which may give more surface active sites and are expected to exhibit higher catalytic activity.
3.2 Catalytic NO oxidation performance
The catalytic activities of the oxide catalysts Co3AlO-500, Co2NiAlO-500, Co3AlO-800 and Co2NiAlO-800 towards NO oxidation are shown in Fig. 7. The thermodynamic equilibrium curve of NO to NO2 under given conditions shows that the equilibrium conversion decreases with the increasing reaction temperature because the reaction is exothermic and reversible.17 In the temperature range 150–350 °C, the catalysts Co3AlO-500 and Co2NiAlO-500 show similar catalytic activity towards NO oxidation, much higher than Co3AlO-800 and Co2NiAlO-800. In detail, at lower temperatures, the NO conversion for all the catalysts are relatively low since the NO oxidation is kinetically limited.1 With the temperature rising, the NO conversion over Co3AlO-500 and Co2NiAlO-500 first increase to the maximum ∼88% at 285 °C and then drop down due to the thermodynamic limitations, while the NO conversion of Co3AlO-800 and Co2NiAlO-800 only increase to the maximum 55.6% and 66.6% at 350 °C, respectively. The larger specific surface areas of the catalysts Co3AlO-500 and Co2NiAlO-500 enabled by the smaller crystallite sizes and mesoporous structure improve their low temperature NO conversion. Similar phenomenon has been reported in the Co3O4 nanowires-array catalysts by Ren et al.19 From the catalytic oxidation performance of the oxide catalysts listed in Table 2, it is noted that the NO conversion for Co3AlO-500 and Co2NiAlO-500 are close to those of the previously reported Co3O4 nanowire-arrays prepared from cobalt nitrate and cobalt acetate19 and La0.9Ba0.1CoO3 perovskites17 and higher than that of Co3O4/CeO2 (ref. 18) and much higher than those of the Co3Al-oxide25 and CoMgAlO-array27 derived from hydrotalcites precursors.| Catalysts | TCalc. (°C) | SSA (m2 g−1) | Temp. (°C) | Conv.NO (%) | Reactant gas mixture | Space velocity/mL g−1 h−1 | Ref. |
|---|---|---|---|---|---|---|---|
| Co3AlO-500 | 500 | 96.2 | 285 | 88.8 | 500 ppm NO, 8% O2, N2 balance | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
This work |
| Co2NiAlO-500 | 500 | 88.6 | 285 | 87.6 | 500 ppm NO, 8% O2, N2 balance | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
This work |
| Co3AlO-800 | 800 | 19.3 | 350 | 55.6 | 500 ppm NO, 8% O2, N2 balance | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
This work |
| Co2NiAlO-800 | 800 | 17.2 | 350 | 66.6 | 500 ppm NO, 8% O2, N2 balance | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
This work |
| Co3O4 nano-arrays | 300 | 122.6 | 275 | 80 | 500 ppm NO, 10% O2, N2 balance | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1 000 h−1 |
19 |
| La0.9Ba0.1CoO3 | 700 | 7.48 | 265 | 93 | 400 ppm NO, 10% O2, N2 balance | 180![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
17 |
| Co3O4/CeO2 | 500 | 21 | 325 | ∼67 | 390 ppm NO, 8% O2, N2 balance | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
18 |
| Co3Al-oxide | 800 | 22 | 300 | 48.8 | 800 ppm NO, 8% O2, N2 balance | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1 000 h−1 |
25 |
| CoMgAlO-array | 500 | 116 | 300 | 46 | 500 ppm NO, 8% O2, N2 balance | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
27 |
The effect (Fig. 8A) of NO concentration (300–700 ppm) on NO conversion of the catalysts Co3AlO-500 and Co2NiAlO-500 was studied. It is noted that the conversion curves for Co3AlO-500 and Co2NiAlO-500 are indistinguishable, similar to that observed in Fig. 7, confirming the similar NO oxidation performance for these two catalysts. Cai et al.51 reported that in the modified Cr/Ce0.2Zr0.8O2 catalyst with rigid benzene-muti-carboxylate ligands, the NO conversion decreased from 63% to 47% with the increase of NO concentration from 300 ppm to 570 ppm, and then almost stayed a constant with the increase of NO concentration from 570 ppm to 660 ppm. Interestingly, in our case, the NO conversion (∼86%) of Co3AlO-500 and Co2NiAlO-500 almost maintained the same as the NO concentration increased from 300 ppm to 700 ppm, different from the phenomenon reported by Cai et al., suggesting the neglectable effect of the NO concentration (300–700 ppm) on the NO conversion over these oxide catalysts and the varied reaction mechanism between our catalysts and the modified Cr/Ce0.2Zr0.8O2 catalysts. These results also mean that the cobalt-based mixed oxide catalysts can keep high NO conversion with a relatively wide NO concentration range, which is very appealing in the NOx removal application.
