A novel strategy for low level laser-induced plasmonic photothermal therapy: the efficient bactericidal effect of biocompatible AuNPs@(PNIPAAM-co-PDMAEMA, PLGA and chitosan)†
Alireza Gharatapeab,
Morteza Milaniac,
Seyed Hossein Rastade,
Mohammad Pourhassan-Moghaddamfg,
Sohrab Ahmadi-Kandjanih,
Soodabeh Davaran*i and
Roya Salehi*j
aDepartment of Medical Nanotechnology, School of Advanced Medical Science, Tabriz University of Medical Science, Tabriz, Iran
bStudent Research Committee, Tabriz University of Medical Science, Tabriz, Iran
cInfectious and Tropical Diseases Research Center, Tabriz University of Medical Science, Tabriz, Iran
dSchool of Medical Sciences, University of Aberdeen, Aberdeen AB24 5DT, UK
eDepartment of Medical Bioengineering, Tabriz University of Medical Sciences, Tabriz, Iran
fDepartment of Medical Biotechnology, School of Advanced Medical Science, Tabriz University of Medical Sciences, Tabriz, Iran
gFuture Industries Institute, University of South Australia, Mawson Lakes Campus, Mawson Lakes, South Australia 5095, Australia
hResearch Institute for Applied Physics and Astronomy, University of Tabriz, Tabriz, Iran
iDrug Applied Research Center and Department of Medicinal Chemistry, Faculty of Pharmacy, Tabriz University of Medical Science, Tabriz, Iran. E-mail: davaran@tbzmed.ac.ir
jResearch Center for Pharmaceutical Nanotechnology and Department of Medical Nanotechnology, School of Advanced Medical Science, Tabriz University of Medical Science, Tabriz, Iran. E-mail: salehiro@tbzmed.ac.ir
First published on 15th November 2016
Abstract
Nowadays the control of multidrug resistant (MDR) and pandrug resistant (PDR) bacteria has been the subject of extensive research. In this study an effective strategy was developed to destroy bacteria using low-level laser photothermal therapy combined with biocompatible surface-modified AuNPs. First, chitosan, poly(N-isopropylacrylamide)-co-(2-dimethylamino ethyl methacrylate) P(NIPAAM-co-DMAEMA) and poly(lactic-co-glycolic acid) (PLGA) modified AuNPs were synthesized and fully characterized. Afterwards, P. aeruginosa and A. baumannii (Two Gram-negative strains of bacteria) were exposed to different doses of low level NIR laser (810 nm) radiation in the presence or absence of the as-prepared surface-modified AuNPs (in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) and the killing efficiency of the radiated laser doses was calculated based on pour-plate colony count for each condition. Chitosan, P(NIPAAM-co-DMAEMA) and PLGA modified AuNPs were synthesized with a core/shell size of 108, 10 and 120 nm, respectively which was confirmed with DLS and TEM studies. Successful polymerization and surface coating of AuNPs was confirmed by FT-IR and 1H NMR. A decreasing trend in the viability of both bacteria was observed along with an increase of the laser dose for all three types of polymer-coated AuNPs. PLGA@AuNPs exhibited the most effective NIR-induced photothermal killing on both bacteria. In other words, 10 J cm−2 and 30 J cm−2 doses were enough to destroy almost all P. aeruginosa, and A. baumannii, respectively. Our study suggests the usefulness of low-level laser in plasmonic photothermal treatment. The suggested strategy, as a new method of anti-bacterial intervention, can be used for the eradication of infections such as wound infections in order to accelerate the healing process. In addition, the offered strategy can be suggested in the treatment of other bio-threats such as cancerous diseases in vivo.
1 ratio) and the killing efficiency of the radiated laser doses was calculated based on pour-plate colony count for each condition. Chitosan, P(NIPAAM-co-DMAEMA) and PLGA modified AuNPs were synthesized with a core/shell size of 108, 10 and 120 nm, respectively which was confirmed with DLS and TEM studies. Successful polymerization and surface coating of AuNPs was confirmed by FT-IR and 1H NMR. A decreasing trend in the viability of both bacteria was observed along with an increase of the laser dose for all three types of polymer-coated AuNPs. PLGA@AuNPs exhibited the most effective NIR-induced photothermal killing on both bacteria. In other words, 10 J cm−2 and 30 J cm−2 doses were enough to destroy almost all P. aeruginosa, and A. baumannii, respectively. Our study suggests the usefulness of low-level laser in plasmonic photothermal treatment. The suggested strategy, as a new method of anti-bacterial intervention, can be used for the eradication of infections such as wound infections in order to accelerate the healing process. In addition, the offered strategy can be suggested in the treatment of other bio-threats such as cancerous diseases in vivo.
