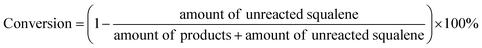The production of bio-jet fuel from Botryococcus braunii liquid over a Ru/CeO2 catalyst
Kun Zhang,
Xu Zhang* and
Tianwei Tan
National Energy R&D Center for Biorefinery, College of Life Science and Technology, Beijing University of Chemical Technology, Beijing 100029, China. E-mail: zhangxu@mail.buct.edu.cn
First published on 11th October 2016
Abstract
In this study, the conversion from Botryococcus braunii liquid to bio-jet fuel via a Ru/CeO2 catalyst was conducted. At first, novel Ru/CeO2 catalysts were prepared in order to increase the catalytic activity. “Ru/CeO2-alcohol thermal” showed the highest activity: the squalane conversion exceeded 90%, an increase of about 22% in comparison with “Ru/CeO2-impregnation”. When the B. braunii liquid was used as the raw material, no obvious catalytic activity was noticed. Therefore, the influence of impurities in the B. braunii liquid on the Ru/CeO2 reaction were preliminarily analysed, confirming that the pigments (such as β-carotenoid) and the oxygen compounds (such as fatty acids) were the main factors influencing the Ru/CeO2 reaction. Based on these results, the reaction process was optimized: prolonging the reaction time and pretreating the B. braunii liquid with H2. Finally, the production of bio-jet fuel over Ru/CeO2 was successfully achieved.
1. Introduction
Biofuels, as one type of promising, renewable and sustainable energy, could be efficiently converted from biomass via catalytic approaches.1 Algal biomass is one type of common feedstock among a variety of lipid producing resources. This interest is obviously explained by the remarkable diversity of microalgae, which comprise about 800![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 different species of which only 35
000 different species of which only 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 have been described.2
000 have been described.2
Among the known oleaginous microalgae, the green alga Botryococcus braunii (B. braunii) has obtained much attention due to its remarkable ability to accumulate a high content of hydrocarbons in the range of 15–75 wt%.3,4 Talukdar3,5–7 et al. assessed the biofuel production potential of B. braunii and determined that B. braunii is economically feasible for the biofuel production. Further, the biosynthesis of B. braunii liquid in bacteria had also been achieved,8,9 which confirms that the B. braunii liquid is possibility to be a promising alternative in biofuel field.
However, Nagano found that the B. braunii liquid is inappropriate to use as a fuel directly due to its adverse natural properties.10 It is necessary to overcome these disadvantages and some corresponding researches have been conducted. Hidalgo studied the direct trans-esterification of B. braunii liquid with continuous methanol reflux to produce biodiesel fuels.11 Watanabe investigated the pyrolysis of B. braunii, and studied the gasification technology of B. braunii over Ni–Fe/Mg/Al catalysts.12,13 Garciano prepared saponite supported cobalt–molybdenum catalysts for upgrading B. braunii liquid.14 Among these researches, most were performed for manufacture of gasoline and diesel fuels, only few were performed towards bio-jet fuel.15 But generally, bio-jet fuel has a greater economic value than gasoline or diesel fuels, which is an attraction point for more and further investigation.
NExBTL and UOP processes (belong to hydro-process) are currently representative commercial processes for the production of bio-jet fuel from non-edible oil, which includes two processes: deoxygenation and isomerization.16–19 Most of the existing works about B. braunii liquid to bio-jet fuel were also focused on this process.15 In contrast to other non-edible oil, B. braunii liquid, particularly the purified B. braunii liquid, is much more special: B. braunii liquid is mainly composed of non-oxygenated hydrocarbons, and the molecular structures of these hydrocarbons are much branched. So the deoxygenation and isomerization actually are undesirable in the case of B. braunii liquid. In this premise, Oya et al. studied the catalytic production process of branched small alkanes from squalane (a B. braunii liquid substitute). Several catalysts (Pd/C, Rh/C, Pt/C, Ru/C, Ir/SiO2, Ru/CeO2 and Ru/SiO2) were compared and characterized. The Ru/CeO2 catalyst showed the highest activity in the same conditions (240 °C, 3.5 MPa H2), the CH2–CH2 bonds located between the branches were selectively dissociated and the isomerization did not happen.20 The Ru/CeO2 catalyst showed an outstanding catalytic potential on the squalane hydrocracking process: high specificity and mild reactive conditions (compared with Pt–Re/SiO2–Al2O3, 330 °C, 5.5 MPa H2).15
However, there are still some problems which restrict the development of B. braunii liquid hydrocracking processes: (1) the commercial squalane, rather than the real B. braunii liquid, was the only used raw material in the Ru/CeO2 reaction, whether the Ru/CeO2 catalyst could work on the B. braunii liquid is not confirmed; (2) the conversion of squalane over impregnated Ru/CeO2 catalyst was too low, only about 70% at four hours. The activity of Ru/CeO2 catalyst needs to be improved.