The effect of O2 concentration (2–9%) on NO conversion of the catalysts Co3AlO-500 and Co2NiAlO-500 was also studied. As Fig. 8B shows, the NO conversion increased slowly from 75.1% to 86.6% with the increase of O2 concentration from 2% to 8% and almost kept the same with the further increase of O2 concentration from 8% to 9%, similar to the phenomenon of the Co3O4 nanowires-array catalysts.19 The relatively high conversion in the whole O2 concentration range (2–9%) of these catalysts suggests that they are reliable to work under both lean-burn and rich-burn conditions, which is quite important for the NOx removal of the internal combustion engines.13,19 The long term isothermal catalytic stability tests of catalysts Co3AlO-500 and Co2NiAlO-500 were studied by monitoring the NO conversion change at the temperature 300 °C. The NO conversion was quantified every 2 hours using the Chemiluminescence NO–NO2–NOx analyzer. Fig. 8C demonstrated stable NO conversion without any degradation during the 20 hours' test and both catalysts maintained the high NO conversion of ∼86%. Moreover, the surface areas and SEM images (Fig. S4†) of the used catalysts Co3AlO-500-u and Co2NiAlO-500-u after the stability tests were also obtained. The surface area of Co3AlO-500-u (58.8 m2 g−1) was lower than Co3AlO-500 (96.2 m2 g−1) along with aggregated nanoplates (Fig. S4a†) instead of flowerlike morphology of fresh Co3AlO-500. While the surface area of Co2NiAlO-500-u (88.0 m2 g−1) was well-kept compared to fresh one (88.6 m2 g−1), in line with its flowerlike morphology (Fig. S4b†). These results imply that Co2NiAlO-500 may maintain the high NO oxidation activities for a longer time than Co3AlO-500.
It is reported that the NO oxidation reaction is a reversible reaction and NO2 formation may inhabit the NO conversion.15,17,19 While in our catalytic tests, the NO oxidation can be reasonably considered as irreversible since the O2 concentration (8%) is high enough compared with the NO concentration (500 ppm). Thus, the reaction rate only depends on the concentration of NO and O2. For the catalysts Co3AlO-500 and Co2NiAlO-500, the reaction orders of NO (1.37 and 1.28) and O2 (0.50 and 0.56) were obtained by the kinetic analysis (Fig. 8(D and E)) through only changing the concentration of NO or O2, respectively. These results indicate that the NO oxidation over the catalysts Co3AlO-500 and Co2NiAlO-500 follows the Eley–Rideal mechanism where NO is gaseous and O2 is at adsorbed state, similar to the previously reported Co3O4 based catalysts for NO oxidation.10,19 The calculated results of the apparent activation energy (Ea) and pre-exponential factor (A) of the catalysts Co3AlO-500 and Co2NiAlO-500 are given in Table S2.† The Ea and A values were obtained by the Arrhenius plot, in which the reaction rate constant k was calculated upon the measured reaction rate r and reaction orders a and b. The Ea values for catalysts Co3AlO-500 and Co2NiAlO-500 obtained in the temperature range of 150–190 °C (Fig. 8F) were 29.12 and 29.84 kJ mol−1, respectively. The similarity of the Ea values between Co3AlO-500 and Co2NiAlO-500 indicates that the active sites and reaction mechanism of the NO oxidation reaction over these catalysts may be similar.19,52 In addition, the catalyst Co3AlO-500 possesses slightly larger pre-exponential factor (A) value (19.44 mol g−1 s−1), compared to Co2NiAlO-500 (18.39 mol g−1 s−1), suggesting the slightly more active sites of Co3AlO-500 exposed.19 Compared to Co3AlO-800 and Co2NiAlO-800, the excellent catalytic NO oxidation performance of Co3AlO-500 and Co2NiAlO-500 can be assigned to the cobalt-based spinel phases with smaller crystallite sizes, larger surface areas and mesoporous structure resulting from topotactic transformation of the hydrotalcite-like precursors at suitable temperature, which may give larger amount of surface active sites.
3.3 Catalytic mechanism
After the microstructures and the catalytic performance of the cobalt-based mixed oxide catalysts have been carefully analyzed, a noticeable problem is why the different catalysts Co3AlO-500 and Co2NiAlO-500 exhibit similar NO oxidation performance. XPS was used as an effective tool to gain information of the surface element compositions and chemical states of the catalysts Co3AlO-500 and Co2NiAlO-500 and the used ones Co3AlO-500-u and Co2NiAlO-500-u after stability tests. The XPS spectra of Co 2p, Ni 2p, Al 2p and O 1s are illustrated in Fig. 9. The binding energies (BE) of Co 2p3/2, Ni 2p3/2, Al 2p and O 1s and the molar ratios of the surface Co3+/Co2+, Ni3+/Ni2+, Oads/Olatt, Co/Ni and (Co + Ni)/Al are summarized in Table 3. The molar ratios of Co3+/Co2+, Ni3+/Ni2+ and Oads/Olatt were obtained by quantitative calculation of the corresponding deconvoluted peak areas. | ||
| Fig. 9 XPS spectra of Co 2p, Ni 2p, Al 2p and O 1s for the oxide catalysts Co3AlO-500 (a), Co2NiAlO-500 (b) and used ones Co3AlO-500-u (c) and Co2NiAlO-500-u (d). | ||
| Catalysts | Binding energy (eV) | Surface molar ratio | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Co2+ | Co3+ | Ni2+ | Ni3+ | Olatt | Oads | Al2p | Co3+/Co2+ | Ni3+/Ni2+ | Oads/Olatt | Co/Ni | (Co + Ni)/Al | |