1. Introduction
Over the last few decades, numerous pathogenic bacterial strains have been resistant to several antibiotics.1–3 There is a requirement to develop a new generation of drugs for overcoming multi-drug resistance (MDR) for health systems worldwide.Photothermal therapy is a minimally invasive method that can induce optical hyperthermia which could be used in clinical treatment to destruct cells.4,5 Single continuous wave (Cw) and pulsed wave are two different methods of photothermal therapy which are based on converting photon energy to heat. Laser radiation absorption by an absorber nanoparticle induces the increase in temperature of host cell, tissue and nearby microenvironment. The laser induced heat destruct the target cell through induction of protein denaturation, cell cavitation, cell rupturing, bubble formation, DNA fragmentation.6–8
Near-infrared (NIR) waves have been used for developing novel theranostic-based methods. Two ranges of NIR, namely (650–950 nm and 1000–1350 nm), are the most important in clinical therapies.9 NIR have been widespreadly used in various applications from sensing to controlled-drug delivery systems. This range of electromagnetic wavelength can penetrate in deep tissue and excite all kind of NIR-activate nanomaterials. When the surface electrons of the nanomaterial are excited by NIR radiation, they quickly lose energy and back down to their stationary state. These process of losing energy is conducted by electron–electron collision.10 NIR-photothermal therapy is emerging theranostic field that can combine via chemotherapy and photodynamic therapy.11,12
Several different nanomaterials, such as gold nanoparticles, quantum dots, carbon nanotubes and graphene oxides have been used as photothermal agents alone or in combination. Among them gold nanoparticle is superior due to unique properties, including biocompatibility, photonic and plasmonic properties and its suitability as carrier in drug and gene delivery.8,13
Low-level laser therapy (LLLT or low-power laser therapy) is application of a low-level laser that emits specific wavelengths of visible light or infrared range which can penetrate deeply into human tissue for therapeutic purposes. In this process no heating-induced damage occurs to the tissue. LLLT does not have any known side effects and even it can be effective to accelerate wound healing and to reduce pain.14,15 Recently, LLLT was used in dentistry16 and joint disorders.17
Antibody or polymer-coated gold nanoparticles – of different size and shape – are used in plasmonic photothermal therapy (PPTT) and photodynamic therapy (PDT).18–22 Moreover, plasmonic photothermal therapy have been successfully demonstrated to kill various bacteria such as Salmonella spp.,23 S. aureus,24 S. epidermidis,25 E. coli,26 E. faecalis.27
It was observed that gold nanoparticles can interact specifically with compounds containing thiol or amine groups.28,29 Chitosan (CS) is a natural polysaccharide derived from chitin which is full of surface dangling amine groups.30 Biocompatibility, biodegradability, hydrophilicity and cell membrane penetration ability makes CS as an excellent biomaterial for delivery of molecules and serving as surface modification agent.31
PNIPAAM is temperature-responsive hydrophilic polymer which could be copolymerized via other monomers such as DMAEMA to synthesize pH- and temperature-sensitive polymer containing of several amine groups.32
PLGA is a polyester copolymer approved by FDA used as encapsulating biomaterial for delivery of DNA, protein, drug and other compounds. Thiolated gold nanoparticles with terminated hydroxyl groups could be polymerized with lactide and glycolide monomers via ring opening polymerization to produced PLGA coated gold nanoparticles.33,34
In the present work we developed novel bactericidal nanoparticles based on NIR photothermal therapy. A better bactericidal efficiency of gold nanoparticle was observed after surface modification. Acinetobacter baumannii and Pseudomonas aeruginosa that are prevalent in infection and nosocomial infections were used in this study. Gold nanoparticles coated with three polymeric compound chitosan, PLGA and P(NIPAAm-co-DMAEMA) to improve cell internalization and increasing photothermal efficiency. The mentioned modified gold nanoparticles could be used to overcome misuse of broad-spectrum antibiotics by induce biological destruction via hyperthermia with a low power NIR laser.
The physicochemical characterization of engineered gold nanoparticles was studied by transmission electron microscopy (TEM), dynamic light scattering (DLS), UV-visible (UV-vis) spectroscopy, Fourier transform infrared spectroscopy (FTIR) and nuclear magnetic resonance (NMR) spectroscopy. The growth of bacteria after photothermal treatment monitored through colony forming unit (CFU) method.
2. Experimental
2.1. Materials and methods
The following analytical-grade chemicals obtained from the mentioned companies and applied without purification: medium molecular weight chitosan (ACROS organic), 2-mercaptoethanol (99%, extra pure, ACROS Organics), glycolic acid (99%, ACROS Organics), DL-lactic acid (85%, ACROS Organics), NIPAAM (97%, Aldrich), DMAEMA (98%, Aldrich), tin(II) 2-ethylhexanoate, Sn(Oct)2 (Aldrich), Tween 20 (Sigma-Aldrich), gold(III) chloride trihydrate, HAuCl4 (≥99.9%, Aldrich), trisodium citrate dehydrate, Na3C6H5O7·2H2O (Merck), nutrient agar (Merck), nutrient broth (Merck), ammonium peroxodisulfate, (NH4)2S2O8 (Merck). All other accessible solvent and reagent were purchase from Sigma-Aldrich Co. water was deionized and purified to a resistance of 18 MΩ. All of glassware wash-downs with aquaregia solution (HCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HNO3; 3
HNO3; 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) firstly, followed by extensive washing with double-deionized water.
1) firstly, followed by extensive washing with double-deionized water.
2.2. Preparation of Au nanoparticles
The Au nanoparticles were synthesized by following a citrate reduction process (Turkevich method). In this process, 100 mL of HAuCl4·3H2O (1 mM) was stirred on heater stirrer (Heidolph Instruments, Germany) at the boiling temperature. Then, 0.12 mg of trisodium citrate dehydrates dissolved in 6 mL deionized water with resistivity of 18 MΩ cm and suddenly added to the above solution under vigorous stirring. Heating was continued and solution color was changed from golden to wine red. Finally stirring was continued till the mixture temperature reached to room temperature. The as prepared AuNPs were stored in dark at 4 °C until used.2.3. Preparation of AuNPs@chitosan
200 mg of medium molecular weight chitosan dissolved in 2% acetic acid solution (20 mL) under 750 rpm stirring in 70–75 °C. Afterwards 20 mL of AuNPs solution added to stock solution of chitosan and the mixture stirred at 900 rpm. After 30 min color of solution was changed to pink and two hours later wine red color was observed. The reaction was continued for 24 h in order to obtain AuNPs@chitosan. Unreacted materials were removed.2.4. Preparation of AuNPs@P(NIPAAM-co-DMAEMA)
AuNPs concentrations which were calculated by Beer's law were 4.1 nM. 1 mL of Tween 20 (10% v/v) was added to 9 mL of citrate-capped AuNPs solution and mixture stirred at room temperature overnight. In the next step 0.01 mM of DMAEM was added to Tween 20–AuNPs with a stirring speed of 500 rpm for overnight.Next, 0.01 mM of NIPAAM was added to the pervious solution and stirred for overnight. Finally the mixture was degassed with nitrogen for 10 min and then 3 μg of ammonium persulfate was added to above mixture (as initiator) and the reaction was continued under nitrogen flow for 24 h. The obtained P(NIPAAM-co-DMAEMA)@AuNPs were stored at 4 °C in the dark.
