Spectroscopic study on spontaneously grown silver@ultra-thin cerium oxide nanostructures
Chi-Hang Tsaia,
Shih-Yun Chenb,
Jenn-Ming Song *c and
Alexandre Gloterd
*c and
Alexandre Gloterd
aGraduate Institute of Applied Science and Technology, National Taiwan University of Science and Technology, Taipei, Taiwan
bDepartment of Materials Science and Engineering, National Taiwan University of Science and Technology, Taipei, Taiwan
cDepartment of Materials Science and Engineering, National Chung Hsing University, Taichung, Taiwan. E-mail: samsong@nchu.edu.tw
dLaboratoire de Physique des Solides, Université Paris-Sud, Orsay, France
First published on 2nd December 2016
Abstract
Ag@CeO2 nanostructures have been recently reported to show unique catalytic properties but synthetic methods for them are limited. This study investigates microstructural characteristics of Ag@CeO2 nanowires and nanoparticles with a spontaneously-grown ultra-thin ceria shell (∼0.5 nm), which were synthesized on CeO2 substrate without using oxide precursors. Elemental mapping and line scanning by electron energy loss spectroscopy (EELS) suggest that the Ce in the CeO2 substrate dissolved in the molten silver nitrate salt and was repelled to the surface of the Ag nanostructures to form continuous oxide shells during the growth of Ag single-crystals. X-ray absorption near edge structure (XANES) spectra verify that the valence of the Ce ions in the oxide layer was between Ce3+ and Ce4+.
Among the common metal oxides, CeO2, which is regarded as a wide band gap semiconductor,1,2 has attracted much interest due to its unique properties.3,4 To enhance functionality and performance, core–shell nanostructured materials have recently been reported and developed, due to the fact that the properties of single material are sometimes limited. The combination of a Ag core and ceria shell is of interest. Ag@CeO2 core–shell nanostructures exhibit good catalytic ability and high chemical selectivity as well. For instance, Khan et al.5 investigated the photocatalytic performance of a Ag/CeO2 nanocomposite and demonstrated that it possesses superior visible light photocatalytic activity compared with pure CeO2. Zhang et al.6 successfully fabricated Ag@CeO2 NPs through hydrothermal method and found that after annealing at 500 °C for 5 h the catalytic properties of Ag@CeO2 NPs are superior to their single structure counterparts (either Ag or CeO2).
Previous works6–11 on the Ag@CeO2 core–shell structures are listed in Table 1. As tabulated, most of them deal with the synthesis of Ag@CeO2 nanoparticles (NPs) and only one study successfully prepared one-dimensional Ag@CeO2. Mondragón-Galicia et al.11 developed the synthesis the Ag@CeO2 nanotubes through sol–gel method. In this method, a hollow structure is formed by reacting the Ag nanowires (NWs) with ammonium hydroxide followed by the migration of Ag atoms. During the heating process, outer-diffusion of the remnant Ag through the interface is faster than the inter-diffusion of the CeO shell, which eventually gives rise to a hollow Ag@CeO2. With respect to Ag@CeO2 nanowires, the relevant reports are quite rare. To have a simple yet effective approach to prepare the Ag@CeO2 nanostructures with adjustable morphologies and dimensions would be a great challenge.
| Metal@oxide | Method | Surfactant (S), precipitation (P) | Size | Core configuration | Precursor |
|---|---|---|---|---|---|
| a Igepal CO-520 (polyoxyethylene nonylphenyl ether), PVP (polyvinylpyrrolidone). | |||||
| Ag@CeO2 NPs6 | Hydro-thermal | PVP (S) | Ag 18.3 nm | Single Ag | Ce(NO3)3·6H2O |
| CeO2 6.2 nm | |||||
| Ag@CeO2 NPs7,8 | Coprecipitation | Ammonia (P) | Ag 28 nm | Ag agglomeration | Ce(NO3)3·6H2O |
| CeO2 14 nm | |||||
| Ag@CeO2 NPs9,10 | Reverse micelle | Igepal CO-520 (S) | Ag 10 nm | Ag agglomeration | Ce(NO3)3·6H2O |
| Cyclohexane (S) | CeO2 3 nm | ||||
| Ag@CeO2 NTs11 | Sol–gel | PVP (S) | Agcore 49 nm | Ag hollow | Ce(NO3)3·6H2O |
| CeO2 shell 78 nm | |||||
This study synthesized Ag@CeO2 nanostructures grown on CeO2 thin film, including nanoparticles and nanowires, with the absence of oxide precursors, templates, and surfactants. Microstructural characteristics of the core–shell nanostructures were studied by TEM, electron energy loss spectroscope (EELS) and X-ray absorption near edge structure (XANES). The growth mechanisms were proposed and discussed.