| Co3AlO-500 | 781.3 | 779.8 | — | — | 530.1 | 531.5 | 73.8 | 1.22 | — | 0.77 | — | 0.84 |
| Co2NiAlO-500 | 780.9 | 779.2 | 854.1 | 855.8 | 529.7 | 531.7 | 73.6 | 1.30 | 1.24 | 1.05 | 1.31 | 1.24 |
| Co3AlO-500-u | 781.6 | 780.1 | — | — | 530.3 | 531.7 | 73.5 | 1.25 | — | 0.82 | — | 0.86 |
| Co2NiAlO-500-u | 781.1 | 779.5 | 854.1 | 855.7 | 529.8 | 531.4 | 73.6 | 1.31 | 1.26 | 1.12 | 1.25 | 1.20 |
The high resolution Co 2p spectra of Co3AlO-500 and Co2NiAlO-500 present two distinct peaks centered at ∼780 eV and ∼795 eV attributed to Co 2p3/2 and Co 2p1/2, respectively.19,32,53 The spin–orbit splitting value is 15.3–15.4 eV, implying the coexistence of Co2+ and Co3+ species.37 By performing the peak-fitting deconvolution, the Co 2p3/2 peaks can be divided into two characteristic peaks at ∼779.2 eV and ∼780.9 eV, assigned to Co3+ and Co2+ in the Co3O4-like and NiCo2O4-like spinel phases.19,32,53 Moreover, it is noted that the BE values of the two deconvoluted peaks of Co 2p3/2 for Co2NiAlO-500 (779.2 eV, 780.9 eV) are both slightly lower than those of Co3AlO-500 (779.8 eV, 781.3 eV). The surface Co3+/Co2+ molar ratio of catalyst Co2NiAlO-500 is 1.30 (Table 3), slightly higher than that of Co3AlO-500 (1.22), suggesting that the Co3+ species are dominant on the surface of these catalysts and the oxidation state of Co can be changed via the addition of Ni due to the strong interaction between Co and Ni.
The high resolution Ni 2p spectrum of Co2NiAlO-500 exhibits two strong peaks at 854.9 eV for Ni 2p3/2 and 872.9 eV for Ni 2p1/2, and two strong satellite peaks at 861.2 eV and 879.3 eV. The Ni 2p3/2 peak is deconvoluted into two peaks at 854.1 and 855.8 eV, ascribed to Ni3+ and Ni3+, respectively,20,38 and the surface Ni3+/Ni2+ molar ratio (Table 3) is 1.24. These results suggest that the Co2NiAlO-500 possesses a surface composition of Co2+, Co3+, Ni2+ and Ni3+, in line with the literature observations for NiCo2O4.54,55 It can be seen that the surface (Co + Ni)/Al molar ratios are 0.84 and 1.24 for Co3AlO-500 and Co2NiAlO-500, respectively, much lower than the bulk (Co + Ni)/Al molar ratios of 2.83 and 2.86, suggesting the surface enrichment of Al on these catalysts.33 While Co2NiAlO-500 possesses higher surface (Co + Ni)/Al molar ratio than Co3AlO-500, indicating that the addition of Ni may prohibit the surface enrichment of Al and increase the surface content of Co and Ni. Moreover, the surface Co/Ni molar ratio (1.31) of Co2NiAlO-500 is much lower than the bulk Co/Ni ratio (2.11), indicating the surface enrichment of Ni on the catalyst. The Al 2p spectra of Co3AlO-500 and Co2NiAlO-500 show a single peak at ∼73.6 eV. The BE values of Al 2p (Table 3) are slightly lower than those of Al2O3 (74.1 eV) as well as CoAl2O4 and NiAl2O4 (73.93 eV),30 confirming the existence of non-stoichiometric cobalt-based spinel phases Co(Co,Al)2O4 and Ni(Co,Al)2O4.
The asymmetrical O 1s peak of catalysts Co3AlO-500 and Co2NiAlO-500 can be deconvoluted into two peaks at BE values of ∼530.1 eV and ∼531.5 eV, ascribed to the lattice oxygen (O2−) and surface adsorbed oxygen (O−, O2−, O22−), respectively.1,32,51,53 It has been reported that the adsorption of gaseous O2 on oxygen vacancies can lead to the formation of surface adsorbed oxygen on the oxide catalysts.20,27 As listed in Table 3, the catalyst Co2NiAlO-500 possesses a much higher surface Oads/Olatt molar ratio (1.05) than Co3AlO-500 (0.77), implying the formation of additional oxygen vacancies due to the partial substitution of octahedral Co3+ by Ni2+.56 When these additional oxygen vacancies are filled up with gaseous O2 leading to the increased Oads/Olatt molar ratio, the neighboring Co2+ may be oxidized into Co3+ to balance the charge, resulting in the higher Co3+/Co2+ molar ratio.1 Generally, the surface adsorbed oxygen is much more reactive in the oxidation reaction than lattice oxygen due to its higher mobility.27,53,57
However, considering the surface Co atomic content of Co3AlO-500 and Co2NiAlO-500 is 13.21% and 5.85%, respectively, thus the surface Co3+ content of Co3AlO-500 and Co2NiAlO-500 is 7.26% and 3.31%, respectively. While the surface Ni atomic content of Co2NiAlO-500 is 4.46%, then the surface Ni3+ content is 2.47%. Thus, the total surface Co3+ and Ni3+ content (5.78%) of Co2NiAlO-500 is slightly lower than the surface Co3+ content (7.26%) of Co3AlO-500. While the surface O atomic content of Co3AlO-500 and Co2NiAlO-500 is 50.4% and 41.2%, respectively, thus, the surface adsorption oxygen content of Co3AlO-500 (21.9%) is close to that of Co2NiAlO-500 (21.1%). Moreover, the obtained BE values of Co2+, Co3+, Ni2+, Ni3+, Oads and Olatt and the molar ratios of Co3+/Co2+, Ni3+/Ni2+ and Oads/Olatt for the used catalysts Co3AlO-500-u and Co2NiAlO-500-u are similar to those of the corresponding fresh catalysts, indicating the almost unchanged surface composition of the used catalysts. These results are in good agreement with the stable NO conversion of Co3AlO-500 and Co2NiAlO-500 during the 20 hours' test.