For the purpose of achieving the production of bio-jet fuel from B. braunii liquid over Ru/CeO2 catalyst, these two problems must be studied and solved. In our work, novel Ru/CeO2 catalysts were prepared (precipitation, impregnation and alcohol thermal) in order to increase the activity of Ru/CeO2 catalyst. Then, the influences of impurities in B. braunii liquid on the reaction were preliminarily analysed for the Ru/CeO2 catalyst almost having no effect on the B. braunii liquid. Finally, based on the results of measurements, the reaction process was optimized. The production of bio-jet fuel was successfully achieved.
2. Material and methods
2.1. Preparation of catalysts
All the Ru/CeO2 samples used in our work were designed for a 5 wt% Ru load. The “Ru/CeO2-impregnation” catalyst was prepared by using CeO2 (calcined at 450 °C; Sigma Aldrich) and Ru(NO)(NO3)3−x(OH)x (Sigma Aldrich) as the starting materials. The commercial CeO2 was impregnated with aqueous Ru(NO)(NO3)3−x(OH)x. Then after drying at 110 °C for 12 h, the catalyst was heated in a N2 stream at 300 °C for 1 h.20The “Ru/CeO2-alcohol thermal” catalyst was prepared by using RuCl3·xH2O (Aladdin), PVP (K 29–32; Xiya Reagent) and ethylene glycol as the starting materials. The RuCl3·xH2O was mixed with ethylene glycol and then PVP was added into this mixture with vigorous stirring. After the mixture became homogeneous, it was transferred to a round flask, and commercial CeO2 was added in the flask, then heated at 198 °C for 6 h under reflux conditions. After cooling to room temperature, the resulting Ru/CeO2 was collected by centrifugation, then rinsed, and dried.21
The “Ru/CeO2-precipitation” catalyst was prepared by co-precipitation method. Ce(NO3)3·6H2O (Sigma Aldrich) and RuCl3·xH2O were dissolved in distilled water. After vigorous stirring at 60 °C for 0.5 h, an aqueous solution of KOH and H2O2 were added dropwise to the aqueous mixture, and the obtained black suspension was kept stirring at 60 °C for 1 h. After cooling to room temperature, the resulting precipitate was centrifuged, washed, and dried. Finally, the precipitate was calcined in the mixture of H2 and N2 at 450 °C for 2 h.22
The catalyst was regenerated with hexane before reusing. The used catalyst was recovered by: washing Ru/CeO2 catalyst with hexane, removing hexane by centrifugation and drying Ru/CeO2 catalyst at 110 °C.20
2.2. Extraction and purification of B. braunii liquid
A dry process was chosen as the extraction process in our work. Four parts were mainly included: aggregation, lyophilisation, hexane extraction and evaporation of solvent. In the hexane extraction process, 1 g lyophilized B. braunii was soaked in 10 mL of n-hexane for 2 min. Then the hexane phase was separated from the extraction residue and collected in a separate flask. Another portion of n-hexane was added to the residue. The same operation was repeated three times.23Purification of B. braunii liquid was carried out as follows: at first, B. braunii liquid was dissolved with hexane. Then a silica gel column was prepared with pure hexane solvent, and four bed volumes of B. braunii liquid/hexane solution were loaded onto the column. Finally, evaporating the solvent, the obtained liquid was the purified B. braunii liquid (B. braunii liquid-P).10,15
2.3. Reaction of squalane/B. braunii liquid
The hydrocracking reaction was performed in a batch reactor. 4 g squalane (Aladdin) or B. braunii liquid, 0.1 g prepared Ru/CeO2 catalyst, a magnetic spinner, a certain amount of impurities and H2 to a pressure of 3.5 MPa/4 MPa were put in a 50 mL autoclave. The start was measured after 60 min heating to 240 °C and terminated after 4 hours/8 hours.2.4. Analysis of B. braunii liquid/productions
Analysis of B. braunii liquid/productions was performed with GC-MS (Agilent 7890B-5977A) in EI modes. The GC was fitted with a HP-5 and programmed for an initial temperature of 40 °C for 1 min, followed by heating at 10 °C min−1 to 300 °C (held for 20 min). Full scans were acquired from m/z 35 to 550. The conversion of squalane was calculated with the formula shown below.Each data point represents the mean result from three independent experiments. And uncertainties reported are standard deviation, which were determined by replicate experiments.