2.5. Preparation of PLGA@AuNPs
1 mL of 2-mercaptoethanol was added to 10 mL of Tween 20–AuNPs solution and stirred overnight (300 rpm) at ambient temperature. Afterward thiolated AuNP solution was dried for 36 h in the vacuum drying oven at 53 °C. Then 0.2 g of lactide and glycolide (each one) was added to thiolated AuNP powder and heated to 90–100 °C for melting of monomers. The mixture was exposed to argon flow for 10 min and then 0.5% wt of Sn(Oct)2 was added to the mixture and the polymerization was continued at 120 °C under argon flow and stirring at 750 rpm for overnight. Finally PLGA@AuNPs was stored at 4 °C in the dark.2.6. Bacteria culture
In this study, standard stain of Acinetobacter baumannii (ATCC 19606) and Pseudomonas aeruginosa (IPM-2) have been used to assess the antimicrobial effect of modified gold nanoparticles based on photothermal therapy. Both strains were cultured in nutrient broth medium and incubated at 35 °C for 24 hour. When the turbidity of the inoculum was equivalent to 0.5 McFarland standards (1.5 × 108 CFU mL−1), stock solution were stored at 4 °C for the next photothermal treatment.2.7. Photothermal bactericidal property of modified AuNPs
The cultured (A. baumannii and P. aeruginosa) strain were washed twice with PBS solution (pH 7.0), collected by centrifugation at 6000 rpm for 10 min and diluted to reach 1.5 × 106 CFU mL−1 250 μL of diluted bacteria inoculum mixed with 250 μL of surface-modified AuNPs in the wells of a 24-well plate individually. The solution vibrated for 3 min and incubated for 2 h at room temperature. Afterwards, NIR laser irradiation (Diode laser Mustang 2000, Russia) applied to mixture with different exposure time at 810 nm.Physical parameter of laser such as power density, spot size, energy density, and output power and exposure time have been calculated with the following formulas:
In this study, 240 mW laser powers with 810 nm wave length was used in order to induce photothermal effects on modified gold nanoparticles. Spot size was calculated according to the circular area that was 2 cm2. Intensity or power density was measured from the following formula (I) and dose or energy density was calculated from formula (II)
| 107*(mW)/2 (cm2) = 53.5 (mW cm−2) | (I) |
| Dose (J cm−2) = power density (W) × time (s) | (II) |
A negative control was included in the experiment without incubation with the nanoparticles. All experiments were done in triplicate.
2.8. Pure plate culture
After irradiation of sample solutions, the content of each well was transferred to a new 25 mL liquid nutrient agar plate and incubated at 37 °C for 24 h. After bacterial growth, colony numbers were calculated.2.9. Characterization
UV-vis spectroscopy of nanoparticles was done using Cecil BioAquarius CE 7250 Double Beam Spectrophotometer (Bio Aquarius, Cecil, UK). Particle size and zeta-potential of modified gold nanoparticles were measured by DLS technique using Nano-ZS, Malvern Instruments (Malvern, UK). All of 1H NMR Spectra were recorded with a Bruker Ultrashield 400 MHz (Germany) using DMSO as solvent. All FT-IR spectra of modified gold nanoparticle recorded with Bruker-Tensor27® FT-IR Spectrometer equipped with a ZnSe Attenuated Total Reflection (ATR). TEM images of core/shell nanoparticles were acquire with Zeiss-Leo 906 TEM operated at 150 kV acceleration voltages (Leo, Germany). TEM images prepared from the evaporated sample on carbon-coated copper grid at room temp.3. Results and discussion
3.1. Preparation of core–shell chitosan@AuNPs, P(NIPAAM-co-DMAEMA)@AuNPs and PLGA@AuNPs
In this study AuNPs were synthesized by citrate reduction process (Turkevich method) (Fig. 1.s in ESI†) and subsequently coated with three different type polymers to produced core–shell structures. The coating procedure of AuNPs with chitosan, PLGA and P(NIPAAM-co-DMAEMA) polymers are illustrated in Fig. 1. These modifications change the surface charge and reactivity of AuNPs. | ||
| Fig. 1 Schematic illustration of mechanistic and synthesis steps of (chitosan (a), DMAEMA-co-NIPAAM (b), PLGA (c))-coated AuNPs. | ||
3.2. Characterization of chitosan@AuNPs
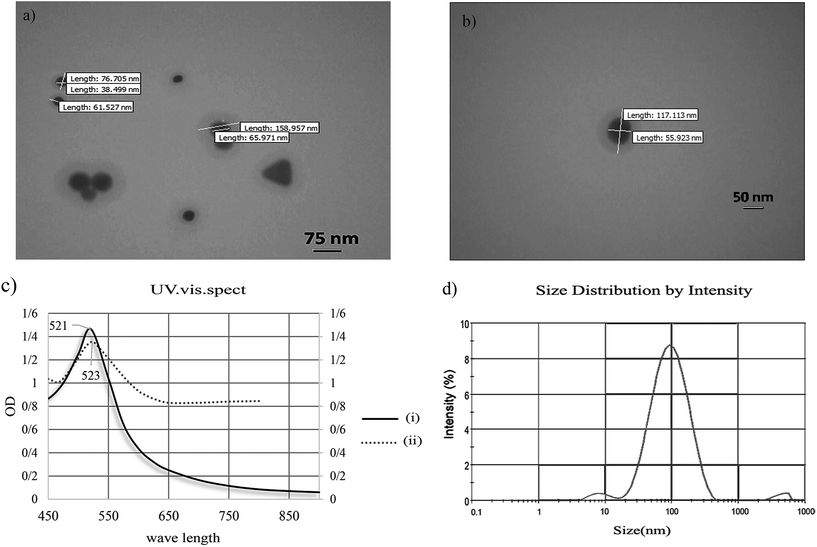
UV-visible spectroscopy results of chitosan@AuNPs showed an absorbance peak at 523 nm. DLS size distribution of 108 nm and average zeta-potential of +17.8 mV for the as-prepared chitosan@AuNPs, indicating high stability of the nanoparticles and the possibility of electrostatic interaction with bacteria membrane. TEM images demonstrated core/shell structure of chitosan@AuNPs, in which, average size of AuNPs core and polymer shell was around 38–65 nm and 38–90 nm, respectively. The whole polymer-coated AuNPs diameter was around 108 nm (Fig. 2).Furthermore, FT-IR analysis proves the formation of polymer-coated AuNPs. More details on FT-IR spectra and Z-potential are found in Fig. 2.s and 3.s (in ESI†), respectively.