With regard to the active sites for the NO oxidation reaction, Ren et al.19 reported that the Co3+ on the surface acted as the active sites of the Co3O4 nanowire-array catalysts, while Feng et al.1 reported that some of the surface adsorbed oxygen species served as the active site over the La-modified SmMn2O5 mullite oxide (LaxSm1−xMn2Oδ) catalysts, and Chen et al.52 reported that Mn4+ ion associated with O ligand served as the active sites over the LaMnO3 perovskite catalysts. If we presume the Co3+ ions associated with surface adsorbed oxygen as the active sites for Co3AlO-500, then the amount of (Co3+ and Ni3+) ions associated with surface adsorbed oxygen active sites for Co2NiAlO-500 may be a little lower than that of Co3AlO-500. However, the catalytic activity of NO oxidation catalyst is not simply determined by the amount of the active sites, but the reducibility of the catalysts and the adsorbed species formed on the surface of the catalysts may also play crucial roles.
H2-TPR experiment was carried out to investigate the reducibility of the catalysts Co3AlO-500 and Co2NiAlO-500. As shown in Fig. 10, the H2-TPR profile of Co3AlO-500 exhibited a weak peak centered at 347 °C and a strong one at 668 °C. The former can be attributed to the reduction of Co3+ to Co2+ in the Co3+-related spinels, while the latter assigned to the reduction of Co2+-related species to Co0.14,20,27,58 The H2-TPR profile of Co2NiAlO-500 presented one weak peak at 292 °C and one strong and asymmetric peak in the temperature range of 300–800 °C. The former is ascribed to the Co3+ to Co2+, similar to Co3AlO-500. While the shoulder (∼500 °C) of the latter peak is probably related to the reduction of Ni2+-related spinels,32 and the main peak centered at 631 °C can be assigned to the reduction of Co2+-related species, the two reduction peaks are overlapped to form the strong and broad peak,20 which is different from that of Co3AlO-500. It should be mentioned that the two reduction peaks of Co2NiAlO-500 shift to lower temperatures compared to Co3AlO-500 and the total H2 consumption of Co2NiAlO-500 (7.40 mmol g−1) is slightly higher than that of Co3AlO-500 (7.24 mmol g−1). These results suggest that Co2NiAlO-500 possesses higher reducibility than Co3AlO-500 due to the existence of strong interaction between Co and Ni in the NiCo2O4-like spinel phase.20
NO-TPD and in situ FT-IR analyses are combined to reveal the adsorbed species formed on the surface of the catalysts during the NO oxidation process. The NO-TPD profiles of Co3AlO-500 and Co2NiAlO-500 (Fig. 11A and B) for NO adsorption alone at 50 °C showed two distinct NO desorption peaks. The strong and broad NO desorption peaks centered at 152 °C are assigned to the decomposition of nitrite species, while the slightly lower and narrower NO desorption peaks around 302–324 °C are assigned to the decomposition of nitrate species.59 Moreover, the weak and broad NO2 desorption peaks around 288–314 °C are also observed, assigned to the decomposition of nitrates species. Although NO alone is adsorbed over Co3AlO-500 and Co2NiAlO-500, nitrates species are formed surprisingly, indicating the existence of active oxygen species on the surface of these catalysts after 8% O2/N2 pre-treatment. Compared to Co3AlO-500, it can be seen that the desorption peaks' areas of NO and NO2 for Co2NiAlO-500 are much larger, indicating that larger amount of nitrite/nitrate species are formed on the catalyst Co2NiAlO-500 at low adsorption temperature. The NO-TPD profiles of Co3AlO-500 and Co2NiAlO-500 (Fig. 11C and D) for NO and O2 co-adsorption at 300 °C show one stronger NO desorption peak around 316–329 °C and one weaker NO2 desorption peak around 280–307 °C, assigned to the decomposition of nitrate species.59 Compared to Co3AlO-500, although the desorption peaks' area of NO for Co2NiAlO-500 is much larger, the desorption peaks' area of NO2 for Co2NiAlO-500 is only slightly larger, indicating the similar desorption amount of NO2 over the two catalysts at high adsorption temperature.