What is more, the UV-Vis spectrophotometer (UV752N) and the Element Analyser (Elementar Vario EL, CHNSO) were also used to analyse the B. braunii liquid.
3. Results and discussion
3.1. Preparation and application of Ru/CeO2 catalysts
The surface morphology and the active site on the surface are significantly affected by the preparation conditions. As shown in Fig. 1a, the Ru/CeO2 catalysts with different preparation methods had distinct squalane conversions. The order of catalytic activity was “Ru/CeO2-alcohol thermal” > “Ru/CeO2-impregnation” > “Ru/CeO2-precipitation”. The “Ru/CeO2-precipitation” catalyst, regarded as a highly catalyst in ammonia synthesis,22 had a lower activity. The probable reasoning could be determined by referring Shin-ichi Oya.20 It was reported that the catalytic activity of Ru/CeO2 was related to the dispersion of loaded ruthenium particles. In the preparation process, the “Ru/CeO2-precipitation” catalyst was precipitated by KOH, and was calcined in the mixture of H2 and N2. John Regalbuto24 regarded these processes probably make the sample has a lower dispersion, which could causes a lower activity. The catalytic activity of “Ru/CeO2-alcohol thermal” exceeded classical “Ru/CeO2-impregnation” by about 22%, which probably was related to the sizes of loaded ruthenium particles.20As shown in Fig. 1b, like with “Ru/CeO2-impregnation”, the CH2–CH2 bonds located between the branches were also selectively dissociated with “Ru/CeO2-alcohol thermal”. The main products had carbon numbers of 9, 14, 15 and 20. And all the structures of these products were substructures of squalane. But the squalane conversion over “Ru/CeO2-alcohol thermal” catalyst was much higher than over previous “Ru/CeO2-impregnation” (70%). The number of conversion had reached above 90%, and the yield of C8–C16 and C17–C20 were separately more than 60% and 20%.
In order to study the stability of “Ru/CeO2-alcohol thermal” catalyst, the “Ru/CeO2-alcohol thermal” was further investigated by reusing catalyst in four experiments. The catalyst was regenerated with hexane before reusing.20 As shown in Fig. 1c, almost the same results were obtained: even after 5 runs, the conversion was still above 90% (90% → 94% → 93% → 89% → 93%), no obvious decrease of the activity was observed.
By changing the preparation methods of Ru/CeO2 catalyst, the classical advantages (high specificity and mild reactive conditions) of Ru/CeO2 catalyst were reserved, and the activity of Ru/CeO2 catalyst was improved (the squalane conversion was up to 90%). What is more, the stability of the new Ru/CeO2 catalyst had also been proven. The “Ru/CeO2-alcohol thermal” catalyst certainly was chosen as the reactive catalyst in the subsequent B. braunii liquid hydrocracking experiments.