3.3. Characterization of P(NIPAAM-co-DMAEMA)@AuNPs
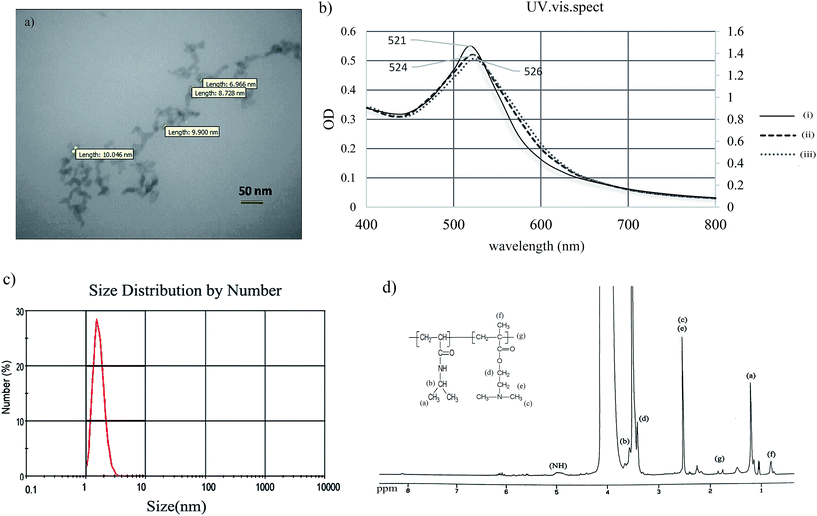
Nucleophilic substitution reaction occurred between two NIPAAM and DMAEMA monomers on AuNPs surface lead to formation of P(NIPAAM-co-DMAEMA)@AuNPs. UV-visible absorption for unmodified gold nanoparticles was 521 nm but when the polymerization occurred on the AuNPs surface, the maximum absorption of resultant P(NIPAAM-co-DMAEMA)@AuNPs red shifted to 526 nm (Fig. 3b). DLS analysis demonstrated average particle size of about 1.6 nm (Fig. 3c). According to the TEM images, polymer-coated AuNPs revealed chained core/shell morphology with core size less than 5 nm. As the original size of synthesized AuNPs without coating in this work were about 20 nm, maybe the possible reason for the small sizes of Au-cores observed in TEM, was due to the additional reduction of gold nanoparticles during polymerization process which, making them smaller than their original size. Z-Potential of the P(NIPAAM-co-DMAEMA)-modified AuNPs was about −0.6 mV which was more neutral in comparison with the bare AuNPs (Fig. s.4†).1H NMR and FT-IR spectra of P(NIPAAM-co-DMAEMA)@AuNPs (Fig. 3d and s.5†) confirmed the successful polymerization of monomers. Characteristic signals of PNIPAAm moiety were appeared at 1.1 (CH3, NIPAAM (a)), 3.9 (CHCH3CH3, NIPAAM (b)) and 1.74 (CH–CH3, NIPAAM (g)). Typical signals of PDMAEMA moiety of copolymer were revealed at 1.83 (CH2CH3, (g)), 2.6 (CH2NHCH3CH3) (e and c) and 3.6 (OCH2CH2, (d)).
3.4. Characterization of PLGA@AuNPs
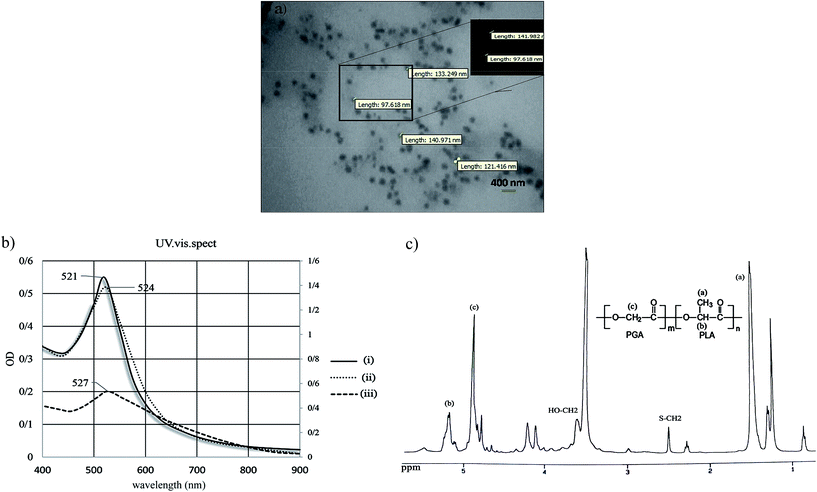
PLGA@AuNPs was synthesized by ring opening polymerization of lactic and glycolic acid that occurred on 2ME-modified AuNPs. UV-vis spectroscopic analysis showed when the 2-mercaptoethanol was reacted with AuNPs (in 10% Tween-20), 6 nm red shift was occurred (Fig. 4b). TEM images showed core/shell structure of PLGA@AuNPs with size range of 90–140 nm (Fig. 4a). The average particles diameter of core and shell size was about 40 and 60 nm, respectively. Possibly FT-IR analysis confirmed modification of AuNPs with PLGA (Fig. s.6†). Also, 1H NMR spectra of PLGA@AuNPs revealed multiplet peaks in which the signals pertaining to lactide moiety were found in 1.48 ppm (OCH(CH3)CO (a), and 5.2 ppm (OCH(CH3)CO (b)). The signal pertaining to glycolide was found in 4.8 ppm (OCH2CO (c)). The presence of ME moiety of polymer was proved by signals appeared at 2.5 S–CH2) and 3.6 ppm (corresponding OCH2) (Fig. 4c).3.5. Bactericidal efficiency of polymer-coated AuNPs based on photothermal therapy
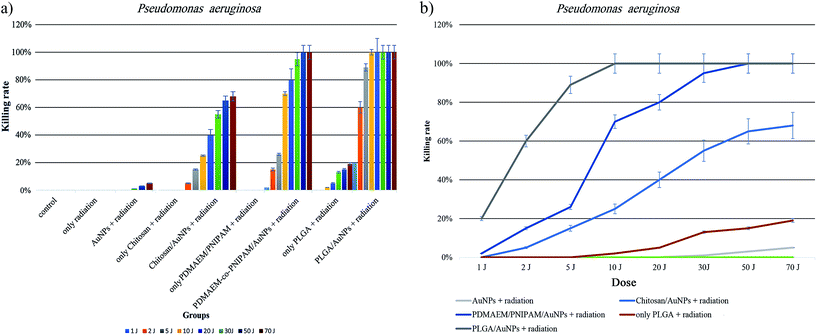
Several controls were included to ensure the specific role of polymer-coated AuNPs photothermal killing under 810 nm continues NIR laser treatment. Using low level laser radiation allow us to propose a safe and harmless laser radiation which can be developed into in vivo studies for eradication of wound infections and other diseases such as cancer. In these experiments, multiple laser exposure times have been tested. Although, 810 nm Mustang's probe has 240 mW laser exposure, it will be less when the nozzles added to the probe for regulating laser beam diameter on spot size.The viability of NIR-treated P. aeruginosa (without AuNPs) was similar to the sample with (bacteria) and (bacteria + AuNPs + NIR), indicating no effect of NIR on bacteria in the absence of AuNPs (Fig. 5 and 6a–e). Also, bare AuNPs did not show significant bactericidal effects on P. aeruginosa after irradiation of 70 J cm−2 (5% killing). However samples who received chitosan–AuNPs + NIR radiation exhibited up to 40% and 68% killing efficiency after exposure to 20 J cm−2 and 70 J cm−2, respectively. After NIR irradiation, a close linear relationship was observed between the number of viable bacteria and energy density. In this case decrease in the viability has occurred with increasing the energy density (Fig. 5 and 6f–i). Only chitosan + NIR radiation-treated bacteria was completely survived that means chitosan does not show NIR-induced photothermal destruction of bacteria (Fig. s.7†). In comparison with the chitosan–AuNPs, P(NIPAAM-co-DMAEMA)@AuNPs + NIR radiation could destruct about 70% and 95% of P. aeruginosa after exposure to 10 J cm−2 and 30 J cm−2 radiations, respectively. With power densities of ≥50 J, almost all of the P. aeruginosa was destroyed. A remarkable killing effect observed when the energy density was increased from 5 J cm−2 to 10 J cm−2 (Fig. 5 and 6j–n). Three reasons may be lead to this extraordinary effect. At first, probably, in case of small size AuNPs, lower energy is required for NIR-induced photothermal killing.35 Also, the small size AuNPs can penetrate easier than bigger ones in to the target bacteria which increase their photothermal efficiency. Furthermore, based on TEM images P(NIPAAM-co-DMAEMA)@AuNPs had chain structure which may causes a high number of AuNPs on the bacteria cell wall, loading to their more intense photothermal effects.
In PLGA@AuNPs + NIR radiation treatment group, 5 J cm−2 dose resulted in the bacterial viability of about 11% and when the energy density increased to 10 J cm−2 almost no viability observed (Fig. 5 and 6o–s). Interestingly, in case of the only PLGA + NIR radiation group a reduced viability observed after exposure to 10 J and reached to 19% with 70 J cm−2 exposures (Fig. 5 and s.7†). This result was in agreement with previous study.36 Therefore high efficiency of PLGA@AuNPs in bacteria killing is attributed to the synergistic accumulation of their NIR-induced photothermal effect. Pseudomonas aeruginosa (IPM-2) killing rate was represented in Table 1.
| Group | 0.5 J | 1 J | 2 J | 5 J | 10 J | 20 J | 30 J | 50 J | 70 J |
|---|---|---|---|---|---|---|---|---|---|
| Control | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| Only radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| AuNPs + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 1% | 3% | 5% |
| Only chitosan + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| Chitosan/AuNPs + radiation | 0% | 0% | 5% | 15% | 25% | 40% | 55% | 65% | 68% |
| Only P(NIPAAM-co-DMAEMA) + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| P(NIPAAM-co-DMAEMA) + radiation | 0% | 2% | 15% | 26% | 70% | 80% | 95% | 100% | 100% |
| Only PLGA + radiation | 0% | 0% | 0% | 0% | 2% | 5% | 13% | 15% | 19% |
| PLGA/AuNPs + radiation | 5% | 20% | 60% | 89% | 100% | 100% | 100% | 100% | 100% |
Similar trends of NIR-induced photothermal effect were observed in both A. baumannii and P. aeruginosa isolates, with a higher resistance of A. baumannii. We believe this phenomenon may be related to size and shape of P. aeruginosa in comparison of A. baumannii. This difference causes to adsorption more number of surface-modified gold nanoparticles on P. aeruginosa surface. Because it has larger surface area than A. baumannii37,38 therefore, it can accept more particles per surface. Another factor may be related to heat-resistance of A. baumannii compared to P. aeruginosa.39,40
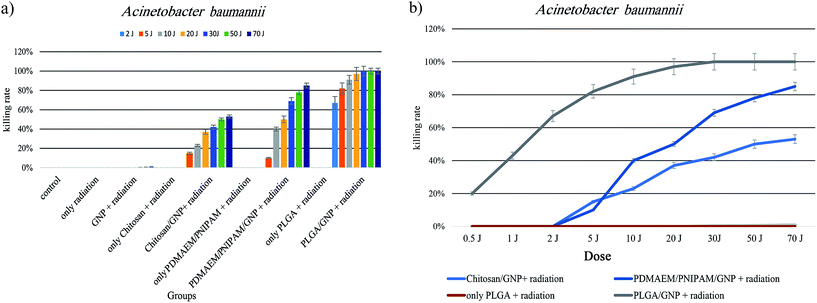
In A. baumannii sample, there was no significant difference in viability of control (only bacteria) group with bare AuNPs + NIR irradiated with energy density of up to 70 J cm−2 (Fig. 7 and 8a–e). Also, none of the polymers (in the pure form) showed NIR-induced photothermal killing of A. baumannii (Fig. 7 and s.8†). This finding suggests that this bacteria isolate is more resistant to NIR-induced photothermal killing in comparison to P. aeruginosa. The reason maybe related to the smaller surface area of A. baumannii and more heat-resistance effect of it, which mentioned above. A near 50% killing was observed in A. baumannii treated with chitosan@AuNPs + NIR radiation after exposure to 50–70 J cm−2 radiations (Fig. 7 and 8f–j). Whereas the killing rate in P. aeruginosa isolate was around 68%. AuNPs@P(NIPAAM-co-DMAEMA) + NIR radiation treatment group displayed weaker effect in 5 J cm−2 radiations than chitosan@AuNPs + NIR radiation group, however, the sharp rise appeared when the laser dose was increased to 10 J cm−2. Accordingly, when the samples were irradiated with 20 J cm−2 and 70 J cm−2 doses, mortality rate reached to 50% and 85% respectively (Fig. 7 and 8k–o). Also killing rate of Acinetobacter baumannii (ATCC-19606) presented in Table 2.
| Group | 0.5 J | 1 J | 2 J | 5 J | 10 J | 20 J | 30 J | 50 J | 70 J |
|---|---|---|---|---|---|---|---|---|---|
| Control | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| Only radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| GNP + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0.40% | 0.60% | 1% |
| Only chitosan + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| Chitosan/GNP + radiation | 0% | 0% | 0% | 15% | 23% | 37% | 42% | 50% | 53% |
| Only P(NIPAAM-co-DMAEMA) + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| P(NIPAAM-co-DMAEMA)/GNP + radiation | 0% | 0% | 0% | 10% | 40% | 50% | 69% | 78% | 85% |
| Only PLGA + radiation | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
| PLGA/GNP + radiation | 20% | 43% | 67% | 82% | 91% | 97% | 100% | 100% | 100% |
Among these three polymer-modified AuNPs, PLGA@AuNPs + NIR radiation group was the most sensitive one to NIR-induced photothermal killing in which survival rate of bacteria reached 20 and 5% after receiving radiation dose of 5 and 20 J cm−2, respectively. Whereas PLGA + NIR radiation group did not show any killing effect on A. baumannii (Fig. 8 and s.8†) (Fig. 8p–t).
Results indicated that bare AuNPs does not significantly destruct bacteria through photothermal therapy which was in agreement with other studies.41 Three polymers; chitosan, PLGA and P(NIPAAM-co-DMAEMA) have been selected nanoparticles surface driven by electrostatics interaction. Chitosan is cationic, biodegradable and biocompatible polymer,42 PLGA has photothermal activity and anionic polymer36 and P(NIPAAM-co-DMAEMA) is biocompatible and temperature-sensitive amphiphilic copolymer which is full of amine groups that can facilitate cell internalization.43,44 Prior studies on PLGA have reported it has partly photothermal effects. The mobility of PLGA molecules under Tg temperature (glass transition temperature) is relatively low, but it is increased with temperature higher than Tg. Local temperature originated from NIR or other external source causes environmental temperature exceeds the Tg of PLGA and photothermal effects are appeared.36
The bactericidal properties of different polymer-coated AuNPs irradiated with NIR compared with other groups (only bare AuNPs, only laser, AuNPs free polymer + NIR, bare AuNPs + NIR). The viability of the bacteria tested (A. baumannii and P. aeruginosa) under NIR treatment were calculated, considering by different energy densities (1, 2, 5, 10, 20, 30, 50, and 70 J cm−2) in continues mode.
Since a few research published about efficacy of photothermal therapy against A. baumannii and P. aeruginosa isolates, and on the other hand, the increasing antibiotic resistance, this study was conducted to evaluate the antibacterial effects of different novel polymer-modified AuNPs as photothermal agents, exposed to low level laser (Mustang 2000, Ga–As diode laser) to kill the bacteria isolates efficiently. Although the received doses and energy level are limited in the suggested method, the coherences effect of the used laser photons are enough to induce photothermal effects due to synergistic effects and higher energy absorption in our developed nanoparticles. In addition to laser type, nanoparticle/bacteria concentration ratio and other parameters such as effective radiation dose are critical factors to inducing photothermal effect. From mechanistic point of view, different shape of AuNPs started to melt at power density ranging from 15–25 mW cm−2 after 15 min exposure time (equal: 13.5–22.5 J cm−2).35
Norman and colleagues, 2008, developed antibody conjugated gold nanorods (GNR) for photothermal therapy treatment against P. aeruginosa isolate. After synthesized GNR they used two layers of polymers (anionic poly(acrylic acid) (PAA) and cationic polyallylamine hydrochloride (PAH)) to provide terminal amine groups for antibody immobilizing. Next, NIR laser (785 nm; 50 mW–10 min) – equivalent to 30 J – was used to irradiate GNRs and bacterial cell viability was counted. Their results were demonstrated that the cell viability of control cells, cells + NIR and cells + GNRs were nearby 80% while the viability of case group who received GNRs + cells + NIR had been decreased to 20%. Whereas, our results shown a near 100% killing by AuNPs-decorated P(NIPAAM-co-DMAEMA) and AuNPs@PLGA. Also a main limitation of the above mentioned study is focusing on only one laser power; however, in our study various laser conditions have been tested in order to optimize the PTT effect of surface bioengineered AuNPs. Thereupon previous studies are in accordance with our efforts.45
In a recent study, Teng and coworkers, 2016, were studied photothermal ablation of P. aeruginosa bacteria with gold nanocrosses. Multibranched gold nanocrosses were synthesized and conjugated with primary antibodies to attach P. aeruginosa, followed by irradiating with NIR laser (800 nm, 3.0 W cm−2 power density, 5 min exposure time) to assess the photothermal effects on bacteria killing. Their results demonstrated complete bacteria destruction. In our study, a new strategy was developed in which low level laser for PTT of surface engineered AuNPs with different polymers was used. Although, the size of gold nano-cross were bigger in compare with our particles, similar killing efficiencies was observed, confirming the usefulness of low power laser photothermal therapy using small AuNPs. It seems that the surface engineering of AuNPs, with charged polymers could increase the interaction of AuNPs with bacteria surface.46
Meziani and colleague, on 2016, synthesized 2,2′-(ethylenedioxy)bis(ethylamine) (EDA) functionalized carbon quantum dots with particles size of 5 nm and their photo-induced bactericidal effect were investigated on E. coli. Their results demonstrated that even though, the initiated optical density of bacteria solution was 1.0, after 1 h it reached to 1.3 in the untreated group while, optical density of bacteria solution in treated groups with different amount of EDA–carbon dot reach to <0.3. Also, pour plate tests showed almost all of bacteria died after 6 h treatment. But effect of different energy densities (increase or decreases) on killing rate was not discussed47 while our study discusses on the effect of energy densities with several experiments as well as in our developed methods complete bacteria killing took place very quickly than mentioned developed carbon quantum dots.