 | ||
| Fig. 11 NO-TPD profiles of Co3AlO-500 (A and C) and Co2NiAlO-500 (B and D) for NO adsorption at 50 °C (A and B) and NO + O2 co-adsorption at 300 °C (C and D). | ||
The adsorbed nitrate species at high adsorption temperature over the catalysts Co3AlO-500 and Co2NiAlO-500 were further revealed by the in situ FT-IR analysis. Fig. 12 showed the in situ FT-IR spectra of the catalysts after the introduction of 500 ppm NO at 300 °C and followed 8% O2 was introduced. NO adsorption on these catalysts was performed first. For Co3AlO-500 (Fig. 12A), after 5 min of exposure, the broad peak at 1577 cm−1 is ascribed to the ν(N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of the chelating bidentate nitrate while the smaller shoulder at 1630 cm−1 is assigned to the ν(N
O) of the chelating bidentate nitrate while the smaller shoulder at 1630 cm−1 is assigned to the ν(N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of the bridged bidentate nitrate and weakly adsorbed NO2.24,60,61 After 5 min of exposure, the catalyst Co2NiAlO-500 (Fig. 12B) exhibits the main peak at 1630 cm−1 and a shoulder at 1598 cm−1, indicating the much larger amount of the bridged bidentate nitrate and weakly adsorbed NO2. In addition, two very weak peaks are observed at 1906 and 1853 cm−1 for Co2NiAlO-500, may be assigned to linear Ni2+–NO species and Ni2+(NO)2 geminal species.62 It is noticed that the intensities of all these peaks are not obviously increased with time, implying that the formation of these nitrates is very fast and the amount of formed nitrates over these catalysts is very limited due to the limited surface active oxygen species.49 When O2 supply is switched on, the intensities of the above mentioned peaks are increased rapidly, and almost reach the strongest after the NO + O2 feeding for 5 min, implying that the introduction of O2 can further promote the oxidation of NO to NO2 and the storage as nitrate species probably due to the complement of the surface active oxygen species. Some new peaks could also be observed. In detail, the weak peaks at 1286–1289 cm−1 and 1034–1037 cm−1 are ascribed to the νasym(NO2) and νsym(NO2) of the chelating bidentate nitrate, while the weak peaks at 1236–1234 cm−1 and 999–1005 cm−1 are ascribed to the νasym(NO2) and νsym(NO2) of the bridged bidentate nitrate.60 A very weak shoulder at 1195 cm−1 for Co3AlO-500 may be attributed to the ν(N–O) vibration of anionic NOx surface species such as NO− and/or bidentate nitrites.63
O) of the bridged bidentate nitrate and weakly adsorbed NO2.24,60,61 After 5 min of exposure, the catalyst Co2NiAlO-500 (Fig. 12B) exhibits the main peak at 1630 cm−1 and a shoulder at 1598 cm−1, indicating the much larger amount of the bridged bidentate nitrate and weakly adsorbed NO2. In addition, two very weak peaks are observed at 1906 and 1853 cm−1 for Co2NiAlO-500, may be assigned to linear Ni2+–NO species and Ni2+(NO)2 geminal species.62 It is noticed that the intensities of all these peaks are not obviously increased with time, implying that the formation of these nitrates is very fast and the amount of formed nitrates over these catalysts is very limited due to the limited surface active oxygen species.49 When O2 supply is switched on, the intensities of the above mentioned peaks are increased rapidly, and almost reach the strongest after the NO + O2 feeding for 5 min, implying that the introduction of O2 can further promote the oxidation of NO to NO2 and the storage as nitrate species probably due to the complement of the surface active oxygen species. Some new peaks could also be observed. In detail, the weak peaks at 1286–1289 cm−1 and 1034–1037 cm−1 are ascribed to the νasym(NO2) and νsym(NO2) of the chelating bidentate nitrate, while the weak peaks at 1236–1234 cm−1 and 999–1005 cm−1 are ascribed to the νasym(NO2) and νsym(NO2) of the bridged bidentate nitrate.60 A very weak shoulder at 1195 cm−1 for Co3AlO-500 may be attributed to the ν(N–O) vibration of anionic NOx surface species such as NO− and/or bidentate nitrites.63
It is noted that the peaks at 1598 cm−1 for chelating bidentate nitrate species in the in situ FT-IR spectra of Co2NiAlO-500 are broader than those of Co3AlO-500 at 1577 cm−1, and accompanied by much higher shoulders, in line with the larger amount of released NO in the NO-TPD profiles. These results suggest that the NiCo2O4-like spinel phase can capture more NO stored as chelating bidentate nitrate than Co3O4-like spinel phase, which may be related to the strong synergistic effect of Co and Ni originating from the hydrotalcite precursor method. Besides the obviously existed chelating bidentate nitrate species in these catalysts, it should be noted that the intensity of the peaks at 1630 cm−1 corresponding to less stable bridged bidentate nitrate and weakly adsorbed NO2 species of Co2NiAlO-500 is much higher than that of Co3AlO-500, while the appearance of the shoulders at 1648 cm−1 of Co3AlO-500 also indicate more production of NO2.61 These phenomena suggest that the two catalysts possess similar amount of less stable bridged bidentate nitrate and weakly adsorbed NO2 species, in line with the NO-TPD results for the co-adsorption of NO and O2 at 300 °C.
By combining the evidence from XPS, H2-TPR, NO-TPD and in situ FT-IR, the reaction pathways for NO oxidation to NO2 over the catalysts Co3AlO-500 and Co2NiAlO-500 are tentatively proposed and depicted in Scheme 1. In brief, the NO oxidation pathways mainly include: (I) the NO oxidation by the Co3+ and/or Ni3+ associated with surface adsorbed oxygen (Co3+/Ni3+–Oads) species, and (II) the NO oxidation by the Co3+ and/or Ni3+ associated with lattice oxygen (Co3+/Ni3+–Olatt) species. In Scheme 1, two kinds of surface reactive species are ideally representative by the cuboids with different colours (Co3+/Ni3+–Oads species: yellow colour; Co3+/Ni3+–Olatt species: light gray colour), while the bulk of the catalysts is representative by the big cuboids with dark gray colour.