As shown in Fig. 2a, the B. braunii liquid-P conversion reached almost 100%, the yield of C17–C20 hydrocarbons was 40%, and the yield of C8–C16 hydrocarbons was 59%. The C8–C16 value of this result is similar to the C8–C16 value (60%) for squalane. In addition, all products in this reaction were stable saturated hydrocarbons and most of them were branched. These properties are ideal for bio-jet fuel.
The hydrocracking result of B. braunii liquid, as shown in Fig. 2b, was widely different. The major conversion products were C27–C33 hydrocarbons. The content of C8–C16 and C17–C20 hydrocarbons were all very poor. And the obtained C15, C16 and C17 hydrocarbons were all linear chain structure hydrocarbons. These results formed a great contrast with the B. braunii liquid-P results.
3.2. Analysis of the effects of impurities on the Ru/CeO2 catalytic reaction
The Ru/CeO2 catalyst achieved a highly efficient catalytic performance for hydrocracking B. braunii liquid-P, but showed a low efficient catalytic performance for hydrocracking B. braunii liquid. In the purifying process, only 80% of B. braunii liquid was available. This ratio is too low to apply in the practical production process. Therefore, using B. braunii liquid-P as the raw material to produce bio-jet fuel is feasible in technique, but not feasible in industrialization.As shown in Fig. 3, the chemical compositions of B. braunii liquid and B. braunii liquid-P were studied by GC-MS, organic elemental analysis, and UV spectrophotometer analysis. The results of UV spectrophotometer analysis showed that there are lots of pigments in B. braunii liquid, and the main pigments were carotenoids (C40H56). But no pigments were detected in B. braunii liquid-P. The results of organic elemental analysis showed that B. braunii liquid and B. braunii liquid-P all mainly consist of four organic elements: C, H, O, N, which is similar to the petroleum feedstock. But the contents of N and O element in B. braunii/B. braunii liquid-P were different. The result of GC-MS showed that the O element existed in B. braunii liquid mainly comes from fatty acids, such as C16H32O2.
 | ||
| Fig. 3 GC-MS, organic elemental analysis and UV spectrophotometer analysis result of B. braunii liquid and B. braunii liquid-P. | ||
Based on the above measurements, there are mainly three differences between B. braunii liquid and B. braunii liquid-P, the pigments, the content of oxygen element (mainly including fatty acids) and the content of nitrogen element (mainly including amine compounds). Among them, the pigments were mainly made up of C and H elements, which are similar to the elemental compositions of squalane. Therefore, the pigments were not regarded as the effect factor of low activity. In order to better display the effects of those variables, the “Ru/CeO2-impregnation” was chosen as the catalyst. The results were shown in Fig. 4.
In the processes of petroleum cracking, nitrogenous compounds could affect the reactions seriously since having an adverse effect on the acid sites located on the solid acids surfaces. This point could be determined by referring Kazuhisa Murata.15 In their paper, the reaction pathways was postulated, the hydrocracking reaction with solid acids mainly occurs on acid sites. What is more, James G. Speight also mentioned this point in the chapter of denitrogenation refining.25 While in our research, it is surprising that nitrogenous compounds had a lower effect on the reaction process (as shown in Fig. 4a), maybe because the hydrocracking reaction over Ru/CeO2 catalyst was not depending on the acid sites.
As shown in Fig. 4b, the oxygen compounds showed a serious impact on the reaction process. Even at a low content of oxygen compounds, the conversion of squalane still had a sharp declining trend. Combined with the content of oxygen element in B. braunii liquid and B. braunii liquid-P, it was well explained that why the results of B. braunii liquid and B. braunii liquid-P were so divergent. The content of O elements played an important role in the Ru/CeO2 reaction process.
When there are oxygen compounds existed in the reaction process, hydrogen and oxygen easily combine to form water. Therefore, there were two possible reasons to explain the decrease: the effect of H2 and the effect of H2O. As shown in Fig. 4c, as the increase of water, the squalane conversion and the content of C8–C16 showed an increasing and then decreasing trend, with a peak of 90% under the optimum reaction conditions. This is probably because a suitable amount of solvent could decrease the viscosity of raw material and increase the hydrogen solubility,26 which are in favour of the hydrogenolysis process. In addition, even if the entire O element in B. braunii liquid were transformed into water, the content of water was still below the number at the inflection point (shown in Fig. 4c). Therefore, the content of water was not the reason for the low activity.