CeO2 thin film with the thickness of 720 nm was fabricated through sol gel method by dipping Si wafer substrate into the gel, for which the solution was prepared from CeCl3O·7H2O, citric acid, and ethanol with a volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5
2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 and stirred for 10 min before aging at room temperature (20 °C) for 2 days. It was then spun at 1000 rpm for 30 s. To achieve well crystallization, annealing of CeO2 films was performed at 500 °C for 8 h in an oxygen atmosphere. Silver nitrate (AgNO3) was used as the precursor to grow Ag@CeO2 nanostructures. In order to obtain Ag@CeO2 NPs, 15 μl of 0.05 M aqueous AgNO3 was dropped onto the UV light-exposed (48 h) CeO2 film, while the Ag@CeO2 NWs was synthesized by dropping the salt solution onto non-UV-exposed CeO2 film. Afterward, the samples were isothermally heated in air at 300 °C for 3 h using an infrared furnace, and cooled down to ambient temperature.
50 and stirred for 10 min before aging at room temperature (20 °C) for 2 days. It was then spun at 1000 rpm for 30 s. To achieve well crystallization, annealing of CeO2 films was performed at 500 °C for 8 h in an oxygen atmosphere. Silver nitrate (AgNO3) was used as the precursor to grow Ag@CeO2 nanostructures. In order to obtain Ag@CeO2 NPs, 15 μl of 0.05 M aqueous AgNO3 was dropped onto the UV light-exposed (48 h) CeO2 film, while the Ag@CeO2 NWs was synthesized by dropping the salt solution onto non-UV-exposed CeO2 film. Afterward, the samples were isothermally heated in air at 300 °C for 3 h using an infrared furnace, and cooled down to ambient temperature.
The nanostructures thus synthesized were removed from CeO2 substrate in alcohol by ultrasonic oscillation for investigation. Phase identification was carried out using X-ray diffraction (XRD, D8 Discover X-ray diffractometer) (incidence angle of 0.3°) with graphite monochromatic Cu Kα radiation (λ = 0.15418 nm) at a scanning rate of 2° per minute from 20° to 70°. The structure and composition distribution of the Ag@CeO2 NPs and NWs were characterized using a transmission electron microscope (TEM). Scanning transmission electron microscopy/electron energy loss spectroscopy (STEM/EELS) was done using a USTEM-NION microscope (USTEM200) and a GATAN EELS modified spectrometer. In order to limit the damage, the microscope was operated at 100 keV.
The X-ray absorption near edge structure (XANES) spectroscopy measurements at the Ce L3-edge were performed using Wigger beamline 17C at National Synchrotron Radiation Research Center (NSRRC), Taiwan, at room temperature, for which the monochromator Si (111) crystals were used. The energy resolution at the Ce L3-edge (5723 eV) was about 0.4 eV.
Fig. 1(a) and (b) show the Ag nanostructures grown on CeO2 substrate. Ag NPs thus produced were with an average diameter of 18 ± 6 nm, while the length and diameter of Ag NWs were 7 μm and 100 ± 9 nm respectively. XRD patterns given in Fig. 1(c) depict that only the diffraction peaks of Ag could be detected. The growth mechanism of zero and one-dimensional Ag on oxide substrates has been proposed elsewhere.12,13 The shape and dimensions of nanocrystals are closely related to the degree of excitation of the oxide substrate by UV exposure, and the amount of photo-induced defects. In brief, UV-excited oxide substrates, e.g. TiO2 and CeO2, can reduce metallic ions to form metallic nanostructure. The reduction of metallic ions is related to the photo-induced defects. Take TiO2 for example, the reduction mechanism of metallic ions are proposed as follows.
 | (1) |
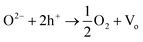 | (2) |
| Ti4+ + e− → Ti3+ | (3) |
| M+ + Ti3+ → M + Ti4+ | (4) |
Fig. 2(a) shows the HRTEM images of an as-synthesized Ag NP, depicting a lattice spacing of 0.23 nm, which could be referred to {111} planes of Ag. Diffraction pattern obtained by Fourier transform was shown in Fig. 2(b). There is only one set of reciprocal lattices, indicating that the Ag nanoparticle is a single crystal. EELS mapping in Fig. 2(c) and elemental line scanning in Fig. 2(d) demonstrate the presence of a very thin shell layer on Ag NPs comprising Ce and O, of which the thickness was about 0.5 nm. The fact that only the lattice fringes of Ag can be observed brings us to believe that the oxide shell was amorphous or with a poor degree of crystallization. Fig. 2(d) further pinpoints the location of O and Ce, which was at the edge of the Ag signals, again verifying the core–shell structure. Elemental line scanning also shows that the oxygen signal was overlapped with Ce but much narrower and close to the outer surface. Our previous study14 suggested that this could be ascribed to the inward diffusion of oxygen and subsequent oxidation.
Fig. 3(a) reveals that the TEM image of a Ag NW and the selected-area electron diffraction pattern, which confirm that the Ag NWs were single-crystalline and grew towards [1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0]. Inserted EDS spectrum verifies the purity of Ag NWs. The EELS mapping (Fig. 3(b)), as well as the elemental line-scanning (Fig. 3(c)), shows that the NWs also exhibited a metal@oxide core–shell structural feature. The ultra-thin spontaneously-grown shell which enriched with Ce and O is shown to be continuous, compact and well attached to the NW surface. The overlapping of Ce and O signals and the deviation in the distribution are identical to those observed in NPs.
0]. Inserted EDS spectrum verifies the purity of Ag NWs. The EELS mapping (Fig. 3(b)), as well as the elemental line-scanning (Fig. 3(c)), shows that the NWs also exhibited a metal@oxide core–shell structural feature. The ultra-thin spontaneously-grown shell which enriched with Ce and O is shown to be continuous, compact and well attached to the NW surface. The overlapping of Ce and O signals and the deviation in the distribution are identical to those observed in NPs.
Fig. 4 presents the normalized XANES spectra of the Ce L3-edge for the Ag@CeO2 NPs and NWs. The comparison with the spectra obtained from CeF3 and CeO2 NPs as the standards are also made. Apparently, the peak at low energy side (around 5730 eV) for both Ag@CeO2 NPs and NWs differs from the standards. In order to estimate the charge state of Ce, all the spectra are subtracted by an arctangent function to exclude the electronic transition to continuum states, and fitted with five Gaussian functions.15–17 In this respect, the deconvoluted components of a representative example for standard CeO2 NPs were illustrated.18–20 Component A arises from a core excited Ce4+ final state with the configuration 2p4f05d*, where 2p denotes a hole in the 2p shell and 5d* refers to the excited electron in the 5d state. The split of component A into two sub peak A1 and A2 is caused by the crystal-field splitting. Component B is attributed mainly to a 2p4f15d* state, where the asterisk stands for a hole in the anion ligand orbital. Component C was assigned as Ce3+, which is transition from the initial electron configuration 2p4f15d0 to the final configuration 2p4f15d*. As for the feature D, it is due to the dipole-forbidden 2p-to-4f transition. The concentration of Ce3+ can be expressed as the ratio of Ic to Itotal, where Ic refers to the intensity of component C and Itotal is the summation of intensities of A, B, C, and D.17 As to Ag@CeO2 NPs and NWs, the calculated ratio of Ic/Itotal was 17 and 20%, respectively, which is much greater than that of reference sample, CeO2 (Ce3+ is ∼8%). Accordingly, it can be inferred that the charge state of Ce in Ag@CeO2 NPs and NWs is between Ce3+ and Ce4+.
Unlike the formation of CeO2 shell on Ag nanoparticles thorough an additive process, e.g. localized surface plasmon resonance photochemical induced interface reaction,21 the growth of thin oxide shell occurs simultaneously with the crystallization of Ag nanostructures in our case. It can be deduced that the segregation of Ce at the Ag surface may take place through the following route. As illustrated in Fig. 5, Ce from CeO2 film dissolved into molten AgNO3 salt during isothermal heating at 300 °C (the melting point of AgNO3 is 212 °C). After that, dissolved Ce in molten salt was repelled to the surface of NPs or NWs during the crystal growth of Ag and then got oxidized. To clarify this, the solubility of Ce in molten AgNO3 salt heated at 300 °C for 3 h was experimentally obtained by measuring the Ce and Ag concentrations dissolved in nitric acid aqueous solution using inductively coupled plasma-mass spectrometry. The measured Ce/Ag value (1.6 wt%), which is closely to the one estimated from the ratio of ceria shell thickness to the Ag wire radius (1.9 wt%), supports our speculation.
 | ||
| Fig. 5 Schematic illustration of proposed mechanism of for the spontaneous formation of thin-oxide shell on Ag nanostructure. | ||
In summary, the microstructural features of Ag@CeO2 core–shell nanostructures by a one-step synthesis method were investigated in this study. Spontaneously-grown CeO2 shell is found to cover the whole surface of nanoparticles or nanowires of Ag. Experimental results suggest that the CeO2 shell is formed by repelling and oxidation of dissolved Ce from the substrate during Ag single-crystal growth. The XAS spectra suggest that the valence of Ce ions in this spontaneous oxide layer is in between Ce3+ and Ce4+.
Acknowledgements
This work was supported primarily by Ministry of Science and Technology of R.O.C. through contracts No. MOST 103-2221-E-005-018 and MOST 104-2119-M-006-012, from which the authors are grateful.Notes and references
- M. M. Khan, S. A. Ansari, D. Pradhan, D. H. Han, J. Lee and M. H. Cho, Ind. Eng. Chem. Res., 2014, 53, 9754 CrossRef CAS.
- S. A. Ansari, M. M. Khan, M. O. Ansari, S. Kalathil, J. Lee and M. H. Cho, RSC Adv, 2014, 4, 16782 RSC.
- M. M. Khan, S. F. Adil and A. Al-Mayouf, J. Saudi Chem. Soc., 2015, 19, 462 CrossRef.
- M. M. Khan, S. A. Ansari, M. O. Ansari, B. K. Min, J. Lee and M. H. Cho, J. Phys. Chem. C, 2014, 118, 9477 CAS.
- M. M. Khan, S. A. Ansari, J. Y. Lee, M. O. Ansari, J. Lee and M. H. Cho, J. Colloid Interface Sci., 2011, 431, 255 CrossRef PubMed.
- J. Zhang, L. Li, X. Huang and G. Li, J. Mater. Chem., 2012, 22, 10480 RSC.
- T. Kayama, K. Yamazaki and H. J Shinjoh, J. Am. Chem. Soc., 2010, 132, 13154 CrossRef CAS PubMed.
- K. Yamazaki, T. Kayama, F. Dong and H. Shinjoh, J. Catal., 2011, 282, 289 CrossRef CAS.
- T. Mitsudome, Y. Mikami, M. Matoba, T. Mizugaki, K. Jitsukawa and K. Kaneda, Angew. Chem., Int. Ed., 2012, 51, 136 CrossRef CAS PubMed.
- T. Mitsudome, M. Matoba, T. Mizugaki, K. Jitsukawa and K. Kaneda, Chem.–Eur. J., 2013, 19, 5255 CrossRef CAS PubMed.
- G. Mondragón-Galicia, R. Pérez-Hernández, C. E. Gutiérrez-Wing and D. Mendoza-Anaya, Phys. Chem. Chem. Phys., 2011, 13, 16756 RSC.
- H. T. Tung, I. G. Chen, J. M. Song and C. W. Yen, J. Mater. Chem., 2009, 19, 2386 RSC.
- J. M. Song, S. Y. Chen, Y. L. Shen, C. H. Tsai, S. W. Feng, H. T. Tung and I. G. Chen, Appl. Surf. Sci., 2013, 285, 450 CrossRef CAS.
- C. H. Tsai, S. Y. Chen, J. M. Song and A. Gloter, Chem. Commun., 2015, 51, 16825 RSC.
- L. Douillard, M. Gautier, N. Thromat, M. Hentriot, M. J. Guittet, J. P. Duraud and G. Tourillon, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 49, 16171 CrossRef CAS.
- Z. Hu, S. Bertram and G. Kaindl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 49, 39 CrossRef CAS.
- G. Kaindl, G. Schmiester and E. V. Sampathkumaran, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 38, 10174 CrossRef CAS.
- P. Nachimuthu, W. C. Shih, R. S. Liu, L. Y. Jang and J. M. Chen, J. Solid State Chem., 2000, 149, 408 CrossRef CAS.
- R. A. Neifeld, M. Croft, T. Mihalisin, C. U. Segre, M. Madigan, M. S. Torikachvili, M. B. Maple and L. E. DeLong, Phys. Rev. B: Condens. Matter Mater. Phys., 1985, 32, 6928 CrossRef CAS.
- A. V. Tsvyashchenko, L. N. Fomicheva, A. A. Sorokin, G. K. Ryasny, B. A. Komissarova, L. G. Shpinkova, K. V. Klementiev, A. V. Kuznetsov, A. P. Menushenkov, V. N. Trofimov, A. E. Primenko and R. Cortes, Phys. Rev. B: Condens. Matter Mater. Phys., 2002, 65, 174513 CrossRef.
- H. X. Zhong, Y. Wei, Y. Z. Yue, L. H. Zhang and Y. Liu, Nanotechnology, 2016, 27, 135701 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |