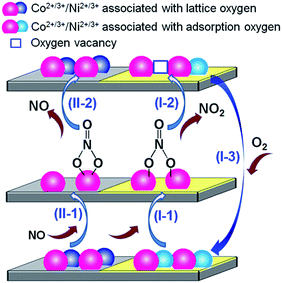 | ||
| Scheme 1 The reaction pathways for NO oxidation on the surface of the cobalt-based mixed oxide catalysts. | ||
For pathway (I), NO was first oxidized by the Co3+/Ni3+–Oads species on the surface of the catalysts, and formed the less stable bridged bidentate nitrate and weakly adsorbed NO2 species, followed by their decomposition into NO2 to generate oxygen vacancies, and finally the gaseous O2 adsorbed onto the oxygen vacancies to replenish the surface adsorbed oxygen and complete the redox cycle. The pathway (I) is in line with the above mentioned Eley–Rideal mechanism. While for pathway (II), the NO was first oxidized by the Co3+/Ni3+–Olatt species on the surface of the catalysts, and then formed the relatively stable chelating bidentate nitrate species, which were prone to decompose into NO, supported by the large amount of the decomposed NO from the nitrate species in the NO-TPD profiles. So pathway (II) is not considered as an effective NO oxidation route. Therefore, the Co3+ and Ni3+ associated with surface adsorbed oxygen and Co3+ associated with surface adsorbed oxygen species are believed to serve as the active sites over the present Co3AlO and Co2NiAlO catalysts, respectively, in the NO oxidation reaction.
Although Co2NiAlO-500 possesses slightly smaller amount of active sites (Co3+ and Ni3+ associated with surface adsorbed oxygen), compared to Co3AlO-500 with active sites (Co3+ associated with surface adsorbed oxygen), catalyst Co2NiAlO-500 exhibits higher reducibility, leading to their similar desorption amount of NO2, thus similar excellent NO oxidation performance. While the lower cost of Co2NiAlO-500 renders it a greater potential for practical applications. Moreover, the exploration of the relationship of the excellent catalytic NO oxidation activity and the microstructure of the facilely synthesized nanoflowerlike oxide catalysts may provide some new insights for the design of hierarchical Co-based oxide catalysts in the NOx removal area.
4. Conclusions
Nonprecious cobalt-based mixed oxide catalysts Co3AlO-500, Co2NiAlO-500, Co3AlO-800 and Co2NiAlO-800 have been fabricated by thermal decomposition of nanoflowerlike hydrotalcite-like precursors Co3Al-HT and Co2NiAl-HT at 500 °C and 800 °C in air. The obtained oxide catalysts mainly consisted of homogeneous and stable non-stoichiometric cobalt-based spinel phases Co(Co,Al)2O4 and Ni(Co,Al)2O4. The catalysts Co3AlO-500 and Co2NiAlO-500 both showed nanoflowerlike morphology, while catalysts Co3AlO-800 and Co2NiAlO-800 both presented disorderly stacked states consisting of severely agglomerated irregular nanoparticles. The catalysts Co3AlO-500 and Co2NiAlO-500 presented excellent NO oxidation performance, with the maximum conversion efficiency ca. 88.8% and 87.6% (285 °C), respectively, which were much higher than the values of Co3AlO-800 and Co2NiAlO-800 (55.6% and 66.6% at 350 °C). The excellent catalytic NO oxidation performance of catalysts Co3AlO-500 and Co2NiAlO-500 could be attributed to the much larger amount of surface active sites endowed by these catalysts with smaller crystallite sizes of cobalt-based spinel phase, higher surface areas, and mesoporous structure. The mechanism study revealed the active sites and oxidation pathways during the NO oxidation reaction over the present catalysts. Although Co2NiAlO-500 possessed slightly smaller amount of active sites (Co3+ and Ni3+ associated with surface adsorbed oxygen), compared to Co3AlO-500 with active sites (Co3+ associated with surface adsorbed oxygen), the reducibility of Co2NiAlO-500 was higher, leading to the similar desorption amount of NO2, thus similar excellent NO oxidation performance of nanoflowerlike catalysts Co3AlO-500 and Co2NiAlO-500. Moreover, catalyst Co2NiAlO-500 has lower cost than Co3AlO-500, rendering it a greater potential for practical applications.Acknowledgements
The authors greatly appreciate the financial support by the National Nature Science Foundation of China (21276015, 21576013, 21521005).References
- Z. J. Feng, J. Q. Wang, X. Liu, Y. W. Wen, R. Chen, H. F. Yin, M. Q. Shen and B. Shan, Catal. Sci. Technol., 2016, 6, 5580–5589 CAS.
- C. H. Kim, G. S. Qi, K. Dahlberg and W. Li, Science, 2010, 327, 1624–1627 CrossRef CAS PubMed.
- J. R. Theis and E. Gulari, Appl. Catal., B, 2007, 74, 40–52 CrossRef CAS.
- X. Auvray, T. Pingel, E. Olsson and L. Olssona, Appl. Catal., B, 2013, 129, 517–527 CrossRef CAS.