When there are oxygen compounds existed in the reaction process, especially the unsaturated fatty acids, the functional group in the reaction system is not only a C–C bond, but the unsaturated bond and C–O bond are also included. What is more, the order of bond energy is unsaturated bond < C–O bond < C–C bond. Therefore, before the C–C bond reacted, the unsaturated bond and C–O bond have already reacted and consumed lots of hydrogen,27 which obviously has an effect on the Ru/CeO2 reaction.
3.3. Achievement of B. braunii liquid hydrocracking process over Ru/CeO2
Based on the analysis above, we know that the Ru/CeO2 had a low activity with B. braunii liquid for the reduction of hydrogen caused by oxygen compounds. Pre-treating by H2 and enhancing H2 pressure were deemed to be effective methods for solving this problem.28 As shown in Fig. 5a and b, pre-treating by H2 and enhancing the H2 pressure all showed excellent effects on the mixture (squalane with ethyl oleate). The conversions were all higher than 90% and the content of C8–C16 were all more than 50%. Therefore, it can be argued that pre-treating with H2 and higher H2 pressure all could totally solve the low activity problem caused by oxygen compounds. | ||
| Fig. 5 (a and b) Squalane with ethyl oleate (c) B. braunii liquid hydrocracking over “Ru/CeO2-alcohol thermal”; conditions: squalane/B. braunii liquid, 4 g; catalyst, 0.1 g; H2, 3.5/4 MPa; T, 513 K. | ||
If the oxygen compounds were the only effect factor in B. braunii liquid reaction process, the result of pre-treating B. braunii liquid should be similar to the result of B. braunii liquid-P; however, the result was not similar. As shown in Fig. 5c, the B. braunii liquid (pretreated by H2) conversion reached almost 100%. The yield of C8–C16 hydrocarbons was 24%, and the yield of C17–C20 hydrocarbons was 49%. All products in this reaction were stable saturated hydrocarbons and most of them were branched. Compared with Fig. 2b, the conversion of B. braunii liquid was significantly promoted. But compared with Fig. 2a, the distribution of products was not well: the content of C20–C25 hydrocarbons was about 25%. The molecular weight of products had obviously increased. These result means that there are some impurities in B. braunii liquid which could affect the products distribution.
The N element had no effect on the reaction, and the O element was removed by H2, the potential influential remaining elements in B. braunii liquid were only C and H. Therefore, the impurities, which could affect the products distribution, most probably were the pigments. The main pigment in B. braunii liquid was β-carotenoid, which molecular formula is C40H56. According to the results of squalane, the main catalytic products of β-carotenoid most probably were C20–C25 hydrocarbons.
In order to eliminate the effect of unsaturated bond, all the experience samples in this part were pretreated by H2. As shown in Fig. 6, when adding 0.1 g pigments (β-carotenoid) in squalane, the contents of C17–C20 and C21–C25 were increased and the content of C8–C16 was decreased, which was similar to the results of Fig. 5c.
According to the results of Kazuhisa Murata15 and Shin-ichi Oya,20 there are three methods, which could be used, to improve the distribution of products: increasing the pressure of H2, prolonging the reaction time and increasing the reaction temperature. As shown in Fig. 6, by prolonging the reaction time, the distribution of products changed. The contents of C17–C20 and C21–C25 were decreased and the content of C8–C16 was increased (above 40%). All products in this reaction were stable saturated hydrocarbons and most of them were also branched. The distribution of products was significantly improved. And the production of bio-jet fuel from B. braunii liquid over Ru/CeO2 was successfully achieved.
4. Conclusions
The conclusions of this study are as follows:(1) “Ru/CeO2-alcohol thermal” catalyst had an outstanding catalytic performance in the reaction of squalane. (2) The reduction of hydrogen caused by oxygen compounds was the main reason for the low activity of B. braunii liquid. (3) The pigments in B. braunii liquid were the main reason for the non-ideal distribution of products.
In order to solve the problems (low activity and non-ideal distribution), the B. braunii liquid was pretreated by H2, and the reaction time was prolonged to 8 hours. Finally, the content of C8–C16 in the products was up to 40%. The production of bio-jet fuel from B. braunii liquid over Ru/CeO2 catalyst was successfully achieved.
This study surely would provide an important reference and basis for the research of bio-jet fuel production from B. braunii.
Acknowledgements
The authors wish to express thanks for the supports from the National High Technology Research and Development 863 Program of China (grant no. 2013AA065804), the Fundamental Research Funds for the Central Universities (YS1407).References
- X. Sheng, N. Li, G. Li, W. Wang, J. Yang, Y. Cong, A. Wang, X. Wang and T. Zhang, Synthesis of high density aviation fuel with cyclopentanol derived from lignocellulose, Sci. Rep., 2015, 5, 9565 CrossRef CAS PubMed.
- I. T. D. Cabanelas, S. S. I. Marques, C. O. de Souza, J. I. Druzian and I. A. Nascimento, Botryococcus, what to do with it? Effect of nutrient concentration on biorefinery potential, Algal Res., 2015, 11, 43–49 CrossRef.
- J. Talukdar, M. C. Kalita, B. C. Goswami, D. D. Hong and H. C. Das, Liquid Hydrocarbon Production Potential of a Novel Strain of the Microalga Botryococcus braunii: Assessing the Reliability of in situ Hydrocarbon Recovery by Wet Process Solvent Extraction, Energy Fuels, 2014, 28, 3747–3758 CrossRef CAS.
- B. Moutel, M. André, D. Kucma, J. Legrand, D. Grizeau, J. Pruvost and O. Gonçalves, Assessing the biofuel production potential of Botryococcus braunii strains by sensitive rapid qualitative chemotyping using chemometrically-assisted gas chromatography-mass spectrometry, Algal Res., 2015, 11, 33–42 CrossRef.
- M. B. Tasić, L. F. R. Pinto, B. C. Klein, V. B. Veljković and R. M. Filho, Botryococcus braunii for biodiesel production, Renewable Sustainable Energy Rev., 2016, 64, 260–270 CrossRef.
- Y. Li, R. B. Moore, J. G. Qin, A. Scott and A. S. Ball, Extractable liquid, its energy and hydrocarbon content in the green alga Botryococcus braunii, Biomass Bioenergy, 2013, 52, 103–112 CrossRef CAS.
- J. Jin, et al., Characteristics of extracellular hydrocarbon-rich microalga Botryococcus braunii for biofuels production: Recent advances and opportunities, Process Biochem., 2016 DOI:10.1016/j.procbio.2015.11.026.
- J. J. Pan, J. O. Solbiati, G. Ramamoorthy, B. S. Hillerich, R. D. Seidel, J. E. Cronan, S. C. Almo and C. D. Poulter, Biosynthesis of Squalene from Farnesyl Diphosphate in Bacteria: Three Steps Catalyzed by Three Enzymes, ACS Cent. Sci., 2015, 1, 77–82 CrossRef CAS PubMed.
- H. R. Thapa, M. T. Naik, S. Okada, K. Takada, I. Molnar, Y. Xu and T. P. Devarenne, A squalene synthase-like enzyme initiates production of tetraterpenoid hydrocarbons in Botryococcus braunii Race L, Nat. Commun., 2016, 7, 11198 CrossRef CAS PubMed.
- S. Nagano, S. Yamamoto, M. Nagakubo, K. Atsumi and M. M. Watanabe, Physical Properties of Hydrocarbon Oils Produced by Botryococcus Braunii: Density, Kinematic Viscosity, Surface Tension, and Distillation Properties, Procedia Environ. Sci., 2012, 15, 73–79 CrossRef CAS.
- P. Hidalgo, G. Ciudad, S. Schober, M. Mittelbach and R. Navia, Biodiesel synthesis by direct transesterification of microalga Botryococcus braunii with continuous methanol reflux, Bioresour. Technol., 2015, 181, 32–39 CrossRef CAS PubMed.
- H. Watanabe, D. Li, Y. Nakagawa, K. Tomishige and M. M. Watanabe, Catalytic gasification of oil-extracted residue biomass of Botryococcus braunii, Bioresour. Technol., 2015, 191, 452–459 CrossRef CAS PubMed.
- H. Watanabe, D. Li, Y. Nakagawa, K. Tomishige, K. Kaya and M. M. Watanabe, Characterization of oil-extracted residue biomass of Botryococcus braunii as a biofuel feedstock and its pyrolytic behavior, Appl. Energy, 2014, 132, 475–484 CrossRef CAS.
- L. O. Garciano, N. H. Tran, G. S. K. Kannangara, A. S. Milev, M. A. Wilson and H. Volk, Developing saponite supported cobalt–molybdenum catalysts for upgrading squalene, a hydrocarbon from the microalgae Botryococcus braunii, Chem. Eng. Sci., 2014, 107, 302–310 CrossRef CAS.
- K. Murata, Y. Liu, M. M. Watanabe, M. Inaba and I. Takahara, Hydrocracking of Algae Oil into Aviation Fuel-Range Hydrocarbons Using a Pt–Re Catalyst, Energy Fuels, 2014, 28, 6999–7006 CrossRef CAS.
- G. Liu, B. Yan and G. Chen, Technical review on jet fuel production, Renewable Sustainable Energy Rev., 2013, 25, 59–70 CrossRef CAS.
- I.-H. Choi, K.-R. Hwang, J.-S. Han, K.-H. Lee, J. S. Yun and J.-S. Lee, The direct production of jet-fuel from non-edible oil in a single-step process, Fuel, 2015, 158, 98–104 CrossRef CAS.
- C. Zhang, X. Hui, Y. Lin and C.-J. Sung, Recent development in studies of alternative jet fuel combustion: progress, challenges, and opportunities, Renewable Sustainable Energy Rev., 2016, 54, 120–138 CrossRef CAS.
- W.-C. Wang and L. Tao, Bio-jet fuel conversion technologies, Renewable Sustainable Energy Rev., 2016, 53, 801–822 CrossRef CAS.
- S. Oya, D. Kanno, H. Watanabe, M. Tamura, Y. Nakagawa and K. Tomishige, Catalytic production of branched small alkanes from biohydrocarbons, ChemSusChem, 2015, 8, 2472–2475 CrossRef CAS PubMed.
- P. da Costa Zonetti, R. Landers and A. J. G. Cobo, Thermal treatment effects on the Ru/CeO2 catalysts performance for partial hydrogenation of benzene, Appl. Surf. Sci., 2008, 254, 6849–6853 CrossRef.
- J. Lin, L. Zhang, Z. Wang, J. Ni, R. Wang and K. Wei, The effect of Ag as a promoter for Ru/CeO2 catalysts in ammonia synthesis, J. Mol. Catal. A: Chem., 2013, 366, 375–379 CrossRef CAS.
- H. Kanda, P. Li, T. Yoshimura and S. Okada, Wet extraction of hydrocarbons from Botryococcus braunii by dimethyl ether as compared with dry extraction by hexane, Fuel, 2013, 105, 535–539 CrossRef CAS.
- J. Regalbuto, Catalyst preparation: science and engineering, CRC Press, 2016 Search PubMed.
- J. G. Speight, Fouling in Refineries, 2015 Search PubMed.
- M. Saajanlehto, P. Uusi-Kyyny and V. Alopaeus, Hydrogen solubility in heavy oil systems: experiments and modeling, Fuel, 2014, 137, 393–404 CrossRef CAS.
- F.-A. Khan, A. Vallat and G. Süss-Fink, Highly selective CC bond hydrogenation in α,β-unsaturated ketones catalyzed by hectorite-supported ruthenium nanoparticles, J. Mol. Catal. A: Chem., 2012, 355, 168–173 CrossRef CAS.
- H. Wang, S. Yan, S. O. Salley and K. Y. S. Ng, Support effects on hydrotreating of soybean oil over NiMo carbide catalyst, Fuel, 2013, 111, 81–87 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |