Monitoring and quantification of the complex bioaccumulation process of mercury ion in algae by a novel aggregation-induced emission fluorogen†
Abstract
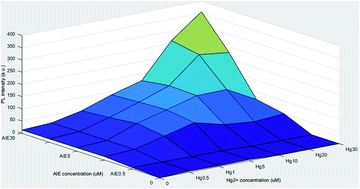
In this study, a novel methodology was developed using a specified aggregation-induced emission fluorogen (AIEgen) to monitor and quantify the complex bioaccumulation process in a microcosm aquatic ecosystem. Mercury ion (Hg2+) was used as the pollutant and Euglena gracilis as a representative algal species in water, to develop this new methodology for understanding the processes of bioaccumulation and biorelease of a heavy metal in algae. AIEgen can easily detect Hg2+ in the environment by the “turn-on” feature, and a relationship was built among photoluminescence (PL) intensity, AIEgen concentration, and Hg2+ concentration. The AIEgen was effectively used for quantifying Hg2+ concentration in the bioaccumulation process by reading the PL intensity of the solution. Bioaccumulation, bioaccumulation efficiency, and the ratio of Hg2+ in Euglena gracilis cells and the environment were carefully characterized by this novel method and the results were further validated with the existing well-established analytical method. The quantitative detection of Hg2+ absorption and release from the algae by the AIEgen demonstrates a novel, green, and sustainable approach to understand the dynamics of Hg2+ between aquatic organisms and the environment.


 Please wait while we load your content...
Please wait while we load your content...