As a result novel developed polymer-modified gold nanospheres in this study could significantly enhance the photothermal effect of as-prepared modified AuNPs and suggested a novel strategy for application of AuNPs in PTT.
4. Conclusion
This study has demonstrated integration of next-generation AuNPs–polymer nanomaterials with low level laser exposure as highly efficient, simple and rapid strategy for destruction of pathogenic bacteria. Three different biocompatible, biodegradable and thermo-sensitive polymer-coated AuNPs were used in order to prove their potential in NIR-induced photothermal therapy. In this system, NIR laser (810 nm) with several different power densities was used. Low-level laser class was used to induce photothermal effect because it has been approved by FDA as safe laser for clinical purposes (used in dentistry and joint disorder). In some conditions, killing rate of bacteria was almost 100%, while untreated groups and control samples did not show any reduction in viability. Additional research will be required to optimize particle size and specific recognition of pathogenic bacteria. Our results proves that the suggested strategy can be used as an principle in designing photothermal-based treatments for controlling various types of pathogenic bacteria.Conflict of interest
The authors report no conflicts of interest.Acknowledgements
This article was written based on a dataset of MSc thesis, registered in Tabriz University of Medical Sciences and financially supported by Drug Applied Research Center (Grant No. 94/2-2/3).References
- A. Tomasz, N. Engl. J. Med., 1994, 330, 1247–1251 CrossRef CAS PubMed.
- J. Davies and D. Davies, Microbiol. Mol. Biol. Rev., 2010, 74, 417–433 CrossRef CAS PubMed.
- H. Nikaido, Annu. Rev. Biochem., 2009, 78, 119 CrossRef CAS PubMed.
- E. B. Dickerson, E. C. Dreaden, X. Huang, I. H. El-Sayed, H. Chu, S. Pushpanketh, J. F. McDonald and M. A. El-Sayed, Cancer Lett., 2008, 269, 57–66 CrossRef CAS PubMed.
- S. Hwang, J. Nam, S. Jung, J. Song, H. Doh and S. Kim, Nanomedicine, 2014, 9, 2003–2022 CrossRef CAS PubMed.
- J. R. Melamed, R. S. Edelstein and E. S. Day, ACS Nano, 2015, 9, 6–11 CrossRef CAS PubMed.
- X. Huang and M. A. El-Sayed, Alexandria Journal of Medicine, 2011, 47, 1–9 CrossRef.
- L. Cheng, C. Wang, L. Feng, K. Yang and Z. Liu, Chem. Rev., 2014, 114, 10869–10939 CrossRef CAS PubMed.
- D. Jaque, L. M. Maestro, B. Del Rosal, P. Haro-Gonzalez, A. Benayas, J. Plaza, E. M. Rodriguez and J. G. Sole, Nanoscale, 2014, 6, 9494–9530 RSC.
- B. Schütte, M. Arbeiter, T. Fennel, G. Jabbari, A. Kuleff, M. Vrakking and A. Rouzée, Nat. Commun., 2015, 6, 8596 CrossRef PubMed.
- Z. Fan, P. P. Fu, H. Yu and P. C. Ray, J. Food Drug Anal., 2014, 22, 3–17 CrossRef CAS PubMed.
- W. Li, J. Peng, L. Tan, J. Wu, K. Shi, Y. Qu, X. Wei and Z. Qian, Biomaterials, 2016, 106, 119–133 CrossRef CAS PubMed.
- P. C. Ray, S. A. Khan, A. K. Singh, D. Senapati and Z. Fan, Chem. Soc. Rev., 2012, 41, 3193–3209 RSC.
- A. N. Gam, H. Thorsen and F. Lønnberg, Pain, 1993, 52, 63–66 CrossRef CAS PubMed.
- W. Posten, D. A. Wrone, J. S. Dover, K. A. Arndt, S. Silapunt and M. Alam, Dermatol. Surg., 2005, 31, 334–340 CrossRef CAS PubMed.
- J. D. Carroll, M. R. Milward, P. R. Cooper, M. Hadis and W. M. Palin, Dent. Mater., 2014, 30, 465–475 CrossRef PubMed.
- F. Ahrari, A. S. Madani, Z. S. Ghafouri and J. Tunér, Laser. Med. Sci., 2014, 29, 551–557 CrossRef PubMed.
- M. A. Mackey, M. R. Ali, L. A. Austin, R. D. Near and M. A. El-Sayed, J. Phys. Chem. B, 2014, 118, 1319–1326 CrossRef CAS PubMed.
- H.-W. Yang, H.-L. Liu, M.-L. Li, I.-W. Hsi, C.-T. Fan, C.-Y. Huang, Y.-J. Lu, M.-Y. Hua, H.-Y. Chou and J.-W. Liaw, Biomaterials, 2013, 34, 5651–5660 CrossRef CAS PubMed.
- W. I. Choi, J.-Y. Kim, C. Kang, C. C. Byeon, Y. H. Kim and G. Tae, ACS Nano, 2011, 5, 1995–2003 CrossRef CAS PubMed.
- X. Huang, I. H. El-Sayed, W. Qian and M. A. El-Sayed, J. Am. Chem. Soc., 2006, 128, 2115–2120 CrossRef CAS PubMed.
- W.-S. Kuo, Y.-T. Chang, K.-C. Cho, K.-C. Chiu, C.-H. Lien, C.-S. Yeh and S.-J. Chen, Biomaterials, 2012, 33, 3270–3278 CrossRef CAS PubMed.
- S. Wang, A. K. Singh, D. Senapati, A. Neely, H. Yu and P. C. Ray, Chem.–Eur. J., 2010, 16, 5600–5606 CrossRef CAS PubMed.
- R. Shokri, M. Salouti and R. S. Zanjani, J. Microbiol., 2015, 53, 116–121 CrossRef CAS PubMed.
- K. C. Black, T. S. Sileika, J. Yi, R. Zhang, J. G. Rivera and P. B. Messersmith, Small, 2014, 10, 169–178 CrossRef CAS PubMed.
- W. C. Huang, P. J. Tsai and Y. C. Chen, Small, 2009, 5, 51–56 CrossRef CAS PubMed.
- O. Khantamat, C.-H. Li, F. Yu, A. C. Jamison, W.-C. Shih, C. Cai and T. R. Lee, ACS Appl. Mater. Interfaces, 2015, 7, 3981–3993 CAS.
- M. B. Haddada, J. Blanchard, S. Casale, J.-M. Krafft, A. Vallée, C. Méthivier and S. Boujday, Gold Bull., 2013, 46, 335–341 CrossRef.
- S. Perumal, Mono- and multivalent interactions between thiol and amine ligands with noble metal nanoparticles, Freie Universität Berlin, 2012 Search PubMed.
- C. Pillai, W. Paul and C. P. Sharma, Prog. Polym. Sci., 2009, 34, 641–678 CrossRef CAS.
- T. Kean and M. Thanou, Adv. Drug Delivery Rev., 2010, 62, 3–11 CrossRef CAS PubMed.
- B. Wang, X.-D. Xu, Z.-C. Wang, S.-X. Cheng, X.-Z. Zhang and R.-X. Zhuo, Colloids Surf., B, 2008, 64, 34–41 CrossRef CAS PubMed.
- S. R. Abulateefeh, S. G. Spain, K. J. Thurecht, J. W. Aylott, W. C. Chan, M. C. Garnett and C. Alexander, Biomater. Sci., 2013, 1, 434–442 RSC.
- H. K. Makadia and S. J. Siegel, Polymers, 2011, 3, 1377–1397 CrossRef CAS PubMed.
- Y. Wang, K. C. Black, H. Luehmann, W. Li, Y. Zhang, X. Cai, D. Wan, S.-Y. Liu, M. Li and P. Kim, ACS Nano, 2013, 7, 2068–2077 CrossRef CAS PubMed.
- W.-L. Chiang, T.-T. Lin, R. Sureshbabu, W.-T. Chia, H.-C. Hsiao, H.-Y. Liu, C.-M. Yang and H.-W. Sung, J. Controlled Release, 2015, 199, 53–62 CrossRef CAS PubMed.
- L. C. Antunes, P. Visca and K. J. Towner, Pathog. Dis., 2014, 71, 292–301 CrossRef CAS PubMed.
- M. N. A. Bhawsar and M. Singh, Int. J., 2014, 2, 778–783 Search PubMed.
- A. Tsuji, Y. Kaneko, K. Takahashi, M. Ogawa and S. Goto, Microbiol. Immunol., 1982, 26, 15–24 CrossRef CAS PubMed.
- S. Sengupta, P. Kumar, A. Ciraj and P. Shivananda, Burns, 2001, 27, 140–144 CrossRef CAS PubMed.
- G. Zhang, X. Sun, J. Jasinski, D. Patel and A. M. Gobin, J. Nanomater., 2012, 2012, 5 Search PubMed.
- C. Peniche, W. Argüelles-Monal, H. Peniche and N. Acosta, Macromol. Biosci., 2003, 3, 511–520 CrossRef CAS.
- T. Kelf, V. Sreenivasan, J. Sun, E. Kim, E. Goldys and A. Zvyagin, Nanotechnology, 2010, 21, 285105 CrossRef CAS PubMed.
- C.-W. Lo, Y. Chang, J.-L. Lee, W.-B. Tsai and W.-S. Chen, PLoS One, 2014, 9, e97627 Search PubMed.
- R. S. Norman, J. W. Stone, A. Gole, C. J. Murphy and T. L. Sabo-Attwood, Nano Lett., 2008, 8, 302–306 CrossRef CAS PubMed.
- C. P. Teng, T. Zhou, E. Ye, S. Liu, L. D. Koh, M. Low, X. J. Loh, K. Y. Win, L. Zhang and M. Y. Han, Adv. Healthcare Mater., 2016, 5, 2122–2130 CrossRef CAS PubMed.
- M. J. Meziani, X. Dong, L. Zhu, L. P. Jones, G. E. LeCroy, F. Yang, S. Wang, P. Wang, Y. Zhao and L. Yang, ACS Appl. Mater. Interfaces, 2016, 8, 10761–10766 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra23213b |
| This journal is © The Royal Society of Chemistry 2016 |