- S. F. Zhong, Y. Y. Sun, H. C. Xin, C. P. Yang, L. Chen and X. B. Li, Chem. Eng. J., 2015, 275, 351–356 CrossRef CAS.
- M. Jabłońska and R. Palkovits, Catal. Sci. Technol., 2016, 6, 49–72 Search PubMed.
- J. J. Yu, J. Cheng, C. Y. Ma, H. L. Wang, L. D. Li, Z. P. Hao and Z. P. Xu, J. Colloid Interface Sci., 2009, 333, 423–430 CrossRef CAS PubMed.
- G. Liu and P. X. Gao, Catal. Sci. Technol., 2011, 1, 552–568 CAS.
- Y. Peng, W. Z. Si, J. M. Luo, W. K. Su, H. Z. Chang, J. H. Li, J. M. Hao and J. Crittenden, Environ. Sci. Technol., 2016, 50, 6442–6448 CrossRef CAS PubMed.
- M. F. Irfan, J. H. Goo and S. D. Kim, Appl. Catal., B, 2008, 78, 267–274 CrossRef CAS.
- H. Hu, S. X. Cai, H. R. Li, L. Huang, L. Y. Shi and D. S. Zhang, ACS Catal., 2015, 5, 6069–6077 CrossRef CAS.
- M. F. Fu, C. T. Li, P. Lu, L. Qu, M. Y. Zhang, Y. Zhou, M. G. Yu and Y. Fang, Catal. Sci. Technol., 2014, 4, 14–25 CAS.
- W. C. Wang, G. McCool, N. Kapur, G. Yuan, B. Shan, M. Nguyen, U. M. Graham, B. H. Davis, G. Jacobs, K. Cho and X. H. (Kelly) Hao, Science, 2012, 337, 832–835 CrossRef CAS PubMed.
- Q. Li, M. Meng, N. Tsubaki, X. G. Li, Z. Q. Li, Y. N. Xie, T. D. Hu and J. Zhang, Appl. Catal., B, 2009, 91, 406–415 CrossRef CAS.
- J. Després, M. Elsener, M. Koebel, O. Kröcher, B. Schnyder and A. Wokaun, Appl. Catal., B, 2004, 50, 73–82 CrossRef.
- E. Lira, L. R. Merte, F. Behafarid, L. K. Ono, L. Zhang and B. R. Cuenya, ACS Catal., 2014, 4, 1875–1884 CrossRef CAS.
- C. Zhou, Z. J. Feng, Y. X. Zhang, L. J. Hu, R. Chen, B. Shan, H. F. Yin, W. G. Wang and A. S. Huang, RSC Adv., 2015, 5, 28054–28059 RSC.
- Y. Yu, L. Zhong, J. Ding, W. Cai and Q. Zhong, RSC Adv., 2015, 5, 23193–23201 RSC.
- Z. Ren, Y. B. Guo, Z. H. Zhang, C. H. Liu and P. X. Gao, J. Mater. Chem. A, 2013, 1, 9897–9906 CAS.
- X. Y. Wang, W. Wen, J. X. Mi, X. X. Li and R. H. Wang, Appl. Catal., B, 2015, 176–177, 454–463 CrossRef CAS.
- F. Cavani, F. Trifirò and A. Vaccari, Catal. Today, 1991, 11, 173–301 CrossRef CAS.
- P. J. Sideris, U. G. Nielsen, Z. H. Gan and C. P. Grey, Science, 2008, 321, 113–117 CrossRef CAS PubMed.
- G. L. Fan, F. Li, D. G. Evans and X. Duan, Chem. Soc. Rev., 2014, 43, 7040–7066 RSC.
- J. J. Yu, Z. Jiang, L. Zhu, Z. P. Hao and Z. P. Xu, J. Phys. Chem. B, 2006, 110, 4291–4300 CrossRef CAS PubMed.
- J. J. Yu, X. P. Wang, L. D. Li, Z. P. Hao, Z. P. Xu and G. Q. Lu, Adv. Funct. Mater., 2007, 17, 3598–3606 CrossRef CAS.
- Z. P. Wang, P. Lu, X. M. Zhang, L. G. Wang, Q. Li and Z. L. Zhang, RSC Adv., 2015, 5, 52743–52753 RSC.
- L. G. Dou, T. Fan and H. Zhang, Catal. Sci. Technol., 2015, 5, 5153–5167 CAS.
- J. Pérez-Ramírez, G. Mul, F. Kapteijn and J. A. Moulijn, J. Mater. Chem., 2001, 11, 821–830 RSC.
- V. Rives, O. Prieto, A. Dubey and S. Kannan, J. Catal., 2003, 220, 161–171 CrossRef CAS.
- H. H. Yi, H. Y. Wang, X. L. Tang, P. Ning, L. L. Yu, D. He and S. Z. Zhao, Ind. Eng. Chem. Res., 2011, 50, 13273–13279 CrossRef CAS.
- E. Genty, R. Cousin, S. Capelle, C. Gennequin and S. Siffert, Eur. J. Inorg. Chem., 2012, 2802–2811 CrossRef CAS.
- S. D. Li, S. P. Mo, J. Q. Li, H. D. Liu and Y. F. Chen, RSC Adv., 2016, 6, 56874–56884 RSC.
- S. Kannan and C. S. Swamy, Catal. Today, 1999, 53, 725–737 CrossRef CAS.
- M. Gabrovska, R. Edreva-Kardjieva, K. Tenchev, P. Tzvetkov, A. Spojakina and L. Petrov, Appl. Catal., A, 2011, 399, 242–251 CrossRef CAS.
- W. X. Tang, X. F. Wu, S. D. Li, W. H. Li and Y. F. Chen, Catal. Commun., 2014, 56, 134–138 CrossRef CAS.
- F. Morales, D. Grandjean, A. Mens, F. M. F. de Groot and B. M. Weckhuysen, J. Phys. Chem. B, 2006, 110, 8626–8639 CrossRef CAS PubMed.
- J. Y. Luo, M. Meng, X. Li, X. G. Li, Y. Q. Zha, T. D. Hu, Y. N. Xie and J. Zhang, J. Catal., 2008, 254, 310–324 CrossRef CAS.
- J. F. Marco, J. R. Gancedo, M. Gracia, J. L. Gautier, E. Ríos and F. J. Berry, J. Solid State Chem., 2000, 153, 74–81 CrossRef CAS.
- J. Preudhomme and P. Tarte, Spectrochim. Acta, Part A, 1971, 27, 1817–1835 CrossRef CAS.
- L. Obalová, K. Jirátová, F. Kovanda, K. Pacultová, Z. Lacný and Z. Mikulová, Appl. Catal., B, 2005, 60, 289–297 CrossRef.
- C. Gennequin, S. Siffert, R. Cousin and A. Aboukaïs, Top. Catal., 2009, 52, 482–491 CrossRef CAS.
- B. Jongsomjit, J. Panpranot and J. G. Goodwin Jr, J. Catal., 2001, 204, 98–109 CrossRef CAS.
- D. L. Li, Y. Y. Ding, X. F. Wei, Y. H. Xiao and L. L. Jiang, Appl. Catal., A, 2015, 507, 130–138 CrossRef CAS.
- M. Adachi-Pagano, C. Forano and J. P. Besse, J. Mater. Chem., 2003, 13, 1988–1993 RSC.
- G. Carja, H. Obata, Y. Kameshima and K. Okada, Microporous Mesoporous Mater., 2007, 98, 150–155 CrossRef CAS.
- S. J. Gregg and K. S. W. Sing, Adsorption, Surface Area and Porosity, Academic Press, New York, 1982 Search PubMed.
- Y. Gou, X. Liang and B. H. Chen, J. Alloys Compd., 2013, 574, 181–187 CrossRef CAS.
- C. H. Wang, X. Zhang, D. C. Zhang, C. Yao and Y. W. Ma, Electrochim. Acta, 2012, 63, 220–227 CrossRef CAS.
- K. Song, E. Cho and Y. M. Kang, ACS Catal., 2015, 5, 5116–5122 CrossRef CAS.
- Y. F. Zhao, X. D. Jia, G. B. Chen, L. Shang, G. I. N. Waterhouse, L. Z. Wu, C. H. Tung, D. O'Hare and T. R. Zhang, J. Am. Chem. Soc., 2016, 138, 6517–6524 CrossRef CAS PubMed.
- W. Cai, Q. Zhong, W. Zhao and Y. F. Bu, Appl. Catal., B, 2014, 158–159, 258–268 CrossRef CAS.
- J. H. Chen, M. Q. Shen, X. Q. Wang, G. S. Qi, J. Wang and W. Li, Appl. Catal., B, 2013, 134–135, 251–257 CrossRef CAS.
- H. Hu, S. X. Cai, H. R. Li, L. Huang, L. Y. Shi and D. S. Zhang, J. Phys. Chem. C, 2015, 119, 22924–22933 CAS.
- B. Cui, H. Lin, Y. Z. Liu, J. B. Li, P. Sun, X. C. Zhao and C. J. Liu, J. Phys. Chem. C, 2009, 113, 14083–14087 CAS.
- F. X. Ma, L. Yu, C. Y. Xu and X. W. (David) Lou, Energy Environ. Sci., 2016, 9, 862–866 CAS.
- Z. Ren, Z. L. Wu, W. Q. Song, W. Xiao, Y. B. Guo, J. Ding, S. L. Suib and P. X. Gao, Appl. Catal., B, 2016, 180, 150–160 CrossRef CAS.
- G. I. Panov, K. A. Dubkov and E. V. Starokon, Catal. Today, 2006, 117, 148–155 CrossRef CAS.
- P. Arnoldy and J. A. Moulijn, J. Catal., 1985, 93, 38–54 CrossRef CAS.
- L. Guo, H. Xian, Q. F. Li, D. Chen, Y. S. Tan, J. Zhang, L. R. Zheng and X. G. Li, J. Hazard. Mater., 2013, 260, 543–551 CrossRef CAS PubMed.
- F. Prinetto, G. Ghiotti, I. Nova, L. Lietti, E. Tronconi and P. Forzatti, J. Phys. Chem. B, 2001, 105, 12732–12745 CrossRef CAS.
- N. Tang, Y. Liu, H. Q. Wang and Z. B. Wu, J. Phys. Chem. C, 2011, 115, 8214–8220 CAS.
- M. Mihaylov and K. Hadjiivanov, Langmuir, 2002, 18, 4376–4383 CrossRef CAS.
- B. Azambre, L. Zenboury, A. Koch and J. V. Weber, J. Phys. Chem. C, 2009, 113, 13287–13299 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra23704e |
| This journal is © The Royal Society of Chemistry 2016